Abstract
With the prevalence of malignant melanoma increasing gradually and the progressive westernization of the Asian lifestyle, it is important to analyze and follow up on the characteristics of malignant melanoma at regular intervals. We identified the characteristics of malignant melanoma by analyzing consecutive patients from a single medical center. We also examined the trend of malignant melanoma and prognostic factors in Asian patients. We investigated 200 consecutive patients with malignant melanoma in a single medical center between 2000 and 2022. Each patient’s sex and age, tumor stage, site of the primary lesion, histological subtype, Breslow thickness, Clark level, and period of survival were collected from the historical medical records of the patients and analyzed. Survival analyses were performed using the Kaplan–Meier method to investigate the prognostic factors. The ratio of man-to-woman was 1:1.53; the most common site of the primary tumor was the lower extremity (60%), and acral lentiginous melanoma was the most common histological subtype (61%). Malignant melanoma commonly occurs in the lower extremities, primarily in the form of the lentiginous subtype. In situ melanomas are most prevalent regarding Breslow thickness, while Clark Level 4 is the most common type of malignant melanoma. Sex and Breslow thickness were significantly associated with the survival rate. However, others were not significant prognostic indicators for survival in this cohort. This study confirmed that the epidemiology of malignant melanoma in Asian patients was maintained without significant change. We also confirmed several significant prognostic indicators for survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Malignant melanoma accounts for only approximately 1% of skin cancers and is not a common type of skin cancer [1]. However, it is a skin cancer with the highest mortality rate due to poor prognosis. Furthermore, the incidence rates and primary sites differ according to race [2]. Therefore, there is a large body of research on the prognosis and incidence of malignant melanoma.
In Asia, from 1992 to 2002, the annual average incidence rate of malignant melanoma was 1.0 per 100,000 people, which is even rarer than that in Caucasians [3]. According to GLOBOCAN 2020 data, the age-standardized incidence and mortality rates of malignant melanoma worldwide are 3.4 and 0.56 per 100,000 people, respectively. In Asia, these rates are lower at 0.42 and 0.21, respectively, than those found globally. However, according to the GLOBOCAN data, the incidence of melanoma is generally on an upward trend both worldwide and in Asia. According to the Korean Statistical Information Service, the age-standardized incidence rate of malignant melanoma in Korea has steadily increased from 0.8 in 1999 to 1.2 in 2020. Additionally, when comparing the mortality rates of patients with malignant melanoma in 31 countries from 1985 to 2015, most countries showed an increase in mortality rates, and particularly Korea showed the highest increase rate for both men and women [4].
However, the incidence of prostate, ovarian, and breast cancers gradually increases in Asians. Additionally, as one of the causes, the Westernized lifestyle is drawing attention. The composition of Korean diets has changed, and the number of overweight patients has increased. This can also be applied to patients with malignant melanoma [5].
Due to its increasing incidence and mortality, the analysis and follow-up of the characteristics of malignant melanoma along a regular cycle are important. Furthermore, since melanoma in Korean patients differs from that of those in Western countries regarding the location and characteristics, and there have not been many large-scale studies, this study aimed to investigate the characteristics of melanoma in Korean patients and compare them with those in patients from other countries [6].
This study analyzed 200 patients and secured the largest number of cases consecutively treated by one surgeon at a single institution in Korea. Additionally, considering that most previously reported large-scale cohort studies are multi-center studies, it is significant that this study can achieve a higher level of data quality. Through this study, the characteristics of malignant melanoma in Korea were confirmed, and the trend of malignant melanoma in Korea was validated by comparing 100 patients who visited the Pusan National University Hospital before 2014 with 100 who came after.
Patients and Methods
Overall, 200 consecutive patients with malignant melanoma who underwent surgery at the Pusan National University Hospital from 2000 to 2022 were investigated. All patients were admitted to the hospital and underwent surgery by a single surgeon who provided continuous care. The historical medical records of each patient were used to obtain information on sex and age, tumor stage, primary lesion site, histopathological subtype, Breslow tumor thickness, Clark level, and survival period.
The histologic subtype was categorized according to the World Health Organization classification as follows: superficial spreading melanoma (SSM), nodular melanoma (NM), acral lentiginous melanoma (ALM), lentigo maligna melanoma (LMM), and mucosal melanoma (MM). According to the American Joint Committee on Cancer (AJCC) staging system, tumor thickness and invasion were assessed using the staging systems of Breslow and Clark, respectively. The interval between the date of diagnosis of the primary cutaneous melanoma and the registered date of death or the date of the last consultation was defined as the survival time.
The 100 patients who visited before 2014 were designated as the A group, and the 100 patients who visited after 2014 were the B group, a total of 200 patients. These groups were compared to confirm whether there were any changes in the characteristics of malignant melanoma over the years.
Statistical Analysis
Multivariate Cox proportional hazard analysis was used. The associations were summarized using a hazard ratio with 95% confidence intervals. The Kaplan–Meier method was used to analyze the survival rate. Furthermore, the associations were summarized according to sex, age, location, type, tumor stage, Breslow thickness, and Clark level. Moreover, the associations were determined using the Kaplan–Meier method (log-rank test) and Gehan–Breslow–Wilcoxon test. The differences were calculated, with the significance being P < 0.05. The statistical analyses were performed using SAS software (version 9.4; SAS Institute, Cary, NC, USA).
Ethics Statement
The Institutional Review Board of Pusan National University Hospital reviewed and approved this study (IRB No. 2201–011-111).
Results
The epidemiological characteristics of the 200 patients who visited Pusan National University Hospital and underwent surgery between September 2000 and May 2022 were analyzed. The median follow-up period was 33 months. All patients' 1-year and 5-year overall survival was 95.5% and 76.5%, respectively. In group A, 64 women and 36 men indicated a man-to-woman ratio of 1:1.78. However, in group B, 57 women and 43 men showed a man-to-woman ratio of 1:1.33.
The mean age of all patients was 63.23(standard deviation [SD] 13.67) years. The average age of the patients in groups A and B was 60.25 (SD 13.71) years and 66.21 (SD 12.97) years, respectively, which indicated a gradual increase in the age of the patients (Table 1). The largest number of patients was those in their 70’s (59, 29.5%), followed by 56 (28%) in their 60’s and 30 (15%) in their 50’s. Moreover, according to the AJCC staging system, stage 2 was the most common (61 patients, 30.5%), followed by stage 3 with 54 (27%) and stage 1 with 46 (23%). (Table 2).
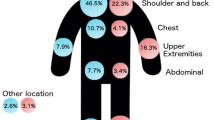
The most common site of the primary tumor was the lower extremity (119 cases, 59.5%), which included the foot (105 cases, 52.5%). The next most common site was the upper extremity (36 cases, 18%), with the same order in groups A and B. There were more patients in the order of the head and neck (28 cases, 14%) and the trunk (17 cases, 8.5%). In group A, there were more patients with sites in the trunk, while in group B, the number of patients with sites in the head, neck, and upper extremities was the same.
Compared with histopathological subtypes, the most common subtype was ALM (122 cases, 61%). The next most common subtypes were 36 cases of NM (18%), 28 of superficial spreading type (14%), 12 of LMM (6%), and two of MM (1%). The prevalence in group A was high in the order ofALM, NM, SSM, and LMM, and in group B ALM, SSM, LMM, NM, and MM. Accordingly, the prevalence of SSM and LMM increased, and the nodular type decreased.
Regarding Breslow thickness, there were 30 cases of in situ melanoma, 28 cases were ≤ 1 mm, 27 were 1–2 mm, 44 were 2–4 mm, and 71 were ≥ 4 mm. A Breslow thickness of ≥ 4 mm was the most common, followed by cases with a thickness of 2–4 mm. When groups A and B were compared, most of the patients were the same, with ≥ 4 mm, while the second most common was 2–4 mm in group A; however, in situ, group B had more cases.
Based on invasion level, when the Clark level was compared, the most frequent type of cases was Level 4, which had 69 cases, followed by Level 5, 45, Level 1, 34, Level 3, 27, and Level 2, 25. Level 4 was the most common type, followed by Level 5 and Level 1 in the order of the number of cases. In groups, A and B, most cases and the second cases were the same.
When the survival rate postoperatively was compared, sex and Breslow thickness were the indicators that showed a significant association with the survival rate. The survival rate of men was lower than that of women (P = 0.042).
The deeper the Breslow depth, the lower the survival rate. From 36 months postoperatively, the survival rate decreased rapidly in patients with a Breslow depth of 1–2 mm and when the survival rate was investigated. However, when the survival rate by age (P = 0.096), Clark level (P = 0.059), Cancer stage (P = 0.063), histological type (P = 0.491), and the primary location of malignant melanoma (P = 0.209) was compared, no significant associations were confirmed (Fig. 1).
a Kaplan–Meier curves showing survival rates in patients according to sex. Sex: men (blue line) and women (red line). b Kaplan–Meier curves showing survival rates in patients according to Breslow thickness. Breslow thickness: under 1.00 mm (blue line), 1.01–2.00 mm (green line), 2.01–4.00 mm (pink line), >4.00 mm (red line). c Kaplan–Meier curves showing survival rates in patients according to age. Age: under 50 (blue line), 50–60 (green line), 60–70 (pink line), 70–80 (purple line), and over 80 years (red line). d Kaplan–Meier curves showing survival rates in patients according to Clark level. Clark level: Clark Level 1 (blue line), Clark Level 2 (green line), Clark Level 3 (pink line), Clark Level 4 (purple line), Clark Level 5 (red line). e Kaplan–Meier curves showing survival rates in patients according to cancer stage. Cancer stage: Stage 0 (blue line), Stage 1 (green line), Stage 2 (pink line), Stage 3 (purple line), Clark Level 4 (red line). f Kaplan–Meier curves showing survival rates in patients according to histological type. Histological type: ALM, acral lentiginous melanoma (blue line); SSM, superficial spreading melanoma (green line); LM, lentigo melanoma (pink line); NM, nodular melanoma (red line). g Kaplan–Meier curves showing survival rates in patients according to tumor location. Tumor location: Head and neck (blue line), upper extremity (purple line), trunk (green line), lower extremity (red line)
Cox’s multivariate analysis identified the following variables as risk factors for malignant melanoma: sex (P = 0.046), Breslow depth of 1–2 mm (P = 0.032) and ≥ 4 mm (P = 0.010), Clark Level 5 (P = 0.019), and cancer stage 4 (P = 0.011) (Table 3).
Discussion
Malignant melanoma is a skin cancer with a high mortality rate, and the location of the onset and the most common types are different in the East and the West. The incidence and mortality rate has also changed gradually over time. Therefore, it is necessary to constantly update the data and understand the trend.
Thus, we analyzed 200 consecutive patients who visited a single institution and obtained a sufficiently large number of statistically significant cases. Additionally, since the patients were operated on by one surgeon who visited the hospital continuously, the survival rate, according to epidemiology, was compared by excluding the factors due to surgical treatment.
Furthermore, it was possible to confirm relatively accurately whether the epidemiological factors changed over time. Moreover, we compared our findings with those of other previously published studies to confirm the characteristics of malignant melanoma in Korean patients (Online Resource 1).
Through a previous study, we have confirmed that the characteristics of patients with malignant melanoma in Korea are similar to those in Asia [6]. Accordingly, this research of our center, which investigated Koreans, will be the same as the characteristics of Asians.
Our results showed that the average age of the patients was 63.2 years old, with the average age in groups A and B being 60.2 years and 66.2 years, respectively, which showed an increasing trend in the average age of patients. The average age of onset of patients with malignant melanoma in Korea has been gradually increasing in previously published reports of patients with malignant melanoma in Korea [6,7,8,9,10].
Regarding the gender ratio, the man-to-woman ratio was 1:78 and 1:33 in groups A and B, respectively, which indicated that while there were more women than men, the incidence rate of female patients decreased gradually. In Korea, according to our survey and previous reports, the prevalence of malignant melanoma in women was higher than that in men [6,7,8,9,10]. Generally, men are more likely to have melanoma, and this tendency has been found to increase as the age of onset increases [11]. In other Asian countries, the man-to-woman ratio in Singapore and Taiwan was 1.7:1 and 1.2:1, respectively. Thus, there have been more female patients, particularly in Korea [12,13,14].
Regarding the Breslow thickness, a deep depth of 4 mm or more was the most common, and no significant differences were found between groups A and B. Regarding the Clark Level, Levels 4 and 5 were high, and it has been demonstrated that the Clark level in Korean patients was higher than that of those from the West [6, 15, 16].
The most common primary tumor site was the lower extremity, which accounted for 59.5% of 119 cases. This number included the foot (105 cases, 52.5%), while the lower extremity, excluding the foot, comprised 14 cases (7%), which suggested that most of the malignant melanomas occurred in the foot. The upper extremity was the second most common site with 36 cases (18%); among these, the hand accounted for a high proportion (25 cases, 12.5%). From the results of this study and other reports published in Korea, it can be confirmed that the most common site of malignant melanomas in Korean patients was the foot area [6,7,8,9,10].
ALM was shown to be the most frequent subtype of melanoma in Asian populations, comprising 41.8%, 86.6%, 50.8%, 47%, and 50% of the patients with melanoma in China, Korea, Hong Kong, Japan, and Singapore, respectively [17, 18]. In most reports, ALM was the most common type in Korea, followed by nodule melanoma and SSM [6,7,8,9,10]. Our findings supported these results. When groups A and B were compared, the ratio of NM decreased in group B, whereas that of SSM and LMM increased. Thus, while ALM was still the most common in Korean patients with melanoma, the ratio of the other subtypes changed.
We investigated the association between age, sex, tumor location, histological subtype, cancer stage, Clark level, Breslow thickness, and survival rate and confirmed that sex and Breslow thickness were significant prognostic factors in the survival rate. Furthermore, considering that this study was a large-scale cohort study of 200 patients, it was confirmed that there was a numerical significance according to age, Clark level, and cancer stage (P < 0.1). However, no significant association was found between the histological type and tumor location (P > 0.1).
We also found that in Korean patients with malignant melanoma, it tended to occur more frequently in women, while men diagnosed with cutaneous melanoma had poorer clinical outcomes and higher disease recurrence, disease progression, and mortality than women.9 This supported the finding of a previous study that indicated that mortality rate ratios were significantly greater in men than in women in the East Asian populations of Japan, Korea, Hong Kong, and Singapore [19].
Breslow thickness has also been found to be the most powerful prognostic factor [20]. If the Breslow thickness was < 0.75 mm, most patients survived; if the Breslow thickness was ≥ 3 mm, the survival rate was approximately 50% [21, 22]. In our study, the mortality rate was higher in patients with a Breslow thickness of ≥ 1 mm than in those with a Breslow thickness of < 1 mm.
The significance of age, Clark level, and cancer stage, 0.05 < P value < 0.1, was not confirmed. However, since this was a large-scale cohort study, it was considered to have numerical significance. Age is a factor that increases the risk of local recurrence; particularly, old age has been associated with a worse prognosis in previous studies [13, 23]. Moreover, studies have suggested that the association with poor prognosis in old age may have been due to a high rate of adverse prognostic features, such as late diagnosis, low awareness of early signs and symptoms of melanoma, ulcers, and high mitotic rates [13]. Although age was not a significant poor prognostic factor in our study (P = 0.096), it was confirmed that older people tended to have a lower survival rate.
The Clark level was the method used before the Breslow method for measuring the depth of invasion. Conversely, due to the difficulty of measurement, it is usually used together with Breslow thickness [24]. However, while the Breslow thickness has been suggested as the most powerful prognostic indicator for survival in the early stage, in the case of the Clark level, according to the findings of some studies, the significance was sometimes not confirmed [25, 26]. In this study, Clark's level was not a significant prognostic factor but had a numerical significance(P = 0.059). Therefore, the deeper the invasion depth measured at the Clark level, the higher the mortality rate. Thus, it can be confirmed that the deeper the malignant melanoma's invasion depth, the higher the mortality rate.
Based on analyses of the AJCC Melanoma Staging Database, cancer stage was a prognostic factor for malignant melanoma [27]. Recently, studies have been conducted to elucidate the correlation between lymph node invasion and poor prognosis [27]. Furthermore, studies on the survival rate and benefit according to whether a sentinel lymph node biopsy was performed are also being conducted [13, 28]. In our study, the higher the cancer stage, the higher the mortality rate, confirming that the cancer stage has a numerical significance (P = 0.063). However, since we did not separate and analyze the cancer stage and the lymph node independently, it is unknown whether the lymph node is a major independent predictor of survival among all patients.
In contrast, in this study, no significant results were found according to the histological type and location of melanoma. Due to the inconsistent statistical data observed in previous studies, it remains controversial whether the anatomic site of melanoma is a significant predictor of survival [29]. Although the discrepancies between these studies have yet to be explained, they may be caused by the diverse sample sizes, inconsistent study designs, and different confounding factors used in the previous studies [13].
The histological type is a frequently mentioned prognostic factor of malignant melanoma.
While some studies have suggested that prognosis may be associated with histologic subtype (e.g., patients with NMs were believed to have a worse prognosis than those with SSM) [10, 30], more recent multivariate analyses have demonstrated that these prognostic differences were more likely to be due to other histologic features, such as Breslow thickness and ulceration histological type [23]. In this study, the survival rate of the nodular type tended to be lower than that of the other histological subtypes. However, no significant results were found according to the histological type of melanoma (P = 0.209). We believe that the ALM type was overly compared to other subtypes, reducing its precise statistical significance.
There is a limitation to this study. The utilization of a hospital-based case series may limit the generalizability of our findings to the broader epidemiology of the entire Korean population. To address this limitation, we included comparisons with our previous hospital-based case series and other Korean studies published at different time intervals, incorporating a substantial number of patients, specifically 200 individuals [6,7,8,9,10].
In conclusion, we confirmed that the characteristics of malignant melanoma in Asian patients were maintained without significant change. We also confirmed that sex and Breslow were significant prognostic factors for survival. But age, Clark level, cancer stage, site of the primary lesion, and histological type were not statistically significant prognostic factors. Therefore, further studies regarding these factors are needed to unveil more details.
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30. https://doi.org/10.3322/caac.21590
Roh MR, Kim J, Chung KY (2010) Treatment and outcomes of melanoma in acral location in Korean patients. Yonsei Med J 51:562–568. https://doi.org/10.3349/ymj.2010.51.4.562
Cormier JN, **ng Y, Ding M et al (2006) Ethnic differences among patients with cutaneous melanoma. Arch Intern Med 166:1907–1914. https://doi.org/10.1001/archinte.166.17.1907
Yang DD, Salciccioli JD, Marshall DC, Sheri A, Shalhoub J (2020) Trends in malignant melanoma mortality in 31 countries from 1985 to 2015. Br J Dermatol 183(6):1056–1064. https://doi.org/10.1111/bjd.19010
Sawada Y, Nakamura M (2021) Daily lifestyle and cutaneous malignancies. Int J Mol Sci 22:5227. https://doi.org/10.3390/ijms22105227
Nam KW, Bae YC, Bae SH, Song KH, Kim HS, Choi YJ (2015) Analysis of the clinical and histopathological patterns of 100 consecutive cases of primary cutaneous melanoma and correlation with staging. Arch Plast Surg 42:746–752. https://doi.org/10.5999/aps.2015.42.6.746
Park KD, Lee SJ, Lee WJ, Kim DW, Chung HY, Cho BC (2007) Clinicopathological features of cutaneous malignant melanoma. Korean J Dermatol 45(2):149–158
Jang HS, Kim JH, Park KH et al (2014) Comparison of melanoma subtypes among Korean patients by morphologic features and ultraviolet exposure. Ann Dermatol 26:485–490. https://doi.org/10.5021/ad.2014.26.4.485
Lee WJ, Lee YJ, Shin HJ et al (2018) Clinicopathological significance of tumor-infiltrating lymphocytes and programmed death-1 expression in cutaneous melanoma: a comparative study on clinical subtypes. Melanoma Res 28:423–434. https://doi.org/10.1097/CMR.0000000000000449
Kim JE, Chung BY, Sim CY et al (2019) Clinicopathologic features and prognostic factors of primary cutaneous melanoma: a multi-center study in Korea. J Korean Med Sci 34:e126. https://doi.org/10.3346/jkms.2019.34.e126
Yuan TA, Lu Y, Edwards K, Jakowatz J, Meyskens FL, Liu-Smith F (2019) Race-, age-, and anatomic site-specific gender differences in cutaneous melanoma suggest differential mechanisms of early- and late-onset melanoma. Int J Environ Res Public Health 16:908. https://doi.org/10.3390/ijerph16060908
Yeo PM, Lim ZV, Tan WDV et al (2021) Melanoma in Singapore: a 20-year review of disease and treatment outcomes. Ann Acad Med Singapore 50:456–466. https://doi.org/10.47102/annals-acadmedsg.2020535
Wu PC, Chen YC, Chen HM, Chen LW (2021) Prognostic factors and population-based analysis of melanoma with sentinel lymph node biopsy. Sci Rep 11:20524. https://doi.org/10.1038/s41598-021-99950-1
Lee HY, Chay WY, Tang MB, Chio MT, Tan SH (2012) Melanoma: differences between Asian and Caucasian patients. Ann Acad Med Singapore 41:17–20
Choi SJ, Bae YC, Moon JS, Nam SB, Kwak HS (2007) An analysis of clinical and histopathological pattern of malignant melanoma. Arch Plast Surg 34:557–561
Song KY, Jung KC, Cho KH, Chi JG, Ham EG (1997) Clinicopathological analysis on the 104 cases of malignant melanoma. J Pathol Transl Med 31(6):566–573
Ishihara K, Saida T, Otsuka F, Yamazaki N, Prognosis and Statistical Investigation Committee of the Japanese Skin Cancer Society (2008) Statistical profiles of malignant melanoma and other skin cancers in Japan: 2007 update. Int J Clin Oncol 13:33–41. https://doi.org/10.1007/s10147-007-0751-1
Chang JWC (2013) Acral melanoma: a unique disease in Asia. JAMA Dermatol 149:1272–1273. https://doi.org/10.1001/jamadermatol.2013.5941
Chen L, ** S (2016) Trends in mortality rates of cutaneous melanoma in East Asian populations. PeerJ 12:1–12. https://doi.org/10.7717/peerj.2809
Davey MG, Miler N, Mclnerney NM (2021) A Review of epidemiology and cancer biology of malignant melanoma. Cureus 13(5):e15087
Gershenwald JE, Scolyer RA, Hess KR et al (2017) Melanoma staging: evidence‐based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin 67:472–492. https://doi.org/10.3322/caac.21409
Blessing K, McLaren KM, McLean A, Davidson P (1991) Thick malignant melanomas (> 3 mm Breslow) with good clinical outcome: a histological study and survival analysis. Histopathology 18(2):143–148. https://doi.org/10.1111/j.1365-2559.1991.tb00842.x
Urist MM, Balch CM, Soong S-J, Shaw HM, Milton GW, Maddox WA (1985) The influence of surgical margins and prognostic factors predicting the risk of local recurrence in 3445 patients with primary cutaneous melanoma. Cancer 55:1398–1402. https://doi.org/10.1002/1097-0142(19850315)55:6%3c1398::aid-cncr2820550639%3e3.0.co;2-a
Lipsker D, Engel F, Cribier B, Velten M, Hedelin G (2007) Trends in melanoma epidemiology suggest three different types of melanoma. Br J Dermatol 157:338–343. https://doi.org/10.1111/j.1365-2133.2007.08029.x
Sanlorenzo M, Osella-Abate S, Ribero S et al (2015) Melanoma of the lower extremities: foot site is an independent risk factor for clinical outcome. Int J Dermatol 54:1023–1029. https://doi.org/10.1111/ijd.12730
Esmaeli B, Wang B, Deavers M et al (2000) Prognostic factors for survival in malignant melanoma of the eyelid skin. Ophthal Plast Reconstr Surg 16:250–257. https://doi.org/10.1097/00002341-200007000-00002
Ariyan S, Berger A (2017) Melanoma. Rubin JP, Neligan P. Plastic surgery, 4th edition, vol 1, chapter 29. Elsevier Health Sciences, p 559–94
Balch CM, Gershenwald JE, Soong SJ et al (2010) Multivariate analysis of prognostic factors among 2,313 patients with stage III melanoma: comparison of nodal micrometastases versus macrometastases. J Clin Oncol 28:2452–2459. https://doi.org/10.1200/JCO.2009.27.1627
Payette MJ, Katz M III, Grant-Kels JM (2009) Melanoma prognostic factors found in the dermatopathology report. Clin Dermatol 27:53–74. https://doi.org/10.1016/j.clindermatol.2008.09.006
Clark WH Jr, Elder DE, Guerry D IV, Epstein MN, Greene MH, Van Horn M (1984) A study of tumor progression: the precursor lesions of superficial spreading and nodular melanoma. Hum Pathol 15:1147–1165. https://doi.org/10.1016/s0046-8177(84)80310-x
Funding
None of the authors has a financial interest in any of the products, devices, or drugs mentioned in this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: Yong Chan Bae.
Data curation: Hoon-Soo Kim, Min-** Kwak, Changryul Claud Yi.
Writing—original draft: Sang Min Park, Ho Yoon Jeong.
Writing—review & editing: Yong Chan Bae, Changryul Claud Yi.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, S.M., Jeong, H.Y., Kim, HS. et al. Epidemiology and Prognostic Factors of Malignant Melanoma Among 200 Asian Patients from a Single Medical Center. Indian J Surg 86, 310–317 (2024). https://doi.org/10.1007/s12262-023-03857-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-023-03857-2