Abstract
Systemic acquired resistance protects plants against a broad spectrum of secondary infections by pathogens. A crucial compound involved in the systemic spread of the threat information after primary pathogen infection is the C9 oxylipin azelaic acid (AZA), a breakdown product of unsaturated C18 fatty acids. AZA is generated during lipid peroxidation in the plastids and accumulates in response to various abiotic and biotic stresses. AZA stimulates the expression of AZELAIC ACID INDUCED1 (AZI1), and a pool of AZI1 accumulates in the plastid envelope in association with AZA. AZA and AZI1 utilize the symplastic pathway to travel through the plasmodesmata to neighbouring cells to induce systemic stress resistance responses in distal tissues. Here, we describe the synthesis, travel and function of AZA and AZI1 and discuss open questions of signal initiation and propagation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Metabolites and proteins involved in systemic resistance
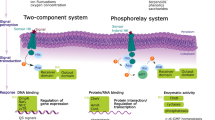
Local infections by pathogens cause resistance in distal tissues and protects them against subsequent pathogen attacks. The resistance in the whole plant is known as systemic acquired resistance (SAR) (Vlot et al. 2021). The resistance in the distal tissue requires mobile signal molecules which are generated at the local infection site and transported to uninfected distal tissues either through the plant body itself or via the air as volatiles (Kachroo and Kachroo 2020; Shine et al. Movement of small proteins or metabolites to systemic tissues occurs often via the phloem (Dinant and Lemoine 2010). Uploading of AZI1 to the phloem occurs via the symplastic transport (Lim et al. 2016) and the protein reaches the phloem via sorting signals which direct it from the outer plastid membrane to the endoplasmic reticulum and plasmodesmata which transverse the cell wall and join the adjacent cells. Being a lipid-binding and membrane-bound protein, AZI1 appears to travel to the phloem companion cells via direct membrane–membrane contact sites, which have been identified at the outer plastid membrane, the endoplasmatic reticulum and the plasma membrane. In case of lipid transfer proteins such as AZI1 these contact sites also allow exchange of the bound lipids (Breuers et al. 2011; Wang and Benning 2012; Helle et al. 2013, Cecchini et al. 2015). Lim et al. (2016) demonstrated that two plasmodesmata-localized proteins regulated SAR function in both, signalling and plasmodesmata gating of AZI1. While PLASMODESMATA LOCALIZING PROTEIN1 (PDLP1) interacts with AZI1, is required for endoplasmatic reticulum-specific localization of AZI1, and contributes to the intracellular portioning of the protein, PDLP5, which impairs plasmodesmata permeability and thus transport of AZA to the neighboring cell. PDLP1 interacts with PDLP5 which regulates the symplastic transport and plasmodesmata gating (Lee et al. 2011; Lim et al. 2016). PDLP5 knockout mutants increase and overexpressor lines restrict general plasmodesmata permeability (Lee et al. 2011; Wang et al. 2013). Importantly, the pdlp1 mutants contained reduced AZI1-GFP protein levels, although the azi1-gfp transcript levels were not affected (Lim et al. 2016). This suggest that PDLP1 affect the stability of AZI1. Furthermore, in the pdlp1 mutant, AZI1 was primarily localized to the outer plastid membrane, whereas in wild-type plants, the majority of the protein is located in extraplastidic compartments. The studies by Lim et al. (2016) highlight the importance of PDLP1 for AZI1 stability and its traveling from the plastids to the plasmodesmata. Jung et al. (2009) showed that AZI1 is important for generating vascular sap that confers disease resistance. AZA and petioles exudates failed to induce systemic immunity in azi1 plants. Pathogen-induced exudates from azi1 were inactive when applied to wild-type plants. Therefore, AZI1 modulates production and/or translocation of a mobile signal(s) during SAR. The AZI1 target in the vascular sap is unknown so far. AZI1 expression is stimulated by exogenously applied AZA (Jung et al. 2009). Whether stimulation of the endogenous AZA levels due to lipid peroxidation under stress in planta is a prerequisite for the AZI1 expression is not known. The best studied biological stimulus for AZI1 activation comes from Arabidopsis leaf infiltration assays with Pseudomonas syringae (cf. Arabidopsis eFP Browser). Similarly effective is the bacterial effector flg22. Late stimulation of AZI1 expression was also observed after co-cultivation of Arabidopsis seedlings with Hyaloperonospora arabidopidis (cf. Arabidopsis eFP Browser). Relatively little is known about the role of AZI1 for other pathogenic or beneficial plant–microbe interactions, including pathogenic fungi, nematodes, insects, mycorrhizal fungi and beneficial endophytes, although some of them can produce AZA. For instance, AZA is produced by P. syringae (Javvadi et al. 2018) or the root colonizing endophytic fungus Piriformospora indica (Kundu et al. 2022), however, whether microbe-synthesized AZA activates AZI1 in plants or participates in defense priming in plants, is not known. Besides biotic stress, expression profiles demonstrate that the AZI11 mRNA level responds also to abiotic stress, however functional analyses are often missing. Xu et al. (2011) showed that the AZI1 transcript level, as well as that of its paralog EARLI1 (Zhang and Schäppi 2007), increases after exposure of Arabidopsis seedlings to cold. The increase of the AZI1 mRNA level was slow, since more than 6 h at 4 °C was required for the induction. The mRNA level declined to basal levels when the plants were transferred back to room temperatures. Overexpression of AZI1 resulted in reduced electrolyte leakage during freezing damage, while AZI1 knockdown and knockout lines showed increased tendencies in cellular damage after freezing treatment. When Saccharomyces cerevisiae cells were transformed with AZI1 under the control of GAL1 promoter, the survival rate of yeast cells harbouring AZI1 increased after freezing treatment. This demonstrates that AZI1 might be multifunctional and associated with cold tolerance of Arabidopsis (Xu et al. 2011). The involvement of AZI1 in cold stress adaptation is further supported by expression profiling of mutants manipulated in cold stress-acclimation genes. Similar results were obtained for Thelunsiella salsuginea (Wong et al. 2006). The ICE-CBF-COR (Inducer of CBF Expression—C-repeat Binding Factor—Cold Regulated) signalling pathway is an important regulator for cold-stress acclimation (Gusain et al. 2023). CBF overexpressors show increased cold tolerance and high levels of AZI1 gene expression (Wong et al. 2006). Likewise, DEHYDRATION-RESPONSIVE-ELEMENT-BINDING PROTEIN1 (DREB1) genes are induced by cold stress, and overexpression of DREB1 induced strong expression of other stress-responsive genes, resulting in increased tolerance to high-salt and freezing stresses (Ito et al. 2006). Among the genes which are up-regulated in the DREB1 overexpressor lines after exposure to cold stress is AZI1 (Maruyama et al. 2004). Besides cold, AZI1 is involved in salinity stress tolerance. Pitzschke et al. (2014b) showed that azi1 mutants are hypersensitive to salt. At 150 mM salt stress, only 7% of the azi1 mutant seeds, 70% of wild-type seeds, and 90% of the seeds of AZI1 overexpressor lines germinated. Furthermore, AZI1 overexpressors thrived better than the azi1 mutants under high salt conditions. Another example provides mutants in with the salt stress signalling gene ZINC FINGER OF ARABIDOPSIS THALIANA12 (ZAT12) was manipulated. The overexpressor lines performed better under salt stress and this was associated with the higher expression levels of AZI1 and EARLI1 (Davletova et al. 2005). Furthermore, exposure of the salt tolerant xero-halophyte Haloxylon salicornicum to salt stress resulted lower stearic acid and palmitic acid levels. Panda et al. (2021) speculated that breakage of lipid membranes might lead to higher accumulation of AZA. In conclusion, AZA is also involved in abiotic stress tolerance in various plant species, such as cold (Davletova et al. 2005) and salt tolerance (Atkinson et al. 2013). When Arabidopsis seedlings are exposed to simultaneous biotic and abiotic stresses, AZI1 was down-regulated in leaves and conferred drought susceptibility when overexpressed (Atkinson et al. 2013). More functional analyses are required to understand the role of AZI1 in abiotic stress responses. The AZA/AZI1 pathway is involved in both biotic and abiotic stress responses in plants, and a comparative analysis of both stimuli might be helpful to throw more light on the molecular mechanism of systemic resistance. AZA accumulates in response to lipid peroxidation and Zöller et al. (2012) showed that lipid peroxidation is predominantly confined to plastid lipids comprising galactolipid and triacylglyceride species during the interaction of Arabidopsis with P. syringae, i.e. biotic stress. 1O2 was identified as the major cause of lipid oxidation under basal conditions, while LOX2- and free radical-catalyzed lipid oxidation substantially contribute to the increase upon pathogen infection (Zöller et al. 2012). It remains to be determined, whether all AZI1-mediated biotic and abiotic stress responses are linked to AZA and lipid peroxidation in the plastids. Barely anything is known about the role of AZA in AZI1-dependent abiotic stress responses and whether these responses are restricted to local tissues or operate systemically. Systemic signal propagation induced by abiotic stresses might be agriculturally important, e.g. for crop plants with roots in cold soil and aerial parts exposed to extreme heat. Finally, the role of the secreted AZI1 in the apoplast or at the plasma membrane for systemic immune responses and local abiotic stress responses has not yet been studied. This is particularly interesting since other members of the HyPRP family which are found in the apoplast, participate in abiotic stress responses (Saikia et al. 2020). The initiation of the AZA/AZI1 signaling at plastids needs to be investigated in more details. AZA is present in roots and shoots, but the plastids and the intraorganellar membrane structure as the site of lipid peroxidation differ substantially in two types of plastids. In both organs, AZI1 has been shown to be associated at least in part with plastids. In particular, in the aerial tissue, HyPRPs are mainly found in epidermal cells. Their plastids play key roles in defense against microbes (cf. Banday et al. 2022). Investigating the role of root plastids for the generation of AZA and the AZA/AZI1 interaction is important for unravelling the function of the signalling compounds in roots. Furthermore, AZI1 and EARLI1 expression is strongly down-regulated in roots upon colonisation by fungi (Banday et al. 2022), whereas this was not observed for other HyPRP genes. This raises the question whether AZI1/EARLI1 might have also other functions in roots, e.g., by controlling root colonisation or entry of fungal hyphae into the roots. Banday et al. (2022) have already demonstrated that HyPRPs regulate the interaction with the plant growth-promoting rhizobacteria Pseudomonas simiae WCS417 in the roots to influence colonization, root system architecture, and/or biomass. Further studies are required to understand the differences in the regulation of these HyPRP genes, as well as function and signalling of the proteins in roots and shoots upon pathogenic and beneficial microbial attacks. Disruption of galactolipids in the plastid membranes by lipid peroxidation generates breakdown products including the oxo-acid AZA which protrude to the aqueous phase. In particular, during membrane repair, this promotes AZA release from the membrane and mobility to neighbouring membranes, either alone or complexed by AZI1. The role of AZI1 for the movement of AZA between membranes requires further attention. Whether membrane disruption during the oxidative process plays a role for AZA movement, should be investigated. Based on the current knowledge about the regulation of the pathway, localisation of AZI1 in or at plastids and its trafficking to other cellular membranes are early events that proceed the activation of the systemic movement of the priming signal of AZA/AZI1 to distal tissue (cf. Cecchini et al. 2021). Plastid targeting of AZI1 is promoted by MAPK3/6, which is activated by biotic (Pitzschke et al. 2009) and abiotic stresses (e.g. Li et al. 2014). Besides MAPK3/6 activation, biotic and abiotic stresses also generate ROS (Takata et al. 2020; Rodriguez et al. 2010), which–in turn—promote plastid-association of AZI1 via MAPK3/6 signaling, but also lipid peroxidation in plastids which generates AZA. It would be interesting to know how the AZI1 and AZA generation is coordinated (Fig. 2). A main question centers around the long-distance transportation of AZA or the AZA signal to systemic tissue. If AZA can travel root-, but not shootwards, one has to postulate different mechanisms for the propagation of the information from the roots to the shoots and from the shoots to the roots. AZA is not water soluble and it is well known that membrane contact sites between plastid envelopes, endoplasmatic reticulum, plasma membrane and membrane material at the plasmodesmata are the sites of exchange of small molecules, including AZA (Andersson et al. 2007; Toulmay and Prinz 2011; Li et al. Ádám AL, Kátay G, Künstler A, Király L (2022) Detection of lipid peroxidation-derived free azelaic acid, a biotic stress marker and other dicarboxylic acids in tobacco by reversed-phase HPLC-MS Under non-derivatized conditions. Methods Mol Biol 2526:191–200. https://doi.org/10.1007/978-1-0716-2469-2_14. (PMID: 35657521) Andersson MX, Goksor M, Sandelius AS (2007) Membrane contact sites: physical attachment between chloroplasts and endoplasmic reticulum revealed by optical manipulation. Plant Signal Behav 2:185–218 Atkinson NJ, Lilley CJ, Urwin PE (2013) Identification of genes involved in the response of Arabidopsis to simultaneous biotic and abiotic stresses. Plant Physiol 162(4):2028–2041. https://doi.org/10.1104/pp.113.222372. (Epub 2013 Jun 25. PMID: 23800991; PMCID: PMC3729780) Banday ZZ, Cecchini NM, Speed DJ, Scott AT, Parent C, Hu CT, Filzen RC, Agbo E, Greenberg JT (2022) Friend or foe: hybrid proline-rich proteins determine how plants respond to beneficial and pathogenic microbes. Plant Physiol 190(1):860–881. https://doi.org/10.1093/plphys/kiac263. (PMID: 35642916; PMCID: PMC9434206) Bell E, Mullet JE (1991) Lipoxygenase gene expression is modulated in plants by water deficit, wounding, and methyl jasmonate. Mol Gen Genet 230:456–462 Bell E, Mullet JE (1993) Characterization of an Arabidopsis lipoxygenase gene responsive to methyl jasmonate and wounding. Plant Physiol 103:1133–1138 Bienert GP, Schjoerring JK, Jahn TP (2006) Membrane transport of hydrogen peroxide. Biochim Biophys Acta 1758:994–1003. https://doi.org/10.1016/j.bbamem.2006.02.015. (Epub 2006 Mar 10 PMID: 16566894) Blée E (2002) Impact of phytooxylipins in plant defense. Trends Plant Sci 7(7):315–322. https://doi.org/10.1016/s1360-1385(02)02290-2. (PMID: 12119169) Blée E, Joyard J (1996) Envelope membranes from spinach chloroplasts are a site of metabolism of fatty acid hydroperoxides. Plant Physiol 110:445–454. https://doi.org/10.1104/pp.110.2.445. (PMID: 12226196; PMCID: PMC157739) Breuers FKH, Bräutigam A, Weber APM (2011) The plastid outer envelope—a highly dynamic interface between plastid and cytoplasm. Front Plant Sci 2:97 Cecchini NM, Steffes K, Schläppi MR, Gifford AN, Greenberg JT (2015) Arabidopsis AZI1 family proteins mediate signal mobilization for systemic defence priming. Nat Commun 6:7658. https://doi.org/10.1038/ncomms8658. (PMID: 26203923) Cecchini NM, Roychoudhry S, Speed DJ, Steffes K, Tambe A, Zodrow K, Konstantinoff K, Jung HW, Engle NL, Tschaplinski TJ, Greenberg JT (2019) Underground azelaic acid-conferred resistance to Pseudomonas syringae in arabidopsis. Mol Plant Microbe Interact 32(1):86–94. https://doi.org/10.1094/MPMI-07-18-0185-R. (Epub 2018 Oct 23 PMID: 30156481) Cecchini NM, Speed DJ, Roychoudhry S, Greenberg JT (2021) Kinases and protein motifs required for AZI1 plastid localization and trafficking during plant defense induction. Plant J 105(6):1615–1629. https://doi.org/10.1111/tpj.15137. (Epub 2021 Feb 20. PMID: 33342031; PMCID: PMC8048937) Conrath U, Beckers GJ, Langenbach CJ, Jaskiewicz MR (2015) Priming for enhanced defense. Annu Rev Phytopathol 53:97–119. https://doi.org/10.1146/annurev-phyto-080614-120132. (Epub 2015 Jun 11 PMID: 26070330) Davletova S, Schlauch K, Coutu J, Mittler R (2005) The zinc-finger protein Zat12 plays a central role in reactive oxygen and abiotic stress signaling in Arabidopsis. Plant Physiol 139(2):847–856. https://doi.org/10.1104/pp.105.068254. (Epub 2005 Sep 23. PMID: 16183833; PMCID: PMC1256000) De Domenico S, Bonsegna S, Horres R, Pastor V, Taurino M, Poltronieri P, Imtiaz M, Kahl G, Flors V, Winter P, Santino A (2012) Transcriptomic analysis of oxylipin biosynthesis genes and chemical profiling reveal an early induction of jasmonates in chickpea roots under drought stress. Plant Physiol Biochem 61:115–122. https://doi.org/10.1016/j.plaphy.2012.09.009. (Epub 2012 Oct 11 PMID: 23141673) Dempsey DA, Klessig DF (2012) SOS - too many signals for systemic acquired resistance? Trends Plant Sci 17(9):538–545. https://doi.org/10.1016/j.tplants.2012.05.011. (Epub 2012 Jun 29 PMID: 22749315) Dinant S, Lemoine R (2010) The phloem pathway: new issues and old debates. CR Biol 333:307–319 Djami-Tchatchou AT, Ncube EN, Steenkamp PA, Dubery IA (2017) Similar, but different: structurally related azelaic acid and hexanoic acid trigger differential metabolomic and transcriptomic responses in tobacco cells. BMC Plant Biol 17(1):227. https://doi.org/10.1186/s12870-017-1157-5. (PMID: 29187153; PMCID: PMC5706331) Du H, Liu H, **ong L (2013) Endogenous auxin and jasmonic acid levels are differentially modulated by abiotic stresses in rice. Front Plant Sci 4:397. https://doi.org/10.3389/fpls.2013.00397. (PMID: 24130566; PMCID: PMC3793129) Dutton C, Hõrak H, Hepworth C, Mitchell A, Ton J, Hunt L, Gray JE (2019) Bacterial infection systemically suppresses stomatal density. Plant Cell Environ 42(8):2411–2421. https://doi.org/10.1111/pce.13570. (Epub 2019 Jun 10. PMID: 31042812; PMCID: PMC6771828) Dvorakova L, Cvrckova F, Fischer L (2007) Analysis of the hybrid proline-rich protein families from seven plant species suggests rapid diversification of their sequences and expression patterns. BMC Genom 8:412 El-Shetehy M, Wang C, Shine MB, Yu K, Kachroo A, Kachroo P (2015) Nitric oxide and reactive oxygen species are required for systemic acquired resistance in plants. Plant Signal Behav 10(9):e998544. https://doi.org/10.1080/15592324.2014.998544 Farmer EE, Mueller MJ (2013) ROS-mediated lipid peroxidation and RES-activated signaling. Annu Rev Plant Biol 64:429–450. https://doi.org/10.1146/annurev-arplant-050312-120132. (Epub 2013 Feb 28 PMID: 23451784) Fernandez-Calvino L, Faulkner C, Walshaw J, Saalbach G, Bayer E, Benitez-Alfonso Y, Maule A (2011) Arabidopsis plasmodesmal proteome. PLoS ONE 6:85. https://doi.org/10.1371/journal.pone.0018880 Froehlich JE, Benning C, Dörmann P (2001a) The digalactosyldiacylglycerol (DGDG) synthase DGD1 is inserted into the outer envelope membrane of chloroplasts in a manner independent of the general import pathway and does not depend on direct interaction with monogalactosyldiacylglycerol synthase for DGDG biosynthesis. J Biol Chem 276:31806–31812. https://doi.org/10.1074/jbc.M104652200 Froehlich JE, Itoh A, Howe GA (2001b) Tomato allene oxide synthase and fatty acid hydroperoxide lyase, two cytochrome P450s involved in oxylipin metabolism, are targeted to different membranes of chloroplast envelope. Plant Physiol 125:306–317. https://doi.org/10.1104/pp.125.1.306 Fu ZQ, Dong X (2013) Systemic acquired resistance: turning local infection into global defense. Annu Rev Plant Biol 64:839–863 Fujii J, Soma Y, Matsuda Y (2023) Biological action of singlet molecular oxygen from the standpoint of cell signaling. Injury Death Mol 28(10):4085. https://doi.org/10.3390/molecules28104085. (PMID: 37241826; PMCID: PMC10223444) Gao QM, Yu K, **a Y, Shine MB, Wang C, Navarre D, Kachroo A, Kachroo P (2014) Mono- and digalactosyldiacylglycerol lipids function nonredundantly to regulate systemic acquired resistance in plants. Cell Rep 9(5):1681–1691. https://doi.org/10.1016/j.celrep.2014.10.069. (Epub 2014 Nov 26 PMID: 25466253) Gao QM, Zhu S, Kachroo P, Kachroo A (2015) Signal regulators of systemic acquired resistance. Front Plant Sci 6:228. https://doi.org/10.3389/fpls.2015.00228. (PMID: 25918514; PMCID: PMC4394658) Gao H, Guo M, Song J, Ma Y, Xu Z (2021) Signals in systemic acquired resistance of plants against microbial pathogens. Mol Biol Rep 48(4):3747–3759. https://doi.org/10.1007/s11033-021-06344-7. (Epub 2021 Apr 24 PMID: 33893927) Gong Q, Wang Y, He L, Huang F, Zhang D, Wang Y, Wei X, Han M, Deng H, Luo L, Cui F, Hong Y, Liu Y (2023) Molecular basis of methyl-salicylate-mediated plant airborne defence. Nature 622:139–148. https://doi.org/10.1038/s41586-023-06533-3. (Epub 2023 Sep 13 PMID: 37704724) Gusain S, Joshi S, Joshi R (2023) Sensing, signalling, and regulatory mechanism of cold-stress tolerance in plants. Plant Physiol Biochem 197:107646. https://doi.org/10.1016/j.plaphy.2023.107646. (Epub 2023 Mar 15 PMID: 36958153) Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322 Hartmann M, Kim D, Bernsdorff F, Ajami-Rashidi Z, Scholten N, Schreiber S, Zeier T, Schuck S, Reichel-Deland V, Zeier J (2017) Biochemical principles and functional aspects of pipecolic acid biosynthesis in plant immunity. Plant Physiol 174:124–153 Hartmann M, Zeier T, Bernsdorff F, Reichel-Deland V, Kim D, Hohmann M, Scholten N, Schuck S, Bräutigam A, Hölzel T, Ganter C, Zeier J (2018) Flavin monooxygenase-generated N-hydroxypipecolic acid is a critical element of plant systemic immunity. Cell 173(2):456-469.e16. https://doi.org/10.1016/j.cell.2018.02.049. (Epub 2018 Mar 22 PMID: 29576453) Helle SC, Kanfer G, Kolar K, Lang A, Michel AH, Kornmann B (2013) Organization and function of membrane contact sites. Biochim Biophys Acta 1833(11):2526–2541. https://doi.org/10.1016/j.bbamcr.2013.01.028. (Epub 2013 Feb 1 PMID: 23380708) Howe GA, Schilmiller AL (2002) Oxylipin metabolism in response to stress. Curr Opin Plant Biol 5:230–236. https://doi.org/10.1016/S1369-5266(02)00250-9 Huang Y, Liu Q, Jibrin M, Mou Z, Dufault N, Li Y, Zhang S (2023) Evaluating nicotinamide adenine dinucleotide for its effects on halo blight of snap bean. Plant Dis 107(3):675–681. https://doi.org/10.1094/PDIS-05-22-1126-RE. (Epub 2023 Mar 19 PMID: 35881875) Ito Y, Katsura K, Maruyama K, Taji T, Kobayashi M, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2006) Functional analysis of rice DREB1/CBF-type transcription factors involved in cold-responsive gene expression in transgenic rice. Plant Cell Physiol 47(1):141–153. https://doi.org/10.1093/pcp/pci230. (Epub 2005 Nov 12 PMID: 16284406) Javvadi SG, Cescutti P, Rizzo R, Lonzarich V, Navarini L, Licastro D, Guarnaccia C, Venturi V (2018) The spent culture supernatant of Pseudomonas syringae contains azelaic acid. BMC Microbiol 28 18(1):199. https://doi.org/10.1186/s12866-018-1352-z Jose-Estanyol M, Gomis-Ruth FX, Puigdomenech P (2004) The eight-cysteine motif, a versatile structure in plant proteins. Plant Physiol Biochem 42:355–365 Jung HW, Tschaplinski TJ, Wang L, Glazebrook J, Greenberg JT (2009) Priming in systemic plant immunity. Science 324(5923):89–91. https://doi.org/10.1126/science.1170025. (PMID: 19342588) Kachroo A, Kachroo P (2020) Mobile signals in systemic acquired resistance. Curr Opin Plant Biol 58:41–47. https://doi.org/10.1016/j.pbi.2020.10.004. (Epub 2020 Nov 14 PMID: 33202317) Kim C, Apel K (2013) Singlet oxygen-mediated signaling in plants: moving from flu to wild type reveals an increasing complexity. Photosynth Res 116:455–464 Kim TJ, Lim GH (2023) Salicylic acid and mobile regulators of systemic immunity in plants: transport and metabolism. Plants 12(5):1013. https://doi.org/10.3390/plants12051013. (PMID: 36903874; PMCID: PMC10005269) Kishimoto K, Matsui K, Ozawa R, Takabayashi J (2008) Direct fungicidal activities of C6-aldehydes are important constituents for defense responses in Arabidopsis against Botrytis cinerea. Phytochemistry 69:2127–2132. https://doi.org/10.1016/j.phytochem.2008.04.023 Koch E, Meier BM, Eiben HG, Slusarenko A (1992) A lipoxygenase from leaves of tomato (Lycopersicon esculentum Mill.) is induced in response to plant pathogenic pseudomonads. Plant Physiol 99(2):571–576. https://doi.org/10.1104/pp.99.2.571 Korenblum E, Dong Y, Szymanski J, Panda S, Jozwiak A, Massalha H, Meir S, Rogachev I, Aharoni A (2020) Rhizosphere microbiome mediates systemic root metabolite exudation by rootto-root signaling. Proc Natl Acad Sci U S A 117(7):3874–3883. https://doi.org/10.1073/pnas.1912130117 Kundu A, Mishra S, Kundu P, Jogawat A, Vadassery J (2022) Piriformospora indica recruits host-derived putrescine for growth promotion in plants. Plant Physiol 28 188(4):2289–2307. https://doi.org/10.1093/plphys/kiab536 Lee JY, Wang X, Cui W, Sager R, Modla S, Czymmek K, Zybaliov B, van Wijk K, Zhang C, Lu H, Lakshmanan V (2011) A plasmodesmata-localized protein mediates crosstalk between cell-to-cell communication and innate immunity in Arabidopsis. Plant Cell 23(9):3353–3373. https://doi.org/10.1105/tpc.111.087742. (Epub 2011 Sep 20. PMID: 21934146; PMCID: PMC3203451) Li M, Kim C (2021) Chloroplast ROS and stress signaling. Plant Commun 3(1):100264. https://doi.org/10.1016/j.xplc.2021.100264. (PMID: 35059631; PMCID: PMC8760138) Li CH, Wang G, Zhao JL, Zhang LQ, Ai LF, Han YF, Sun DY, Zhang SW, Sun Y (2014) The receptor-like kinase SIT1 mediates salt sensitivity by activating MAPK3/6 and regulating ethylene homeostasis in rice. Plant Cell 26(6):2538–2553. https://doi.org/10.1105/tpc.114.125187. (Epub 2014 Jun 6. PMID: 24907341; PMCID: PMC4114950) Li T, **ao Z, Li H, Liu C, Shen W, Gao C (2020) A combinatorial reporter set to visualize the membrane contact sites between endoplasmic reticulum and other organelles in plant cell. Front Plant Sci 11:1280 Lim GH, Shine MB, de Lorenzo L, Yu K, Cui W, Navarre D, Hunt AG, Lee JY, Kachroo A, Kachroo P (2016) Plasmodesmata localizing proteins regulate transport and signaling during systemic acquired immunity in plants. Cell Host Microbe 19(4):541–549. https://doi.org/10.1016/j.chom.2016.03.006. (PMID: 27078071) Liu W, Wang H, Chen Y, Zhu S, Chen M, Lan X, Chen G, Liao Z (2017) Cold stress improves the production of artemisinin depending on the increase in endogenous jasmonate. Biotechnol Appl Biochem 64(3):305–314. https://doi.org/10.1002/bab.1493. (Epub 2016 Oct 10 PMID: 26988377) Losvik A, Beste L, Glinwood R, Ivarson E, Stephens J, Zhu LH, Jonsson L (2017) Overexpression and down-regulation of barley lipoxygenase LOX2.2 affects jasmonate-regulated genes and aphid fecundity. Int J Mol Sci 18(12):2765. https://doi.org/10.3390/ijms18122765 Maruyama K, Sakuma Y, Kasuga M, Ito Y, Seki M, Goda H, Shimada Y, Yoshida S, Shinozaki K, Yamaguchi-Shinozaki K (2004) Identification of cold-inducible downstream genes of the Arabidopsis DREB1A/CBF3 transcriptional factor using two microarray systems. Plant J 38(6):982–993. https://doi.org/10.1111/j.1365-313X.2004.02100.x. (PMID: 15165189) Matsui K (2006) Green leaf volatiles: hydroperoxide lyase pathway of oxylipin metabolism. Curr Opin Plant Biol 9:274–280 Miranda de la Torre JO, Peppino Margutti MY, Lescano López I, Cambiagno DA, Alvarez ME, Cecchini NM (2023) The Arabidopsis chromatin regulator MOM1 is a negative component of the defense priming induced by AZA. BABA and PIP Front Plant Sci 14:1133327. https://doi.org/10.3389/fpls.2023.1133327. (PMID:37229135;PMCID:PMC10203520) Mitra SK, Walters BT, Clouse SD, Goshe MB (2009) An efficient organic solvent based extraction method for the proteomic analysis of arabidopsis plasma membranes. J Proteom Res 8:2752–2767. https://doi.org/10.1021/pr801044y Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498 Mosblech A, Feussner I, Heilmann I (2009) Oxylipins: structurally diverse metabolites from fatty acid oxidation. Plant Physiol Biochem 47:511–517 Nieto-Garibay A, Barraza A, Caamal-Chan G, Murillo-Amador B, Troyo-Diéguez E, Burgoa-Cruz CA, Jaramillo-Limón JN, Loera-Muro A (2022) Habanero pepper (Capsicum chinense) adaptation to water-deficit stress in a protected agricultural system. Funct Plant Biol 49(3):295–306. https://doi.org/10.1071/FP20394. (PMID: 35130477) Oelmüller R (2021) Threat at one end of the plant: What travels to inform the other parts? Int J Mol Sci 22(6):3152. https://doi.org/10.3390/ijms22063152.PMID:33808792;PMCID:PMC8003533 Ohta H, Shida K, Peng YL, Furusawa I, Shishiyama J, Aibara S, Morita Y (1991) A lipoxygenase pathway is activated in rice after infection with the rice blast fungus Magnaporthe grisea. Plant Physiol 97(1):94–98. https://doi.org/10.1104/pp.97.1.94.PMID:16668421;PMCID:PMC1080968 Panda A, Rangani J, Parida AK (2021) Unraveling salt responsive metabolites and metabolic pathways using non-targeted metabolomics approach and elucidation of salt tolerance mechanisms in the xero-halophyte Haloxylon salicornicum. Plant Physiol Biochem 158:284–296. https://doi.org/10.1016/j.plaphy.2020.11.012. (Epub 2020 Nov 16 PMID: 33239222) Pitzschke A, Schikora A, Hirt H (2009) MAPK cascade signalling networks in plant defence. Curr Opin Plant Biol 12(4):421–426. https://doi.org/10.1016/j.pbi.2009.06.008. (Epub 2009 Jul 14 PMID: 19608449) Pitzschke A, Datta S, Persak H (2014a) Mitogen-activated protein kinase-regulated AZI1 - an attractive candidate for genetic engineering. Plant Signal Behav 9(2):e27764. https://doi.org/10.4161/psb.27764. (Epub 2014 Feb 10. PMID: 24518841; PMCID: PMC4091252) Pitzschke A, Datta S, Persak H (2014b) Salt stress in Arabidopsis: lipid transfer protein AZI1 and its control by mitogen-activated protein kinase MPK3. Mol Plant 7(4):722–738. https://doi.org/10.1093/mp/sst157. (Epub 2013 Nov 8. PMID: 24214892; PMCID: PMC3973493) Pitzschke A, Xue H, Persak H, Datta S, Seifert GJ (2016) Post-translational modification and secretion of azelaic acid induced 1 (AZI1), a hybrid proline-rich protein from arabidopsis. Int J Mol Sci 17(1):85. https://doi.org/10.3390/ijms17010085. (PMID: 26771603; PMCID: PMC4730328) Pospíšil P (2016) Production of reactive oxygen species by photosystem ii as a response to light and temperature stress. Front Plant Sci 7:1950. https://doi.org/10.3389/fpls.2016.01950. (PMID: 28082998; PMCID: PMC5183610) Prost I, Dhondt S, Rothe G, Vicente J, Rodriguez MJ, Kift N, Carbonne F, Griffiths G, Esquerré-Tugayé MT, Radi R (1998) Peroxynitrite reactions and diffusion in biology. Chem Res Toxicol 11:720–721 Riedlmeier M, Ghirardo A, Wenig M, Knappe C, Koch K, Georgii E, Dey S, Parker JE, Schnitzler JP, Vlot AC (2017) Monoterpenes Support Systemic Acquired Resistance within and between Plants. Plant Cell 29(6):1440–1459. https://doi.org/10.1105/tpc.16.00898. (Epub 2017 May 23. PMID: 28536145; PMCID: PMC5502447) Rodriguez MC, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649. https://doi.org/10.1146/annurev-arplant-042809-112252. (PMID: 20441529) Rosahl S, Castresana C, Hamberg M, Fournier J (2005) Evaluation of the antimicrobial activities of plant oxylipins supports their involvement in defense against pathogens. Plant Physiol 139(4):1902–1913. https://doi.org/10.1104/pp.105.066274. (Epub 2005 Nov 18. PMID: 16299186; PMCID: PMC1310568) Rustgi S, Springer A, Kang C, von Wettstein D, Reinbothe C, Reinbothe S, Pollmann S (2019) Allene oxide synthase and hydroperoxide lyase, two non-canonical cytochrome P450s in Arabidopsis thaliana and their different roles in plant defense. Int J Mol Sci 20(12):3064. https://doi.org/10.3390/ijms20123064. (PMID: 31234561; PMCID: PMC6627107) Ryter SW, Tyrrell RM (1998) Singlet molecular oxygen (1O2): a possible effector of eukaryotic gene expression. Free Radic Biol Med 24(9):1520–1534. https://doi.org/10.1016/s0891-5849(97)00461-9. (PMID: 9641271) Saikia B, Singh S, Debbarma J, Velmurugan N, Dekaboruah H, Arunkumar KP, Chikkaputtaiah C (2020) Multigene CRISPR/Cas9 genome editing of hybrid proline rich proteins (HyPRPs) for sustainable multi-stress tolerance in crops: the review of a promising approach. Physiol Mol Biol Plants 26(5):857–869. https://doi.org/10.1007/s12298-020-00782-6. (Epub 2020 Apr 20. PMID: 32377037; PMCID: PMC7196567) Shi LN, Lu LX, Ye JR, Shi HM (2022) The endophytic strain ZS-3 enhances salt tolerance in Arabidopsis thaliana by regulating photosynthesis, osmotic stress, and ion homeostasis and inducing systemic tolerance. Front Plant Sci 13:820837. https://doi.org/10.3389/fpls.2022.820837. (PMID: 35386673; PMCID: PMC8977589) Shine MB, **ao X, Kachroo P, Kachroo A (2019) Signaling mechanisms underlying systemic acquired resistance to microbial pathogens. Plant Sci 279:81–86. https://doi.org/10.1016/j.plantsci.2018.01.001. (Epub 2018 Jan 3 PMID: 30709496) Siebers M, Brands M, Wewer V, Duan Y, Hölzl G, Dörmann P (2016) Lipids in plant-microbe interactions. Biochim Biophys Acta 1861:1379–1395. https://doi.org/10.1016/j.bbalip.2016.02.021 Siedow JN (1991) Plant lipoxygenase: structure and function. Annu Rev Plant Physiol Plant Mol Biol 42:145–188 Takata T, Araki S, Tsuchiya Y, Watanabe Y (2020) Oxidative stress orchestrates mapk and nitric-oxide synthase signal. Int J Mol Sci 21(22):8750. https://doi.org/10.3390/ijms21228750. (PMID: 33228180; PMCID: PMC7699490) Toulmay A, Prinz WA (2011) Lipid transfer and signaling at organelle contact sites: the tip of the iceberg. Curr Opin Cell Biol 23:458–463 Triantaphylides C, Krischke M, Hoeberichts FA, Ksas B, Gresser G, Havaux M, Van Breusegem F, Mueller MJ (2008) Singlet oxygen is the major reactive oxygen species involved in photooxidative damage to plants. Plant Physiol 148:960–968 Triantaphylidès C, Havaux M (2009) Singlet oxygen in plants: production, detoxification and signaling. Trends Plant Sci 14(4):219–228. https://doi.org/10.1016/j.tplants.2009.01.008. (Epub 2009 Mar 18 PMID: 19303348) Tripathy BC, Oelmüller R (2012) Reactive oxygen species generation and signaling in plants. Plant Signal Behav 7(12):1621–1633. https://doi.org/10.4161/psb.22455. (Epub 2012 Oct 16. PMID: 23072988; PMCID: PMC3578903) Vandelle E, Delledonne M (2011) Peroxynitrite formation and function in plants. Plant Sci 81:534–539 Vellosillo T, Martínez M, López MA, Vicente J, Cascón T, Dolan L, Hamberg M, Castresana C (2007) Oxylipins produced by the 9-lipoxygenase pathway in Arabidopsis regulate lateral root development and defense responses through a specific signaling cascade. Plant Cell 19(3):831–846. https://doi.org/10.1105/tpc.106.046052. (Epub 2007 Mar 16. PMID: 17369372; PMCID: PMC1867370) Vick BA, Zimmerman DC (1987) The Biochemistry of Plants, ed. Stumpf, P. K. (Academic, New York), Vol. 9:53–90 Vlot AC, Sales JH, Lenk M, Bauer K, Brambilla A, Sommer A, Chen Y, Wenig M, Nayem S (2021) Systemic propagation of immunity in plants. New Phytol 229(3):1234–1250. https://doi.org/10.1111/nph.16953. (Epub 2020 Oct 24 PMID: 32978988) Wang Z, Benning C (2012) Chloroplast lipid synthesis and lipid trafficking through ER-plastid membrane contact sites. Biochem Soc Trans 40:457–463 Wang X, Sager R, Cui W, Zhang C, Lu H, Lee JY (2013) Salicylic acid regulates Plasmodesmata closure during innate immune responses in Arabidopsis. Plant Cell 25(6):2315–2329. https://doi.org/10.1105/tpc.113.110676. (Epub 2013 Jun 7. PMID: 23749844; PMCID: PMC3723628) Wang C, El-Shetehy M, Shine MB, Yu K, Navarre D, Wendehenne D, Kachroo A, Kachroo P (2014) Free radicals mediate systemic acquired resistance. Cell Rep 7(2):348–355. https://doi.org/10.1016/j.celrep.2014.03.032. (Epub 2014 Apr 13 PMID: 24726369) Wang XY, Li DZ, Li Q, Ma YQ, Yao JW, Huang X, Xu ZQ (2016) Metabolomic analysis reveals the relationship between AZI1 and sugar signaling in systemic acquired resistance of Arabidopsis. Plant Physiol Biochem 107:273–287. https://doi.org/10.1016/j.plaphy.2016.06.016. (Epub 2016 Jun 15 PMID: 27337039) Wang C, Liu R, Lim GH, de Lorenzo L, Yu K, Zhang K, Hunt AG, Kachroo A, Kachroo P (2018) Pipecolic acid confers systemic immunity by regulating free radicals. Sci Adv 4(5):eaar4509. https://doi.org/10.1126/sciadv.aar4509. (PMID: 29854946; PMCID: PMC5976275) Wendehenne D, Gao QM, Kachroo A, Kachroo P (2014) Free radical-mediated systemic immunity in plants. Curr Opin Plant Biol 20:127–134. https://doi.org/10.1016/j.pbi.2014.05.012. (Epub 2014 Jun 12 PMID: 24929297) Wittek F, Hoffmann T, Kanawati B, Bichlmeier M, Knappe C, Wenig M, Schmitt-Kopplin P, Parker JE, Schwab W, Vlot AC (2014) Arabidopsis ENHANCED DISEASE SUSCEPTIBILITY1 promotes systemic acquired resistance via azelaic acid and its precursor 9-oxo nonanoic acid. J Exp Bot 65(20):5919–5931. https://doi.org/10.1093/jxb/eru331. (Epub 2014 Aug 11. PMID: 25114016; PMCID: PMC4203127) Wong CE, Li Y, Labbe A, Guevara D, Nuin P, Whitty B, Diaz C, Golding GB, Gray GR, Weretilnyk EA, Griffith M, Moffatt BA (2006) Transcriptional profiling implicates novel interactions between abiotic stress and hormonal responses in Thellungiella, a close relative of Arabidopsis. Plant Physiol 140(4):1437–1450. https://doi.org/10.1104/pp.105.070508. (Epub 2006 Feb 24. PMID: 16500996; PMCID: PMC1435811) Xu ZY, Zhang X, Schläppi M, Xu ZQ (2011) Cold-inducible expression of AZI1 and its function in improvement of freezing tolerance of Arabidopsis thaliana and Saccharomyces cerevisiae. J Plant Physiol 168(13):1576–1587. https://doi.org/10.1016/j.jplph.2011.01.023. (Epub 2011 Apr 14 PMID: 21492954) Yu K, Soares JM, Mandal MK, Wang C, Chanda B, Gifford AN, Fowler JS, Navarre D, Kachroo A, Kachroo P (2013) A feedback regulatory loop between G3P and lipid transfer proteins DIR1 and AZI1 mediates azelaic-acid-induced systemic immunity. Cell Rep 3(4):1266–1278. https://doi.org/10.1016/j.celrep.2013.03.030. (Epub 2013 Apr 18 PMID: 23602565) Zhang Y, Schläppi M (2007) Cold responsive EARLI1 type HyPRPs improve freezing survival of yeast cells and form higher order complexes in plants. Planta 227(1):233–243. https://doi.org/10.1007/s00425-007-0611-2. (Epub 2007 Sep 5 PMID: 17786468) Zhao Q, Liu F, Song C, Zhai T, He Z, Ma L, Zhao X, Jia Z, Song S (2023) Diffusible signal factor primes plant immunity against Xanthomonas campestris pv. campestris (Xcc) via JA signaling in Arabidopsis and Brassica oleracea. Front Cell Infect Microbiol 13:1203582. https://doi.org/10.3389/fcimb.2023.1203582. (PMID: 37404719; PMCID: PMC10315614) Zoeller M, Stingl N, Krischke M, Fekete A, Waller F, Berger S, Mueller MJ (2012) Lipid profiling of the Arabidopsis hypersensitive response reveals specific lipid peroxidation and fragmentation processes: biogenesis of pimelic and azelaic acid. Plant Physiol 160(1):365–378. https://doi.org/10.1104/pp.112.202846. (Epub 2012 Jul 22. PMID: 22822212; PMCID: PMC3440211) None Open Access funding enabled and organized by Projekt DEAL. Funding was received from the Deutsche Forschungsgemeinschaft (CRC1127, project ID: 239748522 to R.O.) PR was supported by the Indian Council of Agricultural Research, New Delhi, India, and the International Max-Planck Research School (Chemical Ecology, Jena). Conceptualization: PR. Writing of manuscript: PR and RO. Authors declare no conflict of interest. Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations. Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. Priya Reddy, Y.N., Oelmüller, R. Lipid peroxidation and stress-induced signalling molecules in systemic resistance mediated by azelaic acid/AZELAIC ACID INDUCED1: signal initiation and propagation.
Physiol Mol Biol Plants 30, 305–316 (2024). https://doi.org/10.1007/s12298-024-01420-1 Received: Revised: Accepted: Published: Issue Date: DOI: https://doi.org/10.1007/s12298-024-01420-1Symplastic transport of AZI1 to the phloem
AZI1 gene activation and abiotic stress
Conclusion and open questions
References
Acknowledgements
Funding
Author information
Authors and Affiliations
Contributions
Corresponding author
Ethics declarations
Conflict of interest
Additional information
Publisher's Note
Rights and permissions
About this article
Cite this article
Keywords