Abstract
In this study, borate was found to selectively increase the stability of ribose over other aldopentoses. Ribose is the only sugar present in both early RNA-based biochemistry and contemporary DNA-based life, and the stability of ribose is of fundamental concern for determining the origin of early RNA-based biochemistry. The formose reaction is a potential process in the prebiotic synthesis of ribose and its stereoisomers arabinose, xylose, and lyxose. Ribose is the least stable of these aldopentoses, raising the fundamental question of whether it was originally a component of primitive RNA or was selected through biotic processes. Borate is known to increase the stability of aldopentoses, but the specific differences in the stabilization achieved among different stereoisomers remain unclear. In this study, it was found that the stabilities of all of the tested pentoses increased with the concentration of added borate, but notably, the stability of ribose increased the most. The predominant formation of complexes between borate and ribose was verified, in agreement with previous studies. This borate complex formation might have sequestered ribose from the isomerization and decomposition reactions, resulting in its selective stabilization. These findings indicate that ribose could have accumulated in borate-rich environments on the early Earth and suggest that ribose-based nucleotides combined with phosphate and nucleobases formed abiotically.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ribose is a carbohydrate component in ribonucleic acids (RNA), which are essential components for gene expression in all living organisms. The discovery of catalytic RNA, also known as ribozymes, has illustrated the extreme significance of RNA for the origin of life (Benner et al. 1989; Joyce 1989; Bartel and Szostak 1993).
The formose reaction, oligomerization of formaldehyde under alkaline conditions (Breslow 1959; Shapiro 1988; Schwartz and Degraaf 1993; Orgel 2004; Kim et al. 2011), is a potential prebiotic process for ribose synthesis. Ribose can be formed in the formose process, along with many other carbohydrates including its own isomers (i.e., the other aldopentoses arabinose, xylose, and lyxose; Breslow 1959; Shapiro 1988; Schwartz and Degraaf 1993; Orgel 2004; Kim et al. 2011). However, formose chemistry is an intricate process involving the formation of sugars, their interconversion, and also their oligomerization to yield even more complex mixtures (Ricardo et al. 2004). In the progressive oligomerization occurring during the formose process, ribose is the most unstable of the aldopentoses (El Khadem et al. 1987; Larralde et al. 1995). This raises the question of whether ribose could have been a component in primordial genetic molecules, in spite of the broad recognition of the importance of RNA in the origin of life (Larralde et al. 1995; Nielsen 2007).
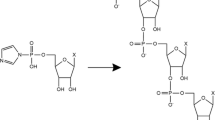
As one possible mechanism leading to the selection of ribose in RNA, the formation of pentose borate complexes has been reported in previous studies (i.e., 2:1 complexes composed of two pentose molecules and one borate and 1:1 complexes composed of one pentose molecule and one borate; Fig. 1; Chapelle and Verchere 1988; Li et al. 2005; Šponer et al. 2008; Pepi et al. 2010). These studies also reported various binding affinities for complexation between borate and the different aldopentoses. Complex formation between pentose and borate stabilizes pentoses (Ricardo et al. 2004; Kim et al. 2011). Although borate has been shown to stabilize aldopentoses, the extent to which the different aldopentoses are stabilized by borate remains unclear because of a lack of a suitable analytical technique.
Boron is cycled on the surface and in shallow areas of the modern Earth. The boron in continental crusts is leached by weathering and then is carried by riverine water in the form of borates to accumulate in oceanic water (Spivack et al. 1987). Clay-rich marine sediments and mafic/ultramafic igneous rocks behave as borate sinks in the ocean (Williams et al. 2001; Boschi et al. 2008). Then, magmatic and/or fluidal processes in the subduction zone liberate boron from the oceanic crust, returning it to the surface environments (Turner et al. 2007). In this modern boron geochemical cycle, continental crusts are important boron reservoirs, but they were most likely less abundant on the early Earth, which raises some ambiguity about whether borates were available in the early Earth’s ocean (Grew et al. 2011).
On the other hand, previous investigators have reported the presence of tourmaline, a borosilicate, in rocks formed ca. 3.8 billion years ago (Gya) in the Isua Supracrustal Belt (Appel 1995; Chaussidon and Uitterdijk Appel 1997; Heijlen et al. 2006). Some tourmaline is present in hydrothermal breccias that were formed by 3.8 Gya submarine hydrothermal alteration (Appel et al. 2001; Heijlen et al. 2006). In addition, tourmaline has also been reported in metasediments in the Isua Supracrustal Belt (Appel 1995; Chaussidon and Uitterdijk Appel 1997). These reports imply that aqueous borate was already available in oceanic water and that certain amounts of borate were sunk into sediments and mafic rocks 3.8 Gya.
In this study, we investigated the effects of borate on the stability of each pentose in alkaline solution. A liquid chromatography/mass spectrometry (LC/MS) technique for quantitative analyses of pentoses in a solution of different isomers was developed here in order to assess the amount of each individual pentose in the mixture (Fig. S1c). In addition, the relative amounts of pentose–borate complexes were assessed in order to elucidate the mechanisms by which borate changes the stabilities of pentoses.
Materials and Methods
Materials
Borax (sodium tetraborate decahydrate; Na2B4O7•10H2O; >99.5 %; Wako Pure Chemical Industries, Ltd.) was used as a borate source. The aldopentoses d-ribose (~99 %; Wako Pure Chemical Industries, Ltd.), d-xylose (99 %; Wako Pure Chemical Industries, Ltd.), d-lyxose (~99 %; Alfa Aesar), and l-arabinose (~99 %; Wako Pure Chemical Industries, Ltd.) were used in this study. For identification of ketopentoses, we used commercial standards for d-ribulose (MP Biomedical, LLC; 99.7 %) and d-xylulose (Sigma-Aldrich; >98 %). Calcium hydroxide (~96 %; MERCK) was used to buffer the pH level. LC/MS-grade acetonitrile (Wako Pure Chemical Industries, Ltd.) and ultrapure water (18.2 MΩ and UV sterilized; prepared with Simplicity UV; Millipore) were used as the eluent for chromatography and solvents for the samples.
Experimental Methods
The stabilities of the aldopentoses were investigated by performing pentose incubation experiments. Borax was dissolved in water to form borate, B(OH)4 −, solutions with either a high concentration (80 mmol/L (mM)) or a low concentration (40 mM). The solutions were then heated to 45 °C. Each aldopentose was added separately to a borate solution sample to a final concentration of 1 mM. Calcium hydroxide was immediately added (21.4 mg/10 mL) to each solution to buffer the pH to 12.0–12.2. A set of samples without borax was prepared for control experiments.
The pentose solutions were incubated at 45 ± 3 °C with continuous stirring. Aliquots of each of the 12 samples were withdrawn at 90 min intervals and immediately analyzed using a high-performance liquid chromatograph (2695 separation module; Waters) connected to a tandem mass spectrometer (Quattro micro API; Waters) (LC/MS). An amino column (TSKgel NH2-100, 2.0 mm I.D. and 150 mm length, 3 μm; TOSOH) was used at 30 °C. A mixture of acetonitrile and ultrapure water (85:15) was adopted as the eluent and a flow rate of 0.5 mL/min was used. Negative-mode electrospray ionization (ESI) was used to ionize the pentoses. The flow rates of the desolvation gas and cone gas were 900 L/h and 90 L/h, respectively. The voltages of the ESI capillary and sampling cone were 2.5 kV and 22 V, respectively. The temperatures of the desolvation gas and source block were 450 °C and 120 °C, respectively. The amounts of the pentoses were measured in single-ion recording mode at m/z = 149.
In addition to the amount of each pentose, the relative amounts of the different pentose–borate complexes in pentose solutions containing borate were measured using a MS technique after 5 min of incubation. Samples were introduced into the mass spectrometer with a syringe at a constant flow rate. During the infusion, the intensities of the peaks in the spectrum that corresponded to free pentoses, 1:1 pentose–borate complexes (Fig. 1), and 2:1 pentose–borate complexes (Fig. 1) were observed at m/z = 149, 193, and 307, respectively. The flow rates of the desolvation gas and cone gas were 900 L/h and 50 L/h, respectively. The voltage of the ESI capillary was 2.5 kV, and the voltage of a sampling cone was 12 V for the 1:1 complex and 30 V for the 2:1 complex. The temperatures of the desolvation gas and source block were 350 °C and 120 °C, respectively.
Results and Discussion
Figure 2 shows the relative intensities of the pentose–borate complex signals measured using a mass spectrometer after 5 min of incubation. These intensities indicate the relative amounts of complexes, under the assumption that the different complexes have identical ionization efficiencies in the mass spectrometer. In the 80 mM borate experimental solutions, both 1:1 and 2:1 pentose–borate complexes were found (Fig. 2b). The intensities of the signals corresponding to 1:1 ribose–borate complexes were far greater than the intensities of any other signals (Fig. 2b and Table S1), which is in agreement with the relative amounts of pentose–borate complexes found in previous mass spectrometry and NMR studies (Chapelle and Verchere 1988; Pepi et al. 2010). For the 40 mM borate experimental solutions, the intensities corresponding to each complex were smaller than those for 80 mM borate solutions (Fig. 2a, b and Table S1). The relative intensities for the different pentoses varied in the order of ribose > xylose > lyxose > arabinose for the 1:1 complexes and xylose and ribose > arabinose > lyxose for the 2:1 complexes (Fig. 2a).
Relative intensities of pentose–borate complex peaks in the mass spectra for a 40 mM borate and b 80 mM borate experimental solutions after 5 min of incubation. Each intensity is normalized to the intensity of the ribose–borate 1:1 complex in the 80 mM borate experiments. The error bars represent ±10 %
Figures 3 and 4 and Table 1 show the changes in the concentrations of the individual pentose molecules during the incubation period. The concentrations decreased significantly within the first 5 min, regardless of the presence or absence of borate. In the experiments without borate, the concentration of ribose was the most extensively reduced among the different pentose molecules during this time, reaching approximately 0.06 mM compared to concentrations of 0.10–0.15 mM for the other pentoses.
Pentose concentrations in all aldopentose incubation experiments: incubation of borate-free solutions containing a ribose, b arabinose, c xylose, and d lyxose; incubation of 40 mM borate solutions containing e ribose, f arabinose, g xylose, and h lyxose; and incubation of 80 mM borate solutions containing i ribose, j arabinose, k xylose, and l lyxose
Curves showing the decreases in concentration for various aldopentoses in a borate-free solution, b 40 mM borate solution, and c 80 mM borate solution. The data were compiled from Fig. 2. The error bars represent ±30 %, and k represents the first-order rate constant for the decrease in ribose concentration after 5 min
After the initial 5 min, the concentration decreased exponentially with incubation time (Fig. 3). The exponential decrease can be characterized by the first-order reaction rate constant k [min−1] in the rate equation −d[P]/dt = k[P], where [P] represents the concentration of a pentose. In the experiments without borate, the ribose concentration decreased the most rapidly, reaching a level after 90 min that is ten times lower than those of the other pentoses (Fig. 4a). The rate constant (k 0) for the decrease in ribose concentration was 0.032 min−1. Previous incubation experiments involving pentoses in alkaline and neutral solutions have also demonstrated selective reduction of the ribose concentration relative to the concentrations of other pentoses (El Khadem et al. 1987; Larralde et al. 1995).
Because no pentose was formed in an amount corresponding to the reduction in the amount of starting molecules during the first 5 min (Fig. 3), it is unlikely that isomerization was a significant factor in the initial concentration decrease. Instead, this decrease was probably due to the formation of complexes between the pentoses and calcium ions in the first 5 min after mixing, after which equilibrium was reached (Lenkinski and Reuben 1976; Symons et al. 1982; Yanagihara et al. 1993). The concentration decrease after 5 min was caused by other factors, including isomerization yielding a ketopentose and a second aldopentose, oligomerization by an aldol reaction forming C–C bonds, fragmentation by a retroaldol reaction that breaks C–C bonds, and dehydration (Speck 1958; El Khadem et al. 1987; Miljković 2009).
In the solutions containing borate, significant reductions in the aldopentose concentrations were also observed within the first 5 min (Fig. 4b, c). The concentration of ribose decreased to the lowest level among all aldopentose concentrations in the 40 mM borate experiments, whereas in the 80 mM borate experiments, the concentration of ribose was higher than that of xylose and lower than that of lyxose (Fig. 4c). Analysis of the complexes showed the formation of pentose–borate complexes within 5 min (Fig. 2), which agrees with a previous study that demonstrated that complexation between pentoses and borate quickly reached equilibrium (Verchere and Hlaibi 1987).
After 5 min, the rate of the decrease in the ribose concentration for the experiment with the 40 mM borate solution was moderate in comparison with that for the experiment with the solution without borate (Figs. 4a, b and S1b) and became almost identical to the rates for the other pentoses (Fig. 4b). The first-order rate constant (k 40) for the decrease in ribose concentration was 0.015 min−1 in the 40 mM borate experiments. In the 80 mM borate experiments, after 5 min, the rates of the decreases in the ribose and xylose concentrations were the smallest among those of all the other aldopentoses, whereas the decrease in lyxose concentration was the fastest (Fig. 4c). The concentration of ribose was therefore the highest after 180 min of incubation (~0.06 mM) and did not change throughout the remainder of the experiment (270 min). The first-order rate constant for the decrease in ribose concentration in the 80 mM solution (k 80) decreased further to 0.002 min−1. In a comparison of all 12 pentose incubation experiments, borate is observed to reduce the rate of the concentration decrease for all aldopentoses after 5 min (Fig. 4). In particular, the concentration decrease rate for ribose was substantially lowered by borate, which indicates that borate is more effective in stabilizing ribose than the other aldopentoses.
Xylulose was formed by incubation of both xylose and lyxose in borate-containing solutions (Fig. 3g, h, k, l). Ribulose was formed by incubation of ribose and also by incubation of arabinose, but only in solutions with higher borate concentrations (Fig. 3i, j). These ketopentoses were not formed in the solutions without borate (Fig. 3a–d). Such formation of ketopentoses indicates that aldose–ketose isomerization is facilitated by the presence of borate. This isomerization had little effect on the stabilization of aldopentoses, as suggested by the larger aldopentose stabilization for the samples yielding more ketopentoses (Fig. 3i–l).
Arabinose and xylose were detected after ribose and lyxose incubation, respectively, in the experiments with both borate-free and 40 mM borate solutions (Fig. 3a, d, e, h). These molecules are attributed to epimerization at the C2 position (Miljković 2009), although they were not observed in the experiments for 80 mM borate solutions (Fig. 3i, l). This indicates that the presence of a high concentration of borate restricts C2 epimerization, which in turn contributes to the stabilization of ribose and lyxose.
It is noteworthy that ribose was the most significantly stabilized aldopentose and had the highest binding affinity with borate (Figs. 2 and 4a, b). Therefore, complex formation with borate may have sequestered ribose from epimerization, oligomerization, fragmentation, and dehydration. Therefore, the improved stability of the ribose complex may have resulted in a smaller decrease in the concentration of uncomplexed ribose (Fig. S2).
Several processes, such as impacts and photochemical reactions, have been proposed as sources of aldehydes on the early Earth (Mukhin et al. 1989; Chyba et al. 1990; Cleaves 2008), which might have initiated formose-like processes. The presence of borosilicate in the Isua Supracrustal Belt suggests that aqueous borate existed in the ocean on the early Earth (Appel 1995; Chaussidon and Uitterdijk Appel 1997; Heijlen et al. 2006). On the other hand, a high concentration of aqueous borate in the open ocean in the prebiotic age might not be plausible (Grew et al. 2011). The oceanic crust likely consisted of mostly mafic to ultramafic rocks well before 3.8 Gyr. Serpentinization of the mafic/ultramafic rocks is known to consume boron and store it in the resulting serpentine (Boschi et al. 2008; Vils et al. 2008). This leads to the possibility that Hadean serpentine could accumulate and store boron even if the borate concentrations in the ocean were low at that time. Hydrothermal circulation in borate-enriched serpentine, such as that in the Lost City hydrothermal system in the modern ocean floor, can create alkaline solutions associated with abundant dissolved borates (Seyfried and Dibble 1980; Kelley et al. 2005; Holm et al. 2006). Therefore, geological settings such as those containing low-temperature hydrothermal fluids could have created borate-rich alkaline environments on the early Earth.
The results of the present study indicate that ribose could have been selectively stabilized and accumulated in borate-rich environments on the prebiotic Earth, facilitating the formation of ribose-based RNA. These results further imply that the preference for ribose in RNA began in the prebiotic stage rather than in later biotic processes.
References
Appel PWU (1995) Tourmalinites in the 3800-Ma-old Isua supracrustal belt, West Greenland. Precambrian Res 72:227–234
Appel PWU, Rollinson HR, Touret JLR (2001) Remnants of an Early Archaean (>3.75 Ga) sea-floor, hydrothermal system in the Isua Greenstone Belt. Precambrian Res 112:27–49
Bartel DP, Szostak JW (1993) Isolation of new ribozymes from a large pool of random sequences. Science 261:1411–1418
Benner SA, Ellington AD, Tauer A (1989) Modern metabolism as a palimpsest of the RNA world. Proc Natl Acad Sci U S A 86:7054–7058
Boschi C, Dini A, Früh-Green GL, Kelley DS (2008) Isotopic and element exchange during serpentinization and metasomatism at the Atlantis Massif (MAR 30°N): insights from B and Sr isotope data. Geochim Cosmochim Acta 72:1801–1823
Breslow R (1959) On the mechanism of the formose reaction. Tetrahedron Lett 1:22–26
Chapelle S, Verchere JF (1988) A 11B and 13C NMR determination of the structures of borate complexes of pentoses and related sugars. Tetrahedron 44:4469–4482
Chaussidon M, Uitterdijk Appel PW (1997) Boron isotopic composition of tourmalines from the 3.8-Ga-old Isua supracrustals, West Greenland: implications on the δ11B value of early Archean seawater. Chem Geol 136:171–180
Chyba CF, Thomas PJ, Brookshaw L, Sagan C (1990) Cometary delivery of organic-molecules to the early Earth. Science 249:366–373
Cleaves HJ (2008) The prebiotic geochemistry of formaldehyde. Precambrian Res 164:111–118
El Khadem HS, Ennifar S, Isbell HS (1987) Contribution of the reaction pathways involved in the isomerization of monosaccharides by alkali. Carbohydr Res 169:13–21
Grew ES, Bada JL, Hazen RM (2011) Borate minerals and origin of the RNA world. Orig Life Evol Biosph 41:307–316
Heijlen W, Appel PWU, Frezzotti ML, Horsewell A, Touret JLR (2006) Metamorphic fluid flow in the northeastern part of the 3.8–3.7 Ga Isua Greenstone Belt (SW Greenland): a re-evalution of fluid inclusion evidence for early Archean seafloor-hydrothermal systems. Geochim Cosmochim Acta 70:3075–3095
Holm NG, Dumont M, Ivarsson M, Konn C (2006) Alkaline fluid circulation in ultramafic rocks and formation of nucleotide constituents: a hypothesis. Geochem Trans 7:7
Joyce GF (1989) RNA evolution and the origins of life. Nature 338:217–224
Kelley DS et al (2005) A serpentinite-hosted ecosystem: the lost city hydrothermal field. Science 307:1428–1434
Kim H et al (2011) Synthesis of carbohydrates in mineral-guided prebiotic cycles. J Am Chem Soc 133:9457–9468
Larralde R, Robertson MP, Miller SL (1995) Rates of decomposition of ribose and other sugars: implications for chemical evolution. Proc Natl Acad Sci U S A 92:8158–8160
Lenkinski RE, Reuben J (1976) Studies of the binding of calcium and lanthanum ions to d-lyxose and d-ribose in aqueous solutions using proton magnetic resonance. J Am Chem Soc 98:3089–3094
Li Q, Ricardo A, Benner SA, Winefordner JD, Powell DH (2005) Desorption/ionization on porous silicon mass spectrometry studies on pentose–borate complexes. Anal Chem 77:4503–4508
Miljković M (2009) Carbohydrates: synthesis, mechanisms, and stereoelectronic effects. Springer, New York
Mukhin LM, Gerasimov MV, Safonova EN (1989) Origin of precursors of organic molecules during evaporation of meteorites and mafic terrestrial rocks. Nature 340:46–48
Nielsen PE (2007) Peptide nucleic acids and the origin of life. Chem Biodivers 4:1996–2002
Orgel LE (2004) Prebiotic chemistry and the origin of the RNA world. Crit Rev Biochem Mol Biol 39:99–123
Pepi F, Garzoli S, Tata A, Giacomello P (2010) Low-energy collisionally activated dissociation of pentose–borate complexes. Int J Mass Spectrom 289:76–83
Ricardo A, Carrigan MA, Olcott AN, Benner SA (2004) Borate minerals stabilize ribose. Science 303:196
Schwartz AW, Degraaf RM (1993) The prebiotic synthesis of carbohydrates: a reassessment. J Mol Evol 36:101–106
Seyfried WE Jr, Dibble WE Jr (1980) Seawater–peridotite interaction at 300 °C and 500 bars: implications for the origin of oceanic serpentinites. Geochim Cosmochim Acta 44:309–321
Shapiro R (1988) Prebiotic ribose synthesis: a critical analysis. Orig Life Evol Biosph 18:71–85
Speck JC (1958) The Lobry de Bruyn-Alberda van Ekenstein transformation. Adv Carbohydr Chem 13:63–103
Spivack AJ, Palmer MR, Edmond JM (1987) The sedimentary cycle of the boron isotopes. Geochim Cosmochim Acta 51:1939–1949
Šponer JE, Sumpter BG, Leszczynski J, Šponer J, Fuentes-Cabrera M (2008) Theoretical study on the factors controlling the stability of the borate complexes of ribose, arabinose, lyxose, and xylose. Chem Eur J 14:9990–9998
Symons MCR, Benbow JA, Pelmore H (1982) Study of calcium ion binding to d-ribose in aqueous solutions using hydroxy-proton resonance shifts. J Chem Soc Faraday Trans 1 78:3671–3677
Turner S, Tonarini S, Bindeman I, Leeman WP, Schaefer BF (2007) Boron and oxygen isotope evidence for recycling of subducted components over the past 2.5 Gyr. Nature 447:702–705
Verchere JF, Hlaibi M (1987) Stability constants of borate complexes of oligosaccharides. Polyhedron 6:1415–1420
Vils F, Pelletier L, Kalt A, Müntener O, Ludwig T (2008) The lithium, boron and beryllium content of serpentinized peridotites from ODP Leg 209 (Sites 1272A and 1274A): implications for lithium and boron budgets of oceanic lithosphere. Geochim Cosmochim Acta 72:5475–5504
Williams LB, Hervig RL, Holloway JR, Hutcheon I (2001) Boron isotope geochemistry during diagenesis. Part I. Experimental determination of fractionation during illitization of smectite. Geochim Cosmochim Acta 65:1769–1782
Yanagihara R, Soeda K, Shiina S, Osanai S, Yoshikawa S (1993) C-2 epimerization of aldoses by calcium ion in basic solutions: a simple system to transform d-glucose and d-xylose into d-mannose and d-lyxose. Bull Chem Soc Jpn 66:2268–2272
Acknowledgments
We appreciate the comments by S. Benner and anonymous reviewers. This work was supported by the Global Education and Research Center for Earth and Planetary Dynamics of Tohoku University and by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science to T.K. (22654063 and 24244084) and Y.F. (23740402).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 444 kb)
Rights and permissions
About this article
Cite this article
Furukawa, Y., Horiuchi, M. & Kakegawa, T. Selective Stabilization of Ribose by Borate. Orig Life Evol Biosph 43, 353–361 (2013). https://doi.org/10.1007/s11084-013-9350-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11084-013-9350-5