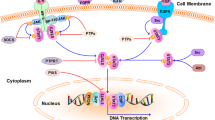
Abstract
Aromatase inhibitors (AIs) are the most effective endocrine treatment for estrogen receptor α-positive (ERα+) postmenopausal breast cancer. Identification of biomarkers that are able to predict AIs responsiveness of patients is a key for successful treatment. The currently used biomarkers for tamoxifen responsiveness, which including ERα as well as progesterone receptor can only predict part of the potential responders to AIs treatment. Sushi domain-containing protein 3 (SUSD3) is a potential novel biomarker of AIs responsiveness. The lack of SUSD3 expression in breast cancer tissue can be an important predictor for non-responsiveness to AI. Here we reviewed the property and function of SUSD3, its usage as a biomarker and the practicability for SUSD3 to become a target for immune therapy. We suggest this protein can be potentially measured or targeted for prevention, diagnostic, and therapeutic purposes for estrogen or progesterone-dependent disorders including breast cancer in women.
Similar content being viewed by others
References
Jemal, A., Bray, F., Center, M. M., Ferlay, J., Ward, E., & Forman, D. (2011). Global cancer statistics. CA: A Cancer Journal for Clinicians, 61, 69–90.
Bulun, S. E., Chen, D., Lu, M., et al. (2007). Aromatase excess in cancers of breast, endometrium and ovary. The Journal of Steroid Biochemistry and Molecular Biology, 106, 81–96.
Dumitrescu, R. G., & Cotarla, I. (2005). Understanding breast cancer risk—where do we stand in 2005? Journal of Cellular and Molecular Medicine, 9, 208–221.
Jones, M. E., Boon, W. C., McInnes, K., Maffei, L., Carani, C., & Simpson, E. R. (2007). Recognizing rare disorders: Aromatase deficiency. Nature Clinical Practice Endocrinology & Metabolism, 3, 414–421.
Simpson, E. R., & Davis, S. R. (2001). Minireview: Aromatase and the regulation of estrogen biosynthesis—some new perspectives. Endocrinology, 142, 4589–4594.
Bulun, S. E., Lin, Z., Imir, G., et al. (2005). Regulation of aromatase expression in estrogen-responsive breast and uterine disease: From bench to treatment. Pharmacological Reviews, 57, 359–383.
Shibaya, M., Matsuda, A., Hojo, T., Acosta, T. J., & Okuda, K. (2007). Expressions of estrogen receptors in the bovine corpus luteum: Cyclic changes and effects of prostaglandin F2alpha and cytokines. Journal of Reproduction and Development, 53, 1059–1068.
Zuo, Y., Berdeaux, R., & Frost, J. A. (2014). The RhoGEF Net1 is required for normal mammary gland development. Molecular Endocrinology, 28(12), 1948–1960.
Oh, Y., Rosenfeld, R., & Ingermann, A.R. (2009). Comprises nucleotide sequences coding insulin-like growth factor binding protein-3 and receptors for diagnosis, prevention and treatment of cancer; antitumor/anticarcinogenic agents. Google Patents, 2009.
Bulun, S. E., Chen, D., Moy, I., Brooks, D. C., & Zhao, H. (2012). Aromatase, breast cancer and obesity: A complex interaction. Trends in Endocrinology and Metabolism: TEM, 23, 83–89.
Diaz-Cruz, E. S., Sugimoto, Y., Gallicano, G. I., Brueggemeier, R. W., & Furth, P. A. (2011). Comparison of increased aromatase versus ERalpha in the generation of mammary hyperplasia and cancer. Cancer Research, 71, 5477–5487.
Yamamoto, M., Hosoda, M., Nakano, K., et al. (2014). p53 accumulation is a strong predictor of recurrence in estrogen receptor-positive breast cancer patients treated with aromatase inhibitors. Cancer Science, 105, 8.
Sabnis, G., Schayowitz, A., Goloubeva, O., Macedo, L., & Brodie, A. (2009). Trastuzumab reverses letrozole resistance and amplifies the sensitivity of breast cancer cells to estrogen. Cancer Research, 69, 1416–1428.
Van Asten, K., Neven, P., Lintermans, A., Wildiers, H., & Paridaens, R. (2014). Aromatase inhibitors in the breast cancer clinic: Focus on exemestane. Endocrine-Related Cancer, 21, R31–R49.
Subik, K., Lee, J. F., Baxter, L., et al. (2010). The expression patterns of ER, PR, HER2, CK5/6, EGFR, Ki-67 and AR by immunohistochemical analysis in breast cancer cell lines. Breast Cancer: Basic and Clinical Research, 4, 35–41.
Lim, S., Janzer, A., Becker, A., et al. (2010). Lysine-specific demethylase 1 (LSD1) is highly expressed in ER-negative breast cancers and a biomarker predicting aggressive biology. Carcinogenesis, 31, 512–520.
Reid, K. B., & Day, A. J. (1989). Structure-function relationships of the complement components. Immunology Today, 10, 177–180.
Norman, D. G., Barlow, P. N., Baron, M., Day, A. J., Sim, R. B., & Campbell, I. D. (1991). Three-dimensional structure of a complement control protein module in solution. Journal of Molecular Biology, 219, 717–725.
Gaboriaud, C., Rossi, V., Bally, I., Arlaud, G. J., & Fontecilla-Camps, J. C. (2000). Crystal structure of the catalytic domain of human complement c1s: A serine protease with a handle. EMBO Journal, 19, 1755–1765.
Moy, I., Todorovic, V., Dubash, A. D., et al. (2014). Estrogen-dependent sushi domain containing 3 regulates cytoskeleton organization and migration in breast cancer cells. Oncogene,. doi:10.1038/onc.2013.
Mello-Grand, M., Singh, V., Ghimenti, C., et al. (2010). Gene expression profiling and prediction of response to hormonal neoadjuvant treatment with anastrozole in surgically resectable breast cancer. Breast Cancer Research and Treatment, 121, 399–411.
Watson, A. P., Evans, R. L., & Egland, K. A. (2013). Multiple functions of sushi domain containing 2 (SUSD2) in breast tumorigenesis. Molecular Cancer Research: MCR, 11, 74–85.
Cui, H., Kamino, H., Nakamura, Y., et al. (2010). Regulation of apoptosis by p53-inducible transmembrane protein containing sushi domain. Oncology Reports, 24, 1193–1200.
Parris, T. Z., Danielsson, A., Nemes, S., et al. (2010). Clinical implications of gene dosage and gene expression patterns in diploid breast carcinoma. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research, 16, 3860–3874.
Hosford, S. R., & Miller, T. W. (2014). Clinical potential of novel therapeutic targets in breast cancer: CDK4/6, Src, JAK/STAT, PARP, HDAC, and PI3K/AKT/mTOR pathways. Journal of Pharmacogenomics and Personalized Medicine, 7, 203–215.
Yip, G. W. (2011). Breast cancer: Novel therapeutic targets. Recent Patents on Anticancer Drug Discovery, 6, 164–165.
Hanstein, B., Djahansouzi, S., Dall, P., Beckmann, M. W., & Bender, H. G. (2004). Insights into the molecular biology of the estrogen receptor define novel therapeutic targets for breast cancer. European Journal of Endocrinology, 150, 243–255.
Hudis, C. A. (2007). Trastuzumab-mechanism of action and use in clinical practice. The New England Journal of Medicine, 357, 39–51.
Kantoff, P. W., Higano, C. S., Shore, N. D., et al. (2010). Sipuleucel-T immunotherapy for castration-resistant prostate cancer. New England Journal of Medicine, 363, 411–422.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhenghong Yu and Enze Jiang have contributed equally.
Rights and permissions
About this article
Cite this article
Yu, Z., Jiang, E., Wang, X. et al. Sushi Domain-Containing Protein 3: A Potential Target for Breast Cancer. Cell Biochem Biophys 72, 321–324 (2015). https://doi.org/10.1007/s12013-014-0480-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0480-9