Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that primarily affects the elderly population. It gradually leads to memory loss, loss of thinking ability, and an overall cognitive decline. However, exhaustive literature is available to suggest that pathological changes in the brain occur decades before the first clinical symptoms appear. This review provides insight into the non-invasive biomarkers for early detection of AD that have been successfully studied in populations across the globe. These biomarkers have been detected in the blood, saliva, breath, and urine samples. Retinal imaging techniques are also reported. In this study, PubMed and Google scholar were the databases employed using keywords “Alzheimer’s disease,” “neurodegeneration,” “non-invasive biomarkers,” “early diagnosis,” “blood-based biomarkers,” and “preclinical AD,” among others. The evaluation of these biomarkers will provide early diagnosis of AD in the preclinical stages due to their positive correlation with brain pathology in AD. Early diagnosis with reliable and timely intervention can effectively manage this disease.

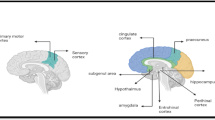
Source: created with BioRender.com
Similar content being viewed by others
Data Availability
Not applicable.
Abbreviations
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid-β
- APOE4:
-
Apolipoprotein E4
- APP:
-
Amyloid precursor protein
- BHT:
-
Butylated hydroxytoluene
- BBB:
-
Blood-brain barrier
- CSF:
-
Cerebrospinal fluid
- CN:
-
Cognitively normal
- ELISA:
-
Enzyme-linked immunosorbent assay
- FBP-PET:
-
18F-Florbetapir amyloid-β PET
- FTP-PET:
-
18F-Flortaucipir tau PET
- FUPLC-MS:
-
Fast ultra-high-performance liquid chromatography coupled with time-of-flight mass spectrometry
- GC-MS:
-
Gas chromatography-mass spectrometry
- GFAP:
-
Glial fibrillary acidic protein
- HS score:
-
Hyperspectral score
- 1H-NMR:
-
1H-Nuclear magnetic resonance
- LC-FTICR-MS:
-
Liquid chromatography/Fourier transform ion cyclotron resonance mass spectrometry
- Lf:
-
Lactoferrin
- MCI:
-
Mild cognitive impairment
- miRNA:
-
MicroRNA
- MCP-1:
-
Monocyte chemoattractant protein-1
- NfL:
-
Neurofilament light chain
- OCT:
-
Optical coherence tomography
- PET:
-
Positron emission tomography
- PS:
-
Presenilin
- PS-OCT:
-
Polarization-sensitive optical coherence tomography
- qRT-PCR:
-
Quantitative reverse transcription-polymerase chain reaction
- rHSI:
-
Retinal hyperspectral imaging, RNFL
- SCD:
-
Subjective cognitive decline
- sCR1:
-
Soluble complement receptor 1
- SD-OCT:
-
Spectral domain optical coherence tomography
- SMC:
-
Subjective memory complainers
- Simoa:
-
Single-molecule array
- TNFR-1:
-
Tumor necrosis factor-receptor-1
- UHPLC-MS:
-
Ultra-high-performance liquid chromatography-mass spectrometry
- UPLC:
-
Ultra-performance liquid chromatography
- VOC:
-
Volatile organic compound
- YKL-40:
-
Chitinase 3-like-protein-1
References
Samhan L, Alfarra A, Abu-Naser SS, Amassi I (2022) Classification of Alzheimer’s disease using convolutional neural networks. Int J Acad Inf Syst Res (IJAISR) 6:18–23
Nichols E, Szoeke CEI, Vollset SE et al (2019) Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol 18:88–106. https://doi.org/10.1016/S1474-4422(18)30403-4
Nichols E, Steinmetz JD, Vollset SE et al (2022) Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the global burden of disease study 2019. Lancet Public Health 7:e105–e125. https://doi.org/10.1016/S2468-2667(21)00249-8
Jahn H (2013) Memory loss in Alzheimer’s disease. In: Dialogues in Clin Neurosci 445–454. https://doi.org/10.31887/DCNS.2013.15.4/hjahn
Tilley L, Morgan KKN (1998) Reviews genetic risk factors in Alzheimer’s disease. J Clin Pathol: Mol Pathol 51(6):293–304. https://doi.org/10.1136/mp.51.6.293
Dubois B, Feldman HH, Jacova C et al (2007) Research criteria for the diagnosis of Alzheimer’s disease: revising the NINCDS-ADRDA criteria. Lancet Neurol 6(8):734–746. https://doi.org/10.1016/S1474-4422(07)70178-3
Reitz C, Brayne C, Mayeux R (2011) Epidemiology of Alzheimer disease. Nat Rev Neurol 7(3):137–152. https://doi.org/10.1038/nrneurol.2011.2
Stockmann J, Verberk IMW, Timmesfeld N et al (2021) Correction to: Amyloid-β misfolding as a plasma biomarker indicates risk for future clinical Alzheimer’s disease in individuals with subjective cognitive decline (Alzheimer’s Research & Therapy, (2020), 12, 1, (169). 10.1186/s13195-020-00738-8). Alzheimers Res Ther 13:1–13. https://doi.org/10.1186/s13195-021-00770-2
Rasmussen J, Langerman H (2019) Alzheimer’s disease – why we need early diagnosis. Degener Neurol Neuromuscul Dis 9:123–130. https://doi.org/10.2147/dnnd.s228939
Goodsell DS (2006) Amyloid-beta precursor protein. RCSB Protein Data Bank. https://doi.org/10.2210/rcsb_pdb/mom_2006_7
Gu L, Guo Z (2013) Alzheimer’s Aβ42 and Aβ40 peptides form interlaced amyloid fibrils. J Neurochem 126:305–311. https://doi.org/10.1111/jnc.12202
Fischer SK, Joyce A, Spengler M et al (2015) Emerging technologies to increase ligand binding assay sensitivitY. AAPS J 17:93–101. https://doi.org/10.1208/s12248-014-9682-8
Beyer L, Stocker H, Rujescu D et al (2022) Amyloid-beta misfolding and GFAP predict risk of clinical Alzheimer’s disease diagnosis within 17 years. Alzheimers Dement. https://doi.org/10.1002/alz.12745
Guo T, Noble W, Hanger DP (2017) Roles of tau protein in health and disease. Acta Neuropathol 133:665–704. https://doi.org/10.1007/s00401-017-1707-9
Moscoso A, Grothe MJ, Ashton NJ et al (2021) Time course of phosphorylated-tau181 in blood across the Alzheimer’s disease spectrum. Brain 144:325–339. https://doi.org/10.1093/brain/awaa399
Wang YL, Chen J, Du ZL et al (2021) Plasma p-tau181 level predicts neurodegeneration and progression to Alzheimer’s dementia: a longitudinal study. Front Neurol 12:. https://doi.org/10.3389/fneur.2021.695696
Barthélemy NR, Horie K, Sato C, Bateman RJ (2020) Blood plasma phosphorylated-tau isoforms track CNS change in Alzheimer’s disease. J Exp Med 217:1–12. https://doi.org/10.1084/JEM.20200861
Benedet AL, Leuzy A, Pascoal TA et al (2020) Stage-specific links between plasma neurofilament light and imaging biomarkers of Alzheimer’s disease. Brain 143:3793–3804. https://doi.org/10.1093/brain/awaa342
Giacomucci G, Mazzeo S, Bagnoli S et al (2022) Plasma neurofilament light chain as a biomarker of Alzheimer’s disease in Subjective Cognitive Decline and Mild Cognitive Impairment. J Neurol 269:4270–4280. https://doi.org/10.1007/s00415-022-11055-5
Silvestro S, Bramanti P, Mazzon E (2019) Role of miRNAs in Alzheimer’s disease and possible fields of application. Int J Mol Sci 20:1–26. https://doi.org/10.3390/ijms20163979
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297. https://doi.org/10.1016/S0092-8674(04)00045-5
Millan MJ (2017) Linking deregulation of non-coding RNA to the core pathophysiology of Alzheimer’s disease: an integrative review. Prog Neurobiol 156:1–68. https://doi.org/10.1016/j.pneurobio.2017.03.004
Serpente M, Fenoglio C, D’Anca M et al (2020) MiRNA profiling in plasma neural-derived small extracellular vesicles from patients with Alzheimer’s disease. Cells 9:. https://doi.org/10.3390/cells9061443
Liu Y, Xu Y, Yu M (2022) MicroRNA-4722–5p and microRNA-615–3p serve as potential biomarkers for Alzheimer’s disease. Exp Ther Med 23:. https://doi.org/10.3892/etm.2022.11166
Wang R, Chopra N, Nho K et al (2022) Human microRNA (miR-20b-5p) modulates Alzheimer’s disease pathways and neuronal function, and a specific polymorphism close to the MIR20B gene influences Alzheimer’s biomarkers. Mol Psychiatry 27:1256–1273. https://doi.org/10.1038/s41380-021-01351-3
Peña-Bautista C, Tarazona-Sánchez A, Braza-Boils A et al (2022) Plasma microRNAs as potential biomarkers in early Alzheimer disease expression. Sci Rep 12:1–10. https://doi.org/10.1038/s41598-022-19862-6
Sabry R, El Sharkawy RE, Gad NM (2020) MiRNA -483–5p as a potential noninvasive biomarker for early detection of Alzheimer’s disease. Egypt J Immunol 27(2):59–72
Nagaraj S, Laskowska-Kaszub K, Debski KJ et al (2017) Profile of 6 microRNA in blood plasma distinguish early stage Alzheimer’s disease patients from non-demented subjects. Oncotarget 8:16122–16143. https://doi.org/10.18632/oncotarget.15109
Ardura-Fabregat A, Boddeke EWGM, Boza-Serrano A et al (2017) Targeting neuroinflammation to treat alzheimer’s disease. CNS Drugs 31(12):1057–1082. https://doi.org/10.1007/s40263-017-0483-3
Prins S, de Kam ML, Teunissen CE, Groeneveld GJ (2022) Inflammatory plasma biomarkers in subjects with preclinical Alzheimer’s disease. Alzheimers Res Ther 14:. https://doi.org/10.1186/s13195-022-01051-2
Cristina d’Abramo C, D’Adamio L, Giliberto L (2020) Significance of blood and cerebrospinal fluid biomarkers for Alzheimer’s disease: sensitivity, specificity and potential for clinical use. In: Villa C (ed) Novel biomarkers in Alzheimer’s disease. J Pers Med MDPI, pp 67–106. https://doi.org/10.3390/jpm10030116
Muramori F, Kobayashi K, Nakamura I (1998) A quantitative study of neurofibrillary tangles, senile plaques and astrocytes in the hippocampal subdivisions and entorhinal cortex in Alzheimer’s disease, normal controls and non-Alzheimer neuropsychiatric diseases. Psychiatry Clin Neurosci 52:. https://doi.org/10.1111/j.1440-1819.1998.tb02706.x
Chatterjee P, Pedrini S, Stoops E et al (2021) Plasma glial fibrillary acidic protein is elevated in cognitively normal older adults at risk of Alzheimer’s disease. Transl Psychiatry 11:1–10. https://doi.org/10.1038/s41398-020-01137-1
Gross AL, Walker KA, Moghekar AR et al (2019) Plasma markers of inflammation linked to clinical progression and decline during preclinical AD. Front Aging Neurosci 11:1–9. https://doi.org/10.3389/fnagi.2019.00229
Morgan AR, Touchard S, Leckey C et al (2019) Inflammatory biomarkers in Alzheimer’s disease plasma. Alzheimers Dement 15:776–787. https://doi.org/10.1016/j.jalz.2019.03.007
Majeed A, Marwick B, Yu H et al (2021) Ophthalmic biomarkers for Alzheimer’s disease: a review. Front Aging Neurosci 13:1–17. https://doi.org/10.3389/fnagi.2021.720167
Koronyo-Hamaoui M, Koronyo Y, Ljubimov AV et al (2011) Identification of amyloid plaques in retinas from Alzheimer’s patients and noninvasive in vivo optical imaging of retinal plaques in a mouse model. Neuroimage 54:S204–S217. https://doi.org/10.1016/j.neuroimage.2010.06.020
Hadoux X, Hui F, Lim JKH et al (2019) Non-invasive in vivo hyperspectral imaging of the retina for potential biomarker use in Alzheimer’s disease. Nat Commun 10:1–12. https://doi.org/10.1038/s41467-019-12242-1
Budenz DL, Anderson DR, Varma R et al (2007) Determinants of normal retinal nerve fiber layer thickness measured by stratus OCT. Ophthalmology 114:1046–1052. https://doi.org/10.1016/j.ophtha.2006.08.046
Kirbas S, Turkyilmaz K, Anlar O et al (2013) Retinal nerve fiber layer thickness in patients with Alzheimer disease. J Neuroophthalmol 33:58–61. https://doi.org/10.1097/WNO.0b013e318267fd5f
Kwon JY, Yang JH, Han JS, Kim DG (2017) Analysis of the retinal nerve fiber layer thickness in alzheimer disease and mild cognitive impairment. Korean J Ophthalmol 31:548. https://doi.org/10.3341/kjo.2016.0118
Lim JKH, Li Q-X, He Z et al (2016) The eye as a biomarker for Alzheimer’s disease. Front Neurosci 10:. https://doi.org/10.3389/fnins.2016.00536
Pircher M, Hitzenberger CK, Schmidt-Erfurth U (2011) Polarization sensitive optical coherence tomography in the human eye. Prog Retin Eye Res 30:431–451. https://doi.org/10.1016/j.preteyeres.2011.06.003
Martins RN, Villemagne V, Sohrabi HR et al (2018) Alzheimer’s disease: a journey from amyloid peptides and oxidative stress, to biomarker technologies and disease prevention strategies-gains from AIBL and DIAN cohort studies. J Alzheimers Dis 62:965–992. https://doi.org/10.3233/JAD-171145
Mark NJA, Henrik I, Kaj Blennow Z et al (n.d.) Salivary biomarkers for alzheimer’s disease and related disorders. Neurol Ther 8:. https://doi.org/10.6084/m9.figshare.9792335
González-Sánchez M, Bartolome F, Antequera D et al (2020) Decreased salivary lactoferrin levels are specific to Alzheimer’s disease. J Clean Prod 57:. https://doi.org/10.1016/j.ebiom.2020.102834
Guo C, Yang ZH, Zhang S et al (2017) Intranasal lactoferrin enhances α-secretase-dependent amyloid precursor protein processing via the ERK1/2-CREB and HIF-1α pathways in an Alzheimer’s disease mouse model. Neuropsychopharmacology 42:2504–2515. https://doi.org/10.1038/npp.2017.8
Dominy SS, Lynch C, Ermini F et al (2019) Porphyromonas gingivalis in Alzheimer’s disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci Adv 5:1–22. https://doi.org/10.1126/sciadv.aau3333
Beydoun MA, Beydoun HA, Hossain S et al (2020) Clinical and bacterial markers of periodontitis and their association with incident all-cause and Alzheimer’s disease dementia in a large national survey. J Alzheimers Dis 75:157–172. https://doi.org/10.3233/jad-200064
Liang Q, Liu H, Li X, Zhang AH (2016) High-throughput metabolomics analysis discovers salivary biomarkers for predicting mild cognitive impairment and Alzheimer’s disease. RSC Adv 6:75499–75504. https://doi.org/10.1039/c6ra16802g
Yilmaz A, Geddes T, Han B et al (2017) Diagnostic biomarkers of Alzheimer’s disease as identified in saliva using 1H NMR-based metabolomics. J Alzheimers Dis 58:355–359. https://doi.org/10.3233/JAD-161226
Zheng J, Dixon RA, Li L (2012) Development of isotope labeling LC-MS for human salivary metabolomics and application to profiling metabolome changes associated with mild cognitive impairment. Anal Chem 84:10802–10811. https://doi.org/10.1021/ac3028307
Rummel NG, Butterfield DA (2022) Altered metabolism in Alzheimer disease brain: role of oxidative stress. Antioxid Redox Signal 36:1289–1305. https://doi.org/10.1089/ars.2021.0177
Tisch U, Schlesinger I, Ionescu R et al (2013) Detection of Alzheimer’s and Parkinson’s disease from exhaled breath using nanomaterial-based sensors. Nanomedicine 8:43–56. https://doi.org/10.2217/nnm.12.105
Emam S, Nasrollahpour M, Colarusso B et al (2020) Detection of presymptomatic Alzheimer’s disease through breath biomarkers. Alzheimers Dement: Diagn, Assess Dis Monit 12:1–9. https://doi.org/10.1002/dad2.12088
Kurbatova N, Garg M, Whiley L et al (2020) Urinary metabolic phenoty** for Alzheimer’s disease. Sci Rep 10:1–17. https://doi.org/10.1038/s41598-020-78031-9
Watanabe Y, Hirao Y, Kasuga K et al (2020) Urinary apolipoprotein C3 is a potential biomarker for Alzheimer’s disease. Dement Geriatr Cogn Dis Extra 10:94–104. https://doi.org/10.1159/000509561
Watanabe Y, Hirao Y, Kasuga K et al (2019) Molecular network analysis of the urinary proteome of Alzheimer’s disease patients. Dement Geriatr Cogn Dis Extra 9:53–65. https://doi.org/10.1159/000496100
Kimball BA, Wilson DA, Wesson DW (2016) Alterations of the volatile metabolome in mouse models of Alzheimer’s disease. Sci Rep 6:1–8. https://doi.org/10.1038/srep19495
Yu J, Kong L, Zhang A et al (2017) High-throughput metabolomics for discovering potential metabolite biomarkers and metabolic mechanism from the APPswe/PS1dE9 transgenic model of Alzheimer’s disease. J Proteome Res 16:3219–3228. https://doi.org/10.1021/acs.jproteome.7b00206
Schindler SE, Bollinger JG, Ovod V et al (2019) High-precision plasma β-amyloid 42/40 predicts current and future brain amyloidosis. Neurology 93:e1647–e1659. https://doi.org/10.1212/WNL.0000000000008081
Heneka MT, Carson MJ, el Khoury J et al (2015) Neuroinflammation in Alzheimer’s disease. Lancet Neurol 14:388–405. https://doi.org/10.1016/S1474-4422(15)70016-5
Acknowledgements
The authors are grateful to Mr. Rohit Joshi and Mr. Abbas Jawadwala for their input. We are thankful to the Institute of Chemical Technology, Mumbai, for the facilities and resources.
Author information
Authors and Affiliations
Contributions
Writing, literature survey, data analysis—Akash Haria; writing, drafting, conceptualization—Niyamat M.A Chimthanawala; editing, supervision—Sadhana Sathaye. All authors have reviewed and approved the final version of the manuscript prior to submission.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Methods
This study was conducted by reviewing papers from reputed journals that have been indexed in databases like PubMed and Google Scholar. The papers chosen were latest in the field of non-invasive biomarkers for early diagnosis of Alzheimer’s disease from 2010 onwards. The keywords selected were “Alzheimer’s disease,” “neurodegeneration,” “non-invasive biomarkers,” “early diagnosis,” “blood-based biomarkers,” “fluid-based biomarkers,” “early-AD,” “mild cognitive impairment,” and “preclinical AD,” among others, as per the scope of the study. Both clinical and preclinical studies of relevance were included. Studies regarding invasive techniques and imaging methods were not included.
Niyamat M. A. Chimthanawala and Akash Haria contributed equally.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chimthanawala, N.M.A., Haria, A. & Sathaye, S. Non-invasive Biomarkers for Early Detection of Alzheimer’s Disease: a New-Age Perspective. Mol Neurobiol 61, 212–223 (2024). https://doi.org/10.1007/s12035-023-03578-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03578-3