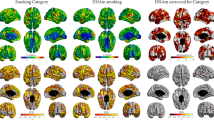
Abstract
Active and passive exposure to tobacco smoke may increase risk of cognitive decline. However, effects of enhanced the aging process on the association of urinary nicotine metabolites with cognitive impairment remain unclear. In this study, 6657 Chinese older adults completed the physical examinations and cognitive tests. We measured urinary nicotine metabolite levels, mitochondrial DNA copy number (mtDNA-CN), and relative telomere length (RTL) and analyzed effects of urinary nicotine metabolites and their interaction with mtDNA-CN or RTL on cognitive impairment by generalized linear models and qg-computation, respectively. Each 1-unit increase in urinary 3-OHCot, 3-OHCotGluc, CotGluc, or NicGluc levels corresponded to a 1.05-, 1.09-, 1.04-, and 0.90-fold increased risk of cognitive impairment. Each 1-quantile increment in the mixture level of 8 nicotine metabolites corresponded to an increment of 1.40- and 1.34-fold risk of cognitive impairment in individuals with longer RTL or low mtDNA-CN. Urinary 3-OHCotGluc and RTL or mtDNA-CN exhibited an additive effect on cognitive impairment in addition to the mixture of 8 nicotine metabolites and mtDNA-CN. The findings suggested that aging process may increase the risk of tobacco-related cognitive impairment.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Abbreviations
- AGR:
-
Albumin/globulin ratio
- AP:
-
The attributable proportion due to interaction
- BMI:
-
Body mass index
- Cot:
-
Cotinine
- CotGluc:
-
Cotinine N-β-d-glucuronide
- CNO:
-
(S)-cotinine N-oxide
- 3-OHCot:
-
Trans-3′-hydroxy cotinine
- HyPyBut:
-
Rac 4-hydroxy-4-(3-pyridyl)butanoic acid dicyclohexylamine salt
- mtDNA-CN:
-
Mitochondrial DNA copy number
- NNO:
-
(1′S, 2′S)-nicotine-1′-oxide
- NicGluc:
-
Nicotine-N-β-glucuronide
- NorNic:
-
(R, S)-nornicotine
- NorCot:
-
(R, S)-nornicotine
- 3-OHCotGluc:
-
Trans-3′-hydroxy cotinine O-β-D-glucuronide
- OR:
-
Odd ratios
- RERI:
-
Relative excess risk due to interaction
- RTL:
-
Relative telomere length
- 95%CI:
-
95% Confidence interval
References
Akhtar WZ, Andresen EM, Cannell MB, Xu X (2013) Association of blood cotinine level with cognitive and physical performance in non-smoking older adults. Environ Res 121:64–70. https://doi.org/10.1016/j.envres.2012.10.013
Akter R, Rahman MH, Kaushik D, Mittal V, Uivarosan D, Nechifor AC, Behl T, Karthika C, Stoicescu M, Munteanu MA, Bustea C, Bungau S (2021) Chemo-preventive action of resveratrol: suppression of p53-a molecular targeting approach. Molecules 26(17):5325. https://doi.org/10.3390/molecules26175325
Argalasova L, Zitnanova I, Vondrova D, Dvorakova M, Laubertova L, Jurkovicova J, Stofko J, Weitzman M, Waczulikova I, Simko M (2019) Self-reported exposure to ets (environmental tobacco smoke), urinary cotinine, and oxidative stress parameters in pregnant women-the pilot study. Int J Environ Res Public Health 16. https://doi.org/10.3390/ijerph16091656
Armanios M (2013) Telomeres and age-related disease: how telomere biology informs clinical paradigms. J Clin Invest 123:996–1002. https://doi.org/10.1172/jci66370
Ashar FN, Zhang Y, Longchamps RJ, Lane J, Moes A, Grove ML, Mychaleckyj JC, Taylor KD, Coresh J, Rotter JI, Boerwinkle E, Pankratz N, Guallar E, Arking DE (2017) Association of mitochondrial DNA copy number with cardiovascular disease. JAMA Cardiol 2:1247–1255. https://doi.org/10.1001/jamacardio.2017.3683
Bai A, ** Y, Huang Y (2020) Impact of secondhand smoke exposure on cognitive function among middle-aged and older women in china: findings from three waves of the china health and retirement longitudinal study. BMJ Open 10:e039824. https://doi.org/10.1136/bmjopen-2020-039824
Baierle M, Nascimento SN, Moro AM, Brucker N, Freitas F, Gauer B, Durgante J, Bordignon S, Zibetti M, Trentini CM, Duarte MM, Grune T, Breusing N, Garcia SC (2015) Relationship between inflammation and oxidative stress and cognitive decline in the institutionalized elderly. Oxid Med Cell Longev 2015:804198. https://doi.org/10.1155/2015/804198
Benowitz NL, Hukkanen J, Jacob P 3rd (2009) Nicotine chemistry, metabolism, kinetics and biomarkers. Handb Exp Pharmacol:29–60. https://doi.org/10.1007/978-3-540-69248-5_2
Buccafusco JJ, Terry AV Jr (2003) The potential role of cotinine in the cognitive and neuroprotective actions of nicotine. Life Sci 72:2931–2942. https://doi.org/10.1016/s0024-3205(03)00226-1
Castellani CA, Longchamps RJ, Sun J, Guallar E, Arking DE (2020) Thinking outside the nucleus: mitochondrial DNA copy number in health and disease. Mitochondrion 53:214–223. https://doi.org/10.1016/j.mito.2020.06.004
Cawthon RM (2009) Telomere length measurement by a novel monochrome multiplex quantitative pcr method. Nucleic Acids Res 37:e21. https://doi.org/10.1093/nar/gkn1027
Chahine MN, Toupance S, El-Hakim S, Labat C, Gautier S, Moussallem T, Yared P, Asmar R, Benetos A (2019) Telomere length and age-dependent telomere attrition: The blood-and-muscle model (1). Can J Physiol Pharmacol 97:328–334. https://doi.org/10.1139/cjpp-2018-0582
Chakravarti D, LaBella KA, DePinho RA (2021) Telomeres: history, health, and hallmarks of aging. Cell 184:306–322. https://doi.org/10.1016/j.cell.2020.12.028
Choi WJ, Lee JW, Cho AR, Lee YJ (2019) Dose-dependent toxic effect of cotinine-verified tobacco smoking on systemic inflammation in apparently healthy men and women: a nationwide population-based study. Int J Environ Res Public Health 16. https://doi.org/10.3390/ijerph16030503
China Development Report (2020) Trend of population aging in china and the strategy. https://wenku.baidu.com/view/0d2fb08a65ec102de2bd960590c69ec3d4bbdb62.html. Accessed 2021.09.08
Dioni L, Hoxha M, Nordio F, Bonzini M, Tarantini L, Albetti B, Savarese A, Schwartz J, Bertazzi PA, Apostoli P, Hou L, Baccarelli A (2011) Effects of short-term exposure to inhalable particulate matter on telomere length, telomerase expression, and telomerase methylation in steel workers. Environ Health Perspect 119:622–627. https://doi.org/10.1289/ehp.1002486
Dong Y, Bi W, Zheng K, Zhu E, Wang S, **ong Y, Chang J, Jiang J, Liu B, Lu Z, Cheng Y (2020) Nicotine prevents oxidative stress-induced hippocampal neuronal injury through α7-nachr/erk1/2 signaling pathway. Front Mol Neurosci 13:557647. https://doi.org/10.3389/fnmol.2020.557647
Echeverria V, Zeitlin R (2012) Cotinine: a potential new therapeutic agent against alzheimer’s disease. CNS Neurosci Ther 18:517–523. https://doi.org/10.1111/j.1755-5949.2012.00317.x
Feigin VL, Nichols E, Alam T, Bannick MS, Beghi E, Blake N, Culpepper WJ, Ray Dorsey E, Elbaz A (2019) Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol 18:459–480. https://doi.org/10.1016/s1474-4422(18)30499-x
Frost F, Roach MJ, Kushner I, Schreiber P (2005) Inflammatory c-reactive protein and cytokine levels in asymptomatic people with chronic spinal cord injury. Arch Phys Med Rehabil 86:312–317. https://doi.org/10.1016/j.apmr.2004.02.009
Gao X, Gào X, Zhang Y, Breitling LP, Schöttker B, Brenner H (2017) Associations of self-reported smoking, cotinine levels and epigenetic smoking indicators with oxidative stress among older adults: a population-based study. Eur J Epidemiol 32:443–456. https://doi.org/10.1007/s10654-017-0248-9
Ge S, Tang X, Wei Z, Dune L, Liu T, Li J, Li C (2020) Smoking and cognitive function among middle-aged adults in china: findings from the china health and retirement longitudinal study baseline survey. J Addict Nurs 31:E5-e12. https://doi.org/10.1097/jan.0000000000000352
Gillum RF, Kwagyan J, Obisesan TO (2011) Smoking, cognitive function and mortality in a U.S. National cohort study. Int J Environ Res Public Health 8:3628–3636. https://doi.org/10.3390/ijerph8093628
Granger DN, Senchenkova E (2010) Inflammation and the microcirculation. Morgan & Claypool Life, San Rafael (CA)
Hallal PC, Andersen LB, Bull FC, Guthold R, Haskell W, Ekelund U, Group, LPASW (2012) Global physical activity levels: Surveillance progress, pitfalls, and prospects. Lancet 380:247–257
He F, Li T, Lin J, Li F, Zhai Y, Zhang T, Gu X, Zhao G (2020) Passive smoking exposure in living environments reduces cognitive function: a prospective cohort study in older adults. Int J Environ Res Public Health 17. https://doi.org/10.3390/ijerph17041402
He J, Pan H, Liang W, **ao D, Chen X, Guo M, He J (2017) Prognostic effect of albumin-to-globulin ratio in patients with solid tumors: a systematic review and meta-analysis. J Cancer 8:4002–4010. https://doi.org/10.7150/jca.21141
Hou L, Wang S, Dou C, Zhang X, Yu Y, Zheng Y, Avula U, Hoxha M, Díaz A, McCracken J, Barretta F, Marinelli B, Bertazzi PA, Schwartz J, Baccarelli AA (2012) Air pollution exposure and telomere length in highly exposed subjects in Bei**g, china: a repeated-measure study. Environ Int 48:71–77. https://doi.org/10.1016/j.envint.2012.06.020
Hukkanen J, Jacob P 3rd, Benowitz NL (2005) Metabolism and disposition kinetics of nicotine. Pharmacol Rev 57:79–115. https://doi.org/10.1124/pr.57.1.3
Jacobson GA, Ferguson SG (2014) Relationship between cotinine and trans-3’-hydroxycotinine glucuronidation and the nicotine metabolite ratio in Caucasian smokers. Biomarkers 19:679–683. https://doi.org/10.3109/1354750x.2014.966254
Jain RB (2021) Comparative analysis of the concentrations of serum cotinine and hydroxycotinine for us children, adolescents, and adults: impact of exposure to environmental tobacco smoke at home and other indoor environments. Environ Sci Pollut Res 28:17627–17635. https://doi.org/10.1007/s11356-020-11838-1
Kauppila TES, Kauppila JHK, Larsson NG (2017) Mammalian mitochondria and aging: an update. Cell Metab 25:57–71. https://doi.org/10.1016/j.cmet.2016.09.017
Keil AP, Buckley JP, O’Brien KM, Ferguson KK, Zhao S, White AJ (2020) A quantile-based g-computation approach to addressing the effects of exposure mixtures. Environ Health Perspect 128:47004. https://doi.org/10.1289/EHP5838
Khorasanchi Z, Bahrami A, Avan A, Jaberi N, Rezaey M, Bahrami-Taghanaki H, Ferns GA, Ghayour-Mobarhan M (2019) Passive smoking is associated with cognitive and emotional impairment in adolescent girls. J Gen Psychol 146:68–78. https://doi.org/10.1080/00221309.2018.1535485
Kim M, Liotta EM, Zee PC, Ganger DR, Ladner DP, Karmarkar A, Peipert JD, Sorond FA, Prabhakaran S, Reid KJ, Naidech AM, Maas MB (2019) Impaired cognition predicts the risk of hospitalization and death in cirrhosis. Ann Clin Transl Neurol 6:2282–2290. https://doi.org/10.1002/acn3.50924
Koyama T, Kuriyama N, Ozaki E, Matsui D, Watanabe I, Miyatani F, Kondo M, Tamura A, Kasai T, Ohshima Y, Yoshida T, Tokuda T, Mizuta I, Mizuno S, Yamada K, Takeda K, Matsumoto S, Nakagawa M, Mizuno T, Watanabe Y (2016) Serum albumin to globulin ratio is related to cognitive decline via reflection of homeostasis: a nested case-control study. BMC Neurol 16:253. https://doi.org/10.1186/s12883-016-0776-z
Lee HC, Wei YH (2000) Mitochondrial role in life and death of the cell. J Biomed Sci 7(2–15):25424
Lee JW, Park KD, Im JA, Kim MY, Lee DC (2010) Mitochondrial DNA copy number in peripheral blood is associated with cognitive function in apparently healthy elderly women. Clin Chim Acta 411:592–596. https://doi.org/10.1016/j.cca.2010.01.024
Li T, Liu W, Yue YJ, Lu SY, Nie LL, Yang XF, Zhu QQ, Zhu B, Wang L, Zhu FQ, Zhou L, Zhang JF, Gao EW, He KW, Liu L, Ye F, Liu JJ, Yuan J, Wang L (2021) Nonlinear dose-response relation between urinary levels of nicotine and its metabolites and cognitive impairment among an elderly population in China. Ecotoxicol Environ Saf 224:112706. https://doi.org/10.1016/j.ecoenv.2021.112706
Liang R, Li J, Tang X, Liu Y (2019) The prognostic role of preoperative systemic immune-inflammation index and albumin/globulin ratio in patients with newly diagnosed high-grade glioma. Clin Neurol Neurosurg 184:105397. https://doi.org/10.1016/j.clineuro.2019.105397
Lin Y, Qiu X, Yu N, Yang Q, Araujo JA, Zhu Y (2016) Urinary metabolites of polycyclic aromatic hydrocarbons and the association with lipid peroxidation: a biomarker-based study between los angeles and bei**g. Environ Sci Technol 50:3738–3745. https://doi.org/10.1021/acs.est.5b04629
Liu L, Liu W, Nie L, Guo Z, Luo Y, Chen W, Liu W, Zhu F, Wang L, Zhang J, Wang X, Li T, Gao E, Zhou L, He K, Huang Y, Yuan C, Zhu Q, Ye F, Liu J (2020) Study design and baseline characteristics of Shenzhen ageing-related disorder cohort in China. BMJ Open 10:e034317. https://doi.org/10.1136/bmjopen-2019-034317
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative pcr and the 2(t)(-delta delta c) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Livingston G, Huntley J, Sommerlad A, Ames D, Ballard C, Banerjee S, Brayne C, Burns A, Cohen-Mansfield J, Cooper C, Costafreda SG, Dias A, Fox N, Gitlin LN, Howard R, Kales HC, Kivimäki M, Larson EB, Ogunniyi A, Orgeta V, Ritchie K, Rockwood K, Sampson EL, Samus Q, Schneider LS, Selbæk G, Teri L, Mukadam N (2020) Dementia prevention, intervention, and care: 2020 report of the lancet commission. Lancet 396:413–446. https://doi.org/10.1016/s0140-6736(20)30367-6
Maeda S, Takeya Y, Oguro R, Akasaka H, Ryuno H, Kabayama M, Yokoyama S, Nagasawa M, Fujimoto T, Takeda M, Onishi-Takeya M, Itoh N, Takami Y, Yamamoto K, Sugimoto K, Inagaki H, Ogawa M, Nakagawa T, Yasumoto S, Masui Y, Arai Y, Ishizaki T, Ikebe K, Gondo Y, Kamide K, Rakugi H (2019) Serum albumin/globulin ratio is associated with cognitive function in community-dwelling older people: the septuagenarians, octogenarians, nonagenarians investigation with centenarians study. Geriatr Gerontol Int 19:967–971. https://doi.org/10.1111/ggi.13751
Maldifassi MC, Atienza G, Arnalich F, López-Collazo E, Cedillo JL, Martín-Sánchez C, Bordas A, Renart J, Montiel C (2014) A new irak-m-mediated mechanism implicated in the anti-inflammatory effect of nicotine via α7 nicotinic receptors in human macrophages. PLoS ONE 9:e108397. https://doi.org/10.1371/journal.pone.0108397
Meyer JN, Leung MCK, Rooney JP, Sendoel A, Hengartner MO, Kisby GE, Bess AS (2013) Mitochondria as a target of environmental toxicants. Toxicol Sci 134:1–17. https://doi.org/10.1093/toxsci/kft102
Mons U, Schöttker B, Müller H, Kliegel M, Brenner H (2013) History of lifetime smoking, smoking cessation and cognitive function in the elderly population. Eur J Epidemiol 28:823–831. https://doi.org/10.1007/s10654-013-9840-9
Pieters N, Janssen BG, Dewitte H, Cox B, Cuypers A, Lefebvre W, Smeets K, Vanpoucke C, Plusquin M, Nawrot TS (2016) Biomolecular markers within the core axis of aging and particulate air pollution exposure in the elderly: a cross-sectional study. Environ Health Perspect 124:943–950. https://doi.org/10.1289/ehp.1509728
Piller M, Gilch G, Scherer G, Scherer M (2014) Simple, fast and sensitive lc–ms/ms analysis for the simultaneous quantification of nicotine and 10 of its major metabolites. J Chromatogr B 951–952:7–15. https://doi.org/10.1016/j.jchromb.2014.01.025
Qin F, Sun C, Ma Y, Liu C, Bai F, Liu N, Luo X, Liu Q, Liu Z (2018) Is low serum albumin concentration associated with the adverse cardiovascular events in stable coronary heart disease? Int J Cardiol 266:260. https://doi.org/10.1016/j.ijcard.2018.02.034
Rovio SP, Pihlman J, Pahkala K, Juonala M, Magnussen CG, Pitkänen N, Ahola-Olli A, Salo P, Kähönen M, Hutri-Kähönen N, Lehtimäki T, Jokinen E, Laitinen T, Taittonen L, Tossavainen P, Viikari JSA, Raitakari OT (2020) Childhood exposure to parental smoking and midlife cognitive function. Am J Epidemiol 189:1280–1291. https://doi.org/10.1093/aje/kwaa052
Smith L, Luchini C, Demurtas J, Soysal P, Stubbs B, Hamer M, Nottegar A, Lawlor RT, Lopez-Sanchez GF, Firth J, Koyanagi A, Roberts J, Willeit P, Waldhoer T, Loosemore M, Abbs AD, Johnstone J, Yang L, Veronese N (2019) Telomere length and health outcomes: an umbrella review of systematic reviews and meta-analyses of observational studies. Ageing Res Rev 51:1–10. https://doi.org/10.1016/j.arr.2019.02.003
Stokes AC, Weiss J, Lundberg DJ, **e W, Kim JK, Preston SH, Crimmins EM (2020) Estimates of the association of dementia with us mortality levels using linked survey and mortality records. JAMA Neurol 77:1543–1550. https://doi.org/10.1001/jamaneurol.2020.2831
Sun N, Youle RJ, Finkel T (2016) The mitochondrial basis of aging. Mol Cell 61:654–666. https://doi.org/10.1016/j.molcel.2016.01.028
World Health Organization (2020) Ageing and health. https://www.Who.Int/china/health-topics/ageing. Accessed 2021.07.06
Wang H, Chen H, Han S, Fu Y, Tian Y, Liu Y, Wang A, Hou H, Hu Q (2021) Decreased mitochondrial DNA copy number in nerve cells and the hippocampus during nicotine exposure is mediated by autophagy. Ecotoxicol Environ Saf 226:112831. https://doi.org/10.1016/j.ecoenv.2021.112831
Wu S, Li X, Meng S, Fung T, Chan AT, Liang G, Giovannucci E, De Vivo I, Lee JH, Nan H (2019) Fruit and vegetable consumption, cigarette smoke, and leukocyte mitochondrial DNA copy number. Am J Clin Nutr 109:424–432. https://doi.org/10.1093/ajcn/nqy286
Yang T, Wang H, **ong Y, Chen C, Duan K, Jia J, Ma F (2020) Vitamin d supplementation improves cognitive function through reducing oxidative stress regulated by telomere length in older adults with mild cognitive impairment: a 12-month randomized controlled trial. J Alzheimers Dis 78:1509–1518. https://doi.org/10.3233/jad-200926
Yeh JK, Lin MH, Wang CY (2019) Telomeres as therapeutic targets in heart disease. JACC Basic Transl Sci 4:855–865. https://doi.org/10.1016/j.jacbts.2019.05.009
Zehravi M, Kabir J, Akter R, Malik S, Ashraf GM, Tagde P, Ramproshad S, Mondal B, Rahman MH, Mohan AG, Cavalu S (2022) A prospective viewpoint on neurological diseases and their biomarkers. Molecules 27(11):3516. https://doi.org/10.3390/molecules27113516
Acknowledgements
We thank all the volunteers who participated in the study and the doctors at the local hospital for screening patients with mild cognitive impairment and dementia using the Mini Mental State Examination score.
Funding
This project is supported by Shenzhen Basic Research Project (JCYJ20190807103401672), Guangdong Medical Science and Technology Research Project (A2020144), Shenzhen Key Medical Discipline Construction Fund (SZXK069), Sanming Project of Medicine in Shenzhen (SZSM201611090).
Author information
Authors and Affiliations
Contributions
J Hou: Investigation, Data curation, Methodology, Formal analysis, Methodology, Writing-Original Draft.
W Liu and B Zhu: investigation, formal analysis, methodology, methodology and writing-review and editing. C Yuan, L Wang, E Gao, J Zhang, and Y Huang: investigation, data curation, writing-review and editing. Q Zhu and T Li: investigation, methodology, formal analysis, and writing-review and editing. D Wu, F Zhu, C Huang, J Huang, S Lai and S Lu: investigation, formal analysis, writing-review & editing. L Nie, L Zhou, F Ye, and X Yang: investigation, methodology, writing-review& editing. J Yuan: conceptualization, methodology, supervision, writing-reviewing and editing. J Liu: conceptualization, methodology, investigation, validation, supervision, funding acquisition, project administration, writing-original draft.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Medical Ethics Research Committee of the Shenzhen Center for Disease Control and Prevention. All participants signed an informed consent form prior to participation in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jian Hou, Chao Huang and Bo Zhu are the co-first authors.
Highlights
• Urinary nicotine metabolites were related to cognitive impairment.
• mtDNA-CN, RTL, or serum AGR was associated with cognitive impairment.
• Urinary 3-OHCotGluc and RTL had an additive effect on cognitive impairment.
• Nicotine metabolite mixture with low mtDNA-CN may synergistically decline cognition.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hou, J., Huang, C., Zhu, B. et al. Effect modification by aging on the associations of nicotine exposure with cognitive impairment among Chinese elderly. Environ Sci Pollut Res 30, 9530–9542 (2023). https://doi.org/10.1007/s11356-022-22392-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22392-3