Abstract
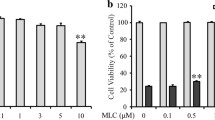
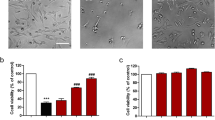
Oxidative stress is considered as one of key factors related to Alzheimer’s disease (AD), while kinetin (KT) has been reported to exert anti-oxidative activities as well as neuroprotective effects both in vivo and in vitro. Thus, in this study, the neuroprotective effects of KT against glutamate-induced oxidative toxicity in HT22 cells were investigated. To evaluate the anti-oxidative capabilities of KT itself, several anti-oxidative assays in vitro were conducted. To evaluate the neuroprotective effects of KT, the levels of intracellular reactive oxygen species (ROS) and calcium influx, mitochondrial membrane potential (MMP), and cell death were measured by flow cytometry. Nuclear translocation of apoptosis inducing factor (AIF) and content of intracellular ATP were also determined. In addition, the phosphorylation levels of apoptosis signal-regulating kinase 1 (ASK-1), c-Jun N-terminal kinase (JNK), and p38 mitogen-activated protein kinases (p38) were evaluated as well. Besides, nuclear translocation of nuclear factor-E2-related factor 2 (Nrf2) and the expression of heme oxygenase-1 (HO-1) were also examined to reveal underlying mechanisms. Results showed that KT rescued cell death, and suppressed the accumulation of intracellular ROS and the increase of intracellular calcium influx. In addition, KT maintained normal function of mitochondria and inhibited the phosphorylation of ASK-1, JNK, and p38. KT also promoted nuclear translocation of Nrf2 and enhanced the expression of HO-1 both at protein and mRNA level. Importantly, blockage of Nrf2 almost completely abolished the neuroprotective effects of KT, while blockage of HO-1 expression partly neutralized its neuroprotective effects. Our results indicated that KT can protect HT22 cells from glutamate-induced cell death by activating Nrf2 pathway and inducing expression of HO-1, suggesting KT might be a drug candidate for treatment of AD and other neurodegenerative disorders related to oxidative stress.









Similar content being viewed by others
References
Albrecht P, Lewerenz J, Dittmer S, Noack R, Maher P, Methner A (2010) Mechanisms of oxidative glutamate toxicity: the glutamate/Cystine antiporter system x(c)(−) as a neuroprotective drug target CNS Neurol Disord-Drug. Targets 9:373–382
Barciszewski J, Siboska GE, Pedersen BO, Clark BFC, Rattan SIS (1997a) Furfural, a precursor of the cytokinin hormone kinetin, and base propenals are formed by hydroxyl radical damage of DNA. Biochem Biophys Res Commun 238:317–319. https://doi.org/10.1006/bbrc.1997.7315
Barciszewski J, Siboska GE, Pedersen BO, Clark BFC, Rattan SIS (1997b) A mechanism for the in vivo formation of N-6-furfuryladenine, kinetin, as a secondary oxidative damage product of DNA. FEBS Lett 414:457–460. https://doi.org/10.1016/s0014-5793(97)01037-5
Barciszewski J, Rattan SIS, Siboska G, Clark BFC (1999) Kinetin—45 years on Plant Sci 148:37-45 doi:https://doi.org/10.1016/s0168-9452(99)00116-8
Barciszewski J, Massino F, BFC C (2007) Kinetin—a multiactive molecule. Int J Biol Macromol 40:182–192. https://doi.org/10.1016/j.ijbiomac.2006.06.024
Barone E (2016) Oxidative stress and Alzheimer disease: where do we stand? Curr Alzheimer Res 13:108–111. https://doi.org/10.2174/156720501302160101123849
Breyer A, Elstner M, Gillessen T, Weiser D, Elstner E (2007) Glutamate-induced cell death in neuronal HT22 cells is attenuated by extracts from St John’s wort (Hypericum perforatum L). Phytomedicine 14:250–255. https://doi.org/10.1016/j.phymed.2007.02.001
Buendia I, Michalska P, Navarro E, Gameiro I, Egea J, Leon R (2016) Nrf2-ARE pathway: an emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol Ther 157:84–104. https://doi.org/10.1016/j.pharmthera.2015.11.003
Celik I, Tuluce Y (2006) Effects of indoleacetic acid and kinetin on lipid peroxidation and antioxidant defense in various tissues of rats. Pest Biochem Physiol 84:49–54. https://doi.org/10.1016/j.pestbp.2005.05.004
Cooper AJL, Jeitner TM (2016) Central role of glutamate metabolism in the maintenance of nitrogen homeostasis in normal and hyperammonemic brain. Biomolecules:6. https://doi.org/10.3390/biom6020016
Dong J, Cai L, Zhu X, Huang X, Yin T, Fang H, Ding Z (2014) Antioxidant activities and phenolic compounds of cornhusk, Corncob and Stigma Maydis. J Braz Chem Soc 25:1956–1964. https://doi.org/10.5935/0103-5053.20140177
Elphick LM, Hawat M, Toms NJ, Meinander A, Mikhailov A, Eriksson JE, GEN K (2008) Opposing roles for caspase and calpain death proteases in L-glutamate-induced oxidative neurotoxicity. Toxicol Appl Pharmacol 232:258–267. https://doi.org/10.1016/j.taap.2008.07.008
Esteras N, Dinkova-Kostova AT, Abramov AY (2016) Nrf2 activation in the treatment of neurodegenerative diseases: a focus on its role in mitochondrial bioenergetics and function. Biol Chem 397:383–400. https://doi.org/10.1515/hsz-2015-0295
Filpa V, Moro E, Protasoni M, Crema F, Frigo G, Giaroni C (2016) Role of glutamatergic neurotransmission in the enteric nervous system and brain-gut axis in health and disease. Neuropharmacology 111:14–33. https://doi.org/10.1016/j.neuropharm.2016.08.024
Fujisawa T, Ichijo H (2014) ASK1-MAP kinase signaling pathway as a therapeutic target for human diseases. Nihon rinsho Jpn J Clin Med 72:957–965
Fukui M, Song JH, Choi JY, Choi HJ, Zhu BT (2009) Mechanism of glutamate-induced neurotoxicity in HT22 mouse hippocampal cells. Eur J Pharmacol 617:1–11. https://doi.org/10.1016/j.ejphar.2009.06.059
Huang YD, Mucke L (2012) Alzheimer Mechanisms and Therapeutic Strategies. Cell 148:1204–1222. https://doi.org/10.1016/j.cell.2012.02.040
Ichijo H et al (1997) Induction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science 275:90–94. https://doi.org/10.1126/science.275.5296.90
Jablonska-Trypuc A, Matejczyk M, Czerpak R (2016) N6-Benzyladenine and kinetin influence antioxidative stress parameters in human skin fibroblasts. Mol Cell Biochem 413:97–107. https://doi.org/10.1007/s11010-015-2642-5
Johnson DA, Johnson JA (2015) Nrf2—a therapeutic target for the treatment of neurodegenerative diseases. Free Radic Biol Med 88:253–267. https://doi.org/10.1016/j.freeradbiomed.2015.07.147
Kanninen K et al (2009) Intrahippocampal injection of a lentiviral vector expressing Nrf2 improves spatial learning in a mouse model of Alzheimer's disease. Proc Natl Acad SciU S A 106:16505–16510. https://doi.org/10.1073/pnas.0908397106
Kelebek H, Selli S, Kadiroglu P, Kola O, Kesen S, Ucar B, Cetiner B (2017) Bioactive compounds and antioxidant potential in tomato pastes as affected by hot and cold break process. Food Chem 220:31–41. https://doi.org/10.1016/j.foodchem.2016.09.190
Lewerenz J, Maher P (2015) Chronic glutamate toxicity in neurodegenerative diseases—what is the evidence? Front Neurosci 9. https://doi.org/10.3389/fnins.2015.00469
Li MY, Ouyang WQ, Wu XL, Zheng Y, Gao R, Ouyang X, An L (2014a) Kinetin protects D-galactose-induced aging rats from splenic oxidative damage. Pak Vet J 34:386–390
Li MY, Ouyang WQ, Wu XL, Zheng Y, Wei YP, An L (2014b) Kinetin inhibits apoptosis of aging spleen cells induced by D-galactose in rats. J Vet Sci 15:353–359. https://doi.org/10.4142/jvs.2014.15.3.353
Li MY, Ouyang WQ, Li J, Si LF, Li X, Guo JJ, Li HF (2016) Effects of kinetin on thymus and immune function of aging Rats. Pak Vet J 36:356–362
Liu YM, Zhang ZQ, Yang XF (2011) Kinetin protects against lipid peroxidation and improves antioxidant status in cultured astrocytes and mouse brain exposed to D-galactose. Afr J Biotechnol 10:11721–11727
Luca M, Luca A, Calandra C (2015) The role of oxidative damage in the pathogenesis and progression of Alzheimer's disease and vascular dementia. Oxid Med Cell Longev. https://doi.org/10.1155/2015/504678
Minorsky PV (2003) Kinetin: the elixir of life? Plant Physiol 132:1135–1136. https://doi.org/10.1104/pp.900083
Orrenius S, Gogvadze V, Zhivotovsky B (2015) Calcium and mitochondria in the regulation of cell death. Biochem Biophys Res Commun 460:72–81. https://doi.org/10.1016/j.bbrc.2015.01.137
Prasad KN (2016) Simultaneous activation of Nrf2 and elevation of antioxidant compounds for reducing oxidative stress and chronic inflammation in human Alzheimer's disease. Mech Ageing Dev 153:41–47. https://doi.org/10.1016/j.mad.2016.01.002
Ramsey CP et al (2007) Expression of Nrf2 in neurodegenerative diseases. J Neuropathol Exp Neurol 66:75–85. https://doi.org/10.1097/nen.0b013e31802d6da9
Rattan SIS, Clark BFC (1994) Kinetin delays the onset of aging characteristics in human fibroblasts. Biochem Biophys Res Commun 201:665–672. https://doi.org/10.1006/bbrc.1994.1752
Rosales-Corral S, Tan DX, Manchester L, Reiter RJ (2015) Diabetes and Alzheimer disease, Two overlap** pathologies with the same background: oxidative stress. Oxid Med Cell Longev. https://doi.org/10.1155/2015/985845
Sakauchi C, Wakatsuki H, Ichijo H, Hattori K (2016) Pleiotropic properties of ASK1. Biochim Biophys Acta 1861:3030–3038. https://doi.org/10.1016/j.bbagen.2016.09.028
Sharma SP, Kaur J, SIS R (1997) Increased longevity of kinetin-fed Zaprionus fruitflies is accompanied by their reduced fecundity and enhanced catalase activity. Biochem Mol Biol Int 41:869–875
Son Y, Byun SJ, Pae HO (2013) Involvement of heme oxygenase-1 expression in neuroprotection by piceatannol, a natural analog and a metabolite of resveratrol, against glutamate-mediated oxidative injury in HT22 neuronal cells. Amino Acids 45:393–401. https://doi.org/10.1007/s00726-013-1518-9
Song J, Park KA, Lee WT, Lee JE (2014) Apoptosis signal regulating kinase 1 ( ASK1): potential as a therapeutic target for Alzheimer's disease. Int J Mol Sci 15:2119–2129. https://doi.org/10.3390/ijms15022119
Sun JH, **ang GQ, Liu YM, Cao T, Li MY, Shang YH, Ouyang WQ (2012) Protective Effect of Kinetin on Iso-Induced Myocardial Ischemia in vitro and in vivo. J Anim Vet Adv 11:4617–4624
Tang LG, Ouyang WQ, Yang DY, Liu YJ, Chen H (2012) The effect of maize plumule extracts on rat manunary gland hyperplasia. J Anim Vet Adv 11:1995–1999
Wang Y, Wang Z, Handa CL, Xu J (2017) Effects of ultrasound pre-treatment on the structure of beta-conglycinin and glycinin and the antioxidant activity of their hydrolysates. Food Chem 218:165–172. https://doi.org/10.1016/j.foodchem.2016.09.069
Wanitphakdeedecha R, Meeprathom W, Manuskiatti W (2015) Efficacy and safety of 0.1% Kinetin cream in the treatment of photoaging skin. Indian J Dermatol Venereol Leprol 81:547. https://doi.org/10.4103/0378-6323.157446
Yamazaki H, Tanji K, Wakabayashi K, Matsuura S, Itoh K (2015) Role of the Keap1/Nrf2 pathway in neurodegenerative diseases. Pathol Int 65:210–219. https://doi.org/10.1111/pin.12261
Yan Y, Wei CL, Zhang WR, Cheng HP, Liu J (2006) Cross-talk between calcium and reactive oxygen species signaling. Acta Pharmacol Sin 27:821–826. https://doi.org/10.1111/j.1745-7254.2006.00390.x
Acknowledgements
This work was supported by Major Science and Technology Innovation Special Fund of Shaanxi Province (no. K332020916) and Science and Technology Co-ordination Innovation Project of Shaanxi Province (no. K3310216076).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wei, Y., Liu, D., Zheng, Y. et al. Neuroprotective Effects of Kinetin Against Glutamate-Induced Oxidative Cytotoxicity in HT22 Cells: Involvement of Nrf2 and Heme Oxygenase-1. Neurotox Res 33, 725–737 (2018). https://doi.org/10.1007/s12640-017-9811-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-017-9811-0