Abstract
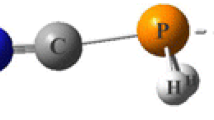
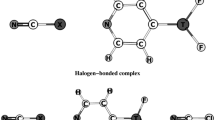
The substituent effects on cooperativity between pnicogen and halogen bonds in ternary complexes involving pyrimidine with substituents X in the 5-position (X = CN, NC, CF3, Br, Cl, F, H, CH3, C(CH3)3, NH2, N(CH3)2), H2FP, and ClF are investigated using theoretical calculations at the MP2/aug-cc-pVDZ level. The reduced density gradient (RDG) analysis indicates that the halogen and pnicogen interactions clearly stand out as attractive interactions. The calculated synergetic energies in ternary complexes are positive, which demonstrates the antagonist effect of pnicogen and halogen interactions upon each other. According to geometrical parameters, binding energies, molecular electrostatic potentials (MEPs), and the results of natural bond orbital (NBO), atoms in molecules (AIM), and nuclear magnetic resonance (NMR) analyses, pnicogen and halogen bonds are weakened in the ternary complexes where two pnicogen and halogen bonds coexist. The results indicate reduced electron density values, second-order perturbation energies, charge transfer values, and two-bonded spin–spin coupling constants for both pnicogen and halogen bonds in the ternary complex as compared to the isolated binary systems. Also, the results show that substituent effects on cooperativity between N…P pnicogen and N…Cl halogen bonds in considered system can be expressed by Hammett constants.










Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Knowles RR, Jacobsen EN (2010) Attractive noncovalent interactions in asymmetric catalysis: links between enzymes and small molecule catalysts. Proc Natl Acad Sci U S A 107:20678–20685
Desiraju GR (2010) Crystal engineering: a brief overview. J Chem Sci 122:667–675
Guthrie F (1863) XXVIII.—on the iodide of iodammonium. J Chem Soc 16:239–244
Dumas JM, Peurichard H, Gomel M (1978) CX4...base interactions as models of weak charge-transfer interactions: comparison with strong charge-transfer and hydrogen-bond interactions. J Chem Res (S) 2:54–57
Bent HA (1968) Structural chemistry of donor-acceptor interactions. Chem Rev 68:587–648
Hassel O (1970) Structural aspects of interatomic charge-transfer bonding. Science 170:497–502
Auffinger P, Hays FA, Westhof E, Ho PS (2004) Halogen bonds in biological molecules. Proc Natl Acad Sci U S A 101:16789–16794
Metrangolo P, Neukirch H, Pilati T, Resnati G (2005) Halogen bonding based recognition processes: a world parallel to hydrogen bonding. Acc Chem Res 38:386–395
Persch E, Dumele O, Diederich F (2015) Molecular recognition in chemical and biological systems. Angew Chem Int Ed 54:3290–3327
Wilcken R, Zimmermann MO, Lange A, Joerger AC, Boeckler FM (2013) Principles and applications of halogen bonding in medicinal chemistry and chemical biology. J Med Chem 56:1363–1388
Metrangolo P, Meyer F, Pilati T, Resnati G, Terraneo G (2008) Halogen bonding in supramolecular chemistry. Angew Chem Int Ed 47:6114–6127
Cavallo G, Metrangolo P, Milani R, Pilati T, Priimagi A, Resnati G, Terraneo G (2016) The halogen bond. Chem Rev 116:2478–2601
Bulfield D, Huber SM (2016) Halogen bonding in organic synthesis and organocatalysis. Chem Eur J 22:14434–14450
Berger G, Soubhye J, Meyer F (2015) Halogen bonding in polymer science: from crystal engineering to functional supramolecular polymers and materials. Polym Chem 6:3559–3580
Alcock NW (1972) Secondary bonding to nonmetallic elements. In Advances in inorganic chemistry and radiochemistry. Academic Press.
Clark T, Hennemann M, Murray JS, Politzer P (2007) Halogen bonding: the σ-hole. J Mol Model 13:291–296
Murray JS, Lane P, Clark T, Politzer P (2007) σ-hole bonding: molecules containing group VI atoms. J Mol Model 13:1033–1038
Murray JS, Lane PA, Politzer P (2007) A predicted new type of directional noncovalent interaction. Int J Quantum Chem 107:2286–2292
Murray JS, Lane P, Politzer P (2009) Expansion of the σ-hole concept. J Mol Model 15:723–729
Politzer P, Murray JS, Clark T (2010) Halogen bonding: an electrostatically-driven highly directional noncovalent interaction. Phys Chem Chem Phys 12:7748–7757
Politzer P, Murray JS, Clark T (2013) Halogen bonding and other σ-hole interactions: a perspective. Phys Chem Chem Phys 15:11178–11189
Politzer P, Murray JS, Clark T, Resnati G (2017) The σ-hole revisited. Phys Chem Chem Phys 19:32166–32178
Hill WE, Silva-Trivino LM (1978) Preparation and characterization of di (tertiary phosphines) with electronegative substituents. 1. Symmetrical derivatives Inorg Chem 17:2495–2498
Hill WE, Silva-Trivino LM (1979) Preparation and characterization of di (tertiary phosphines) with electronegative substituents. 2. Unsymmetrical derivatives Inorg Chem 18:361–364
Kilian P, Slawin AM, Woollins JD (2003) Naphthalene-1, 8-diyl bis (halogenophosphanes): novel syntheses and structures of useful synthetic building blocks. Chem Eur J 9:215–222
Zahn S, Frank R, Hey-Hawkins E, Kirchner B (2011) Pnicogen bonds: a new molecular linker? Chem Eur J 17:6034–6038
Bauza A, Quinonero D, Deya PM, Frontera A (2012) Pnicogen–π complexes: theoretical study and biological implications. Phys Chem Chem Phys 14:14061–14066
Saparov B, He H, Zhang X, Greene R, Bobev S (2010) Synthesis, crystallographic and theoretical studies of the new Zintl phases Ba2Cd2Pn3 (Pn = As, Sb), and the solid solutions (Ba1–xSrx)2Cd2Sb3 and Ba2Cd2(Sb1–xAsx)3. Dalton Trans 39:1063–1070
Vickaryous WJ, Healey ER, Berryman OB, Johnson DW (2005) Synthesis and characterization of two isomeric, self-assembled arsenic− thiolate macrocycles. Inorg Chem 44:9247–9252
Alkorta I, Elguero J, Del Bene JE (2013) Pnicogen-bonded cyclic trimers (PH2X)3 with X = F, Cl, OH, NC, CN, CH3, H, and BH2. J Phys Chem A 117:4981–4987
Adhikari U, Scheiner S (2011) Comparison of P⋯ D (D= P, N) with other noncovalent bonds in molecular aggregates. J Chem Phys 135:184306
Setiawan D, Kraka E, Cremer D (2015) Strength of the pnicogen bond in complexes involving group Va elements N, P, and As. J Phys Chem A 119:1642–1656
Politzer P, Murray JS, Janjić GV, Zarić SD (2014) σ-Hole interactions of covalently-bonded nitrogen, phosphorus and arsenic: a survey of crystal structures. Crystals 4:12–31
Sarkar S, Pavan MS, Row TG (2015) Experimental validation of ‘pnicogen bonding’ in nitrogen by charge density analysis. Phys Chem Chem Phys 17:2330–2334
Solimannejad M, Gholipour A (2013) Revealing substituent effects on the concerted interaction of pnicogen, chalcogen, and halogen bonds in substituted s-triazine ring. Struct Chem 24:1705–1711
Scheiner S (2011) Effects of substituents upon the P···N noncovalent interaction: the limits of its strength. J Phys Chem A 115:11202–11209
Bagheri S, Masoodi HR, Akrami-Mohajeri AR (2017) A theoretical survey of substituent effects on the properties of pnicogen and hydrogen bonds in cationic complexes of PH4+ with substituted benzonitrile. J Mol Graph Model 77:64–71
Del Bene JE, Alkorta I, Sanchez-Sanz G, Elguero J (2011) 31P–31P spin–spin coupling constants for pnicogen homodimers. Chem Phys Lett 512:184–187
Del Bene JE, Alkorta I, Sanchez-Sanz G, Elguero J (2012) Structures, binding energies, and spin–spin coupling constants of geometric isomers of pnicogen homodimers (PHFX)2, X= F, Cl, CN, CH3, NC. J Phys Chem A 116:3056–3060
Shukla R, Chopra D (2016) “Pnicogen bonds” or “chalcogen bonds”: exploiting the effect of substitution on the formation of P⋯Se noncovalent bonds. Phys Chem Chem Phys 18:13820–13829
Shukla R, Chopra D (2015) Exploring the role of substitution on the formation of Se··· O/N noncovalent bonds. J Phys Chem B 119:14857–14870
George J, Deringer VL, Dronskowski R (2014) Cooperativity of halogen, chalcogen, and pnictogen bonds in infinite molecular chains by electronic structure theory. J Phys Chem A 118:3193–3200
Esrafili MD, Vakili M, Solimannejad M (2014) Cooperative effects in pnicogen bonding:(PH2F)2–7 and (PH2Cl)2–7 clusters. Chem Phys Lett 609:37–41
Tondro T, Roohi H (2020) Substituent effects on the halogen and pnictogen bonds characteristics in ternary complexes 4-YPhNH2··· PH2 F··· ClX (Y= H, F, CN, CHO, NH2, CH3, NO2 and OCH3, and X= F, OH, CN, NC, FCC and NO2): a theoretical study. J Chem Sci 132:1–21
Alkorta I, Blanco F, Deya PM, Elguero J, Estarellas C, Frontera A, Quinonero D (2010) Cooperativity in multiple unusual weak bonds. Theor Chem Acc 126:1–4
Grabowski SJ (2013) Cooperativity of hydrogen and halogen bond interactions. Theor Chem Acc 132:1347
Masoodi HR, Bagheri S, Ranjbar M (2016) Theoretical study of cooperativity between hydrogen bond-hydrogen bond, halogen bond-halogen bond and hydrogen bond-halogen bond in ternary FX… diazine… XF (X= H and Cl) complexes. Mol Phys 114:3464–3474
Bagheri S, Masoodi HR, Yousofvand A (2016) Exploring the role of substituents on cooperativity between N⋯ HF and CH⋯ F hydrogen bonds in ternary systems involving aromatic azine: substituted complexes of s-triazine: HF: s-triazine as a working model. Comput Theor Chem 1092:12–18
Esrafili MD, Ghanbari M, Mohammadian-Sabet F (2014) Substituent effects on cooperativity of pnicogen bonds. J Mol Model 20:1–9
Bauzá A, Quiñonero D, Frontera A, Deyà PM (2011) Substituent effects in halogen bonding complexes between aromatic donors and acceptors: a comprehensive ab initio study. Phys Chem Chem Phys 13:20371–20379
Zhang Q, Smalley A, Zhu Z, Xu Z, Peng C, Chen Z, Yao G, Shi J, Zhu W (2020) Computational study of the substituent effect of halogenated fused-ring heteroaromatics on halogen bonding. J Mol Model 26:1–2
Abadleh M, Peifer C, Kinkel K, Schollmeyer D, Laufer S (2007) 4-[5-(4-Fluorophenyl)-3-isopropylisoxazol-4-yl] pyridin-2 (1H)-one. Acta Crystallogr Sect E Struct Rep Online 63:1423–1425
Voth AR, Hays FA, Ho PS (2007) Directing macromolecular conformation through halogen bonds. Proc Natl Acad Sci USA 104:6188–6193
Varadwaj A, Varadwaj PR, Marques HM, Yamashita K (2022) Definition of the pnictogen bond: a perspective. Inorg 10:149
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V,Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09 (revision A.02). Gaussian, Inc, Wallingford
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, New York
Biegler König B, Schönbohm J (2002) Update of the AIM2000-program for atoms in molecules. J Comput Chem 23:1489–1494
Rozas I, Alkorta I, Elguero J (2000) Behavior of ylides containing N, O, and C atoms as hydrogen bond acceptors. J Am Chem Soc 122:11154–11161
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
Glendening ED, Badenhoop JK, Reed AE, Carpenter JE, Bohmann JA, Morales CM, Weinhold F (2001) Theoretical Chemistry Institute: University of Wisconsin. Madison, WI, 2001NBO
Lu T, Chen F (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33:580–592
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J Mol Graphics 14:33–38
Ditchfield R (1974) Self-consistent perturbation theory of diamagnetism: I. A gauge-invariant LCAO method for NMR chemical shifts. Mol Phys 27:789–807
Adamo C, Barone V (1999) Toward reliable density functional methods without adjustable parameters: the PBE0 model. J Chem Phys 110:6158–6170
Grudova MV, Kubasov AS, Khrustalev VN, Novikov AS, Kritchenkov AS, Nenajdenko VG, Borisov AV, Tskhovrebov AG (2022) Exploring supramolecular assembly space of cationic 1, 2, 4-selenodiazoles: effect of the substituent at the carbon atom and anions. Mol 27:1029
Johnson ER, Keinan S, Mori-Sanchez P, Contreras-Garcia J, Cohen AJ, Yang W (2010) Revealing noncovalent interactions. J Am Chem Soc 132:6498–6506
Isaacs NS (1995) Physical organic chemistry, 2nd edn. Longman, London
Mottishaw JD, Erck AR, Kramer JH, Sun H, Koppang M (2015) Electrostatic potential maps and natural bond orbital analysis: visualization and conceptualization of reactivity in Sanger’s reagent. J Chem Educ 92:1846–1852
Khan I, Panini P, Khan SU, Rana UA, Andleeb H, Chopra D, Hameed S, Simpson J (2016) Exploiting the role of molecular electrostatic potential, deformation density, topology, and energetics in the characterization of S···N and Cl···N supramolecular motifs in crystalline triazolothiadiazoles. Cryst Growth Des 16:1371–1386
Murray JS, Politzer P (2017) Molecular electrostatic potentials and noncovalent interactions. Wiley Interdiscip Rev Comput Mol Sci 7:1326
Politzer P, Truhlar DG (2013) Chemical applications of atomic and molecular electrostatic potentials: reactivity, structure, scattering, and energetics of organic, inorganic, and biological systems. Springer Science & Business Media
Murray JS, Politzer P (2011) The electrostatic potential: an overview. Wiley Interdiscip Rev Comput Mol Sci 1:153–163
Politzer P, Murray JS (2018) σ-holes and π-holes: similarities and differences. J Comput Chem 39:464–471
Veccham SP, Lee J, Mao Y, Horn PR, Head-Gordon M (2021) A non-perturbative pairwise-additive analysis of charge transfer contributions to intermolecular interaction energies. Phys Chem Chem Phys 23:928–943
Golubev NS, Shenderovich IG, Smirnov SN, Denisov GS, Limbach HH (1999) Nuclear scalar spin–spin coupling reveals novel properties of low-barrier hydrogen bonds in a polar environment. Chem Eur J 5:492–497
Pavia DL, Lampman GM, Kriz GS (2007) Introduction to spectroscopy, 3rd edn. Thomson Brooks/Cole Press, Pacific Grove, California
Acknowledgements
The authors thank the Vali-e-Asr University of Rafsanjan for financial supports and Computational Quantum Chemistry Laboratory for computational facilities.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Theoretical calculations, data collection, and analysis were performed by Sotoodeh Bagheri, Mona Vasfi, and Hamid Reza Masoodi. The first draft of the manuscript was written by Sotoodeh Bagheri, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This material (partially or in full) is the authors’ own original work, which has not been previously published elsewhere.
The paper is not currently being considered for publication elsewhere.
The paper reflects the authors’ own research and analysis in a truthful and complete manner.
Consent for publication
All authors approved the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bagheri, S., Vasfi, M. & Masoodi, H.R. Computational analyses of cooperativity between pnicogen and halogen bonds in H2FP:pyrimidine: ClF complex. Struct Chem 35, 569–582 (2024). https://doi.org/10.1007/s11224-023-02212-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02212-1