Abstract
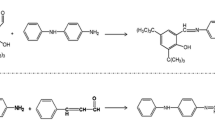
Novel antioxidants based on polymerized 2,2,4-trimethyl-1,2-dihydroquinoline (TMQ) are synthesized. TMQ is chemically modified to obtain new derivatives, namely; Ester, Hydarzide, Oxadiazole and Triazole. The synthesized polymers are characterized by Fourier transforms infrared spectroscopy (FTIR) and proton-nuclear magnetic resonance (1HNMR). The formed polymers are evaluated as antioxidants for styrene-butadiene rubber (SBR) composites. TMQ and the prepared polymers are added to SBR by a concentration of 1 phr and their antioxidant activity is compared. The rheometric characteristics and the mechanical properties including tensile strength, elongation at break, modulus at 100% elongation and hardness are studied. The results show that the prepared polymer vulcanizates are characterized by high mechanical properties compared to the commercial TMQ. The tensile strength equals 14.92 MPa, 18.3 MPa, 20.43 MPa, 21.59 MPa, 19.5 MPa and 18.92 MPa for blank, TMQ, Ester, Hydrazide, Oxadiazole and triazole vulcanizates, respectively. The prepared vulcanizates were subjected to thermo-oxidative aging for two, four, six and seven days. Mechanical properties are measured after aging and their retained values are estimated. The results show that the prepared compounds have high antioxidant activity compared to commercial TMQ. The retained values of tensile strength after aging for seven days are 50.27%, 71.97%, 80.90%, 86.66%, 80.15%, 72.99%, for blank, TMQ, Ester, Hydrazide, Oxadiazole and triazole vulcanizates, respectively. The hydrazide polymer exhibits the highest antioxidant efficiency among the prepared polymers. It has retained tensile strength of 86.66% after aging for seven days. It is also found that the rubber vulcanizates containing the synthesized polymers have the lowest swelling values and consequently higher crosslink density leading to high mechanical properties and high antioxidant efficiency.














Similar content being viewed by others
Data availability
The data included in this paper are available upon request.
References
Abdelsalam AA, Araby Sh, El-Sabbagh SH, Abdelmoneim A, Hassan MA (2021) A comparative study on mechanical and rheological properties of ternary rubber blends. Polym Polym Compos 29(1):15–28
Shah R, Pratt DA (2016) Determination of key hydrocarbon autoxidation products by fluorescence. J Org Chem 81(15):6649–6656
Cibulková Z, Šimon P, Lehocký P, Kosár K, Chochulová A (2009) DSC study of antioxidant activity of selected p-phenylenediamines in styrene-butadiene rubber. J Therm anal Calorm 97(2):535–540
Zhao W, Dufresne A, Li A, An H, Shen C, Yu P, Jiang X, Wang R, Zhang L (2023) Use of lignin-based crude carbon dots as effective antioxidant for natural rubber. Int J Biol Macromol 253:126594
Zou Y, He J, Tang Z, Zhu L, Liu F (2020) Structural and mechanical properties of styrene–butadiene rubber/silica composites with an interface modified in-situ using a novel hindered phenol antioxidant and its samarium complex. Compos Sci Technol 188:107984
Wu W, Zeng X, Li H, Lai X, **e H (2015) Synthesis and antioxidative properties in natural rubber of novel macromolecular hindered phenol antioxidants containing thioether and urethane groups. Polym Degrad Stab 111:232–238
Sun Y, Huang J, He J, Tang Z, Zhu L, Liu F (2021) Effects of a novel hindered phenol samarium complex on the thermo-oxidative aging of styrene-butadiene rubber/silica composites. Polym Degrad Stab 185:109482
Klinpituksa P, Kiarttisarekul A, Kaesaman A (2014) Rubber bound phenolic antioxidant and its application in thermoplastic elastomer. Adv Mater Res 3:175–183
Abd El-Ghaffar MA, Shaffei KA, Abdelwahab N (2014) Evaluation of some conducting polymers as novel antioxidants for rubber vulcanizates. Int J Polym Sci Article ID 893542. https://doi.org/10.1155/2014/893542
Al-Malaika S, Ibrahim AQ, Rao MJ, Scott G (1992) Mechanisms of antioxidant action: photoantioxidant activity of polymer-bound hindered amines. II. Bis acrylates. J Appl Polym Sci 44:1287–1296
Herdan JM, Giurginca M (1993) Grafting antioxidants. V phenols with mercaptoheterocyclic substituents as antioxidants for dienic rubbers. Polym Degrad Stab 41:157–162
Giurginca M, Herdan JM, Cira L, Valeanu G, Ivan G (1992) Grafted mercapto-1,3,5-triazinic antioxidants for elastomers. Polym Degrad Stab 36:53–57
Illarionova MS, Ushmarin NF, Kapitonova MA, Koltsov NI (2017) The influence of antioxidants on the properties of rubber based on butadiene-nitrile caoutchouc. Butlerov Commun 51:126–131
Zhang J, Zhang H, Chen F, Liu M (2019) Improving stability of mechanical properties for nitrile butadiene rubber composite by carbon nanotube with antioxidant loading distribution. Polym compos, 2019, doi: 10.1002/pc.24929.
Flory P (1946) Effects of molecular structure on physical properties of butyl rubber. J Ind Eng Chem 38:417–436
Zidan TA, El-Sabbagh SH, Yehia AA (2022) Synthesis and evaluation of methyl 3, 4, 5-trihydroxybenzoate and methyl 3, 4, 5-trihydroxybenzohydrazide as additives for natural rubber composites. Egypt J Chem 65(2):531–538
Zidan TA (2024) Rhodanine-chitosan hydrogel as a novel antioxidant for acrylonitrile-butadiene rubber composites. J Vinyl Addit Technol 30(1):349–358
Zidan TA, Kandil HS (2024) Investigation of novel polyanilines as new antioxidants for EPDM rubber composites. J Appl Polym Sci 141:e54924
Fadda AA, Abdel-Rahman AA-H, El-Sayed WA, Zidan TA, Badria FA (2011) Synthesis of novel 1,3,4-oxadiazole derivatives and their nucleoside analogs with antioxidant, and antitumor activities. Chem Hetero Comp 7:1045–1054
Rana SM, Islam M, Saeed H, Rafique H, Majid M, Aqeel MT, Imtiaz F, Ashraf Z (2023) Synthesis, computational studies, antioxidant and anti-inflammatory bio-evaluation of 2,5-disubstituted1,3,4-oxadiazole derivatives. Pharmaceut 16:1045–1064
Bhandari Sh V, Nagras OG, Kuthe PV, Sarkate AP, Waghamare KS, Pansare DN, Chaudhari SY, Mawale Sh N, Belwate MC (2023) Design, synthesis, molecular docking and antioxidant evaluation of Benzimidazole-1,3,4 oxadiazole derivatives. J Mol Struct 1276:134747
Dawbaa S, Nuha D, Evren AE, Cankiliç MY, Yurttaş L, Turan G (2023) New oxadiazole/triazole derivatives with antimicrobial and antioxidant properties. J Mol Struct 1282:135213
Tay NF, Duran M, Kayagil İ, Yurttaş L, Göger G, Göger F, Demirci F, Demirayak Ş (2022) Synthesis, antimicrobial and antioxidant activities of pyridyl substituted thiazolyl triazole derivatives. Braz J Pharm Sci 58:e191026–e191054
Aggarwal R, Hooda M, Kumar P, Sumran G (2022) Vision on synthetic and medicinal facets of 1,2,4–triazolo[3,4–b][1,3,4]thiadiazine scafold. Top Curr Chem 380:10–75
Verbanac D, Malik R, Chand M, Kushwaha Kh, Vashist M, MatijaŠić M, Stepanić V, Perić M, Paljetak HČ, Saso L, Jain SC (2016) Synthesis and evaluation of antibacterial and antioxidant activity ofnovel 2-phenyl-quinoline analogs derivatized at position 4 with aromatically substituted 4H-1,2,4-triazoles. J Enzyme Inhib Med Chem 31(S2):104–110
Pachuta-Stec A (2022) Antioxidant activity of 1,2,4-Triazole and its derivatives: a Mini-review. Mini-Rev Med Chem 22(7):1081–1094
Kandil H, Ward AA, Zidan TA (2023) Amino-terminated hyperbranched poly(phenylene oxide) as a novel antioxidant for nitrile-butadiene rubber. Polym Bull. https://doi.org/10.1007/s00289-023-05054-3
Koopaei MN, Assarzadeh MJ, Almasirad A, Ghasemi-Niri SF, Amini M, Kebriaeezadeh A, Koopaei NN, Ghadimi M, Tabei A (2013) Synthesis and analgesic activity of Novel Hydrazide and Hydrazine derivatives. Iran J Pharm Res 12:721–727
Abdelrehim EM (2021) Synthesis and screening of New [1,3,4]Oxadiazole, [1,2,4]Triazole, and [1,2,4]Triazolo[4,3-b][1,2,4]triazole derivatives as potential Antitumor agents on the Colon carcinoma cell line (HCT-116). ACS Omega 6:1687–1696
Abacl G, Rollas S, Tatar E (2013) Development of a new and efficient synthesis method of 1,2,4-Triazole-5-Thione derivatives. Marmara Pharm J 17:181–186
Zhao S, Li J, Yan Z, Lu T, Liu R, Han X, Cai C, Sh Z, Wang H (2021) Preparation of lignin-based filling antioxidant and its application in styrene-butadiene rubber. J Appl Polym Sci. https://doi.org/10.1002/app.51281
Manaila E, Stelescu MD, Craciun G (2018) Degradation studies realized on natural rubber and plasticized potato starch based eco-composites obtained by peroxide cross-linking. J Mol Sci 19:2862–2880
Abdel-Hakim A, El-Mogy SA, Abou-Kandil A (2021) Novel modification of styrene butadiene rubber/acrylic rubber blends to improve mechanical, dynamic mechanical, and swelling behavior for oil sealing applications. Polym Polym Compos 29(9S):S959–S968
Manaila E, Stelescu MD, Craciun G, Ighigeanu D (2016) Wood sawdust/natural rubber ecocomposites cross-linked by electron beam irradiation. Mater 9:503–526
Nawwar G, Yakout S, El-Sadiek MSA, El-Sabbagh S (2011) Synthesis and evaluation of new antioxidants for styrene butadiene rubber. Pigm Resin Technol 40(6):399–409
Abdelsalam AA, Ward AA, Abdel-Naeem G, Mohamed WS, El–Sabbagh SH (2023) Effect of alumina modified by silane on the mechanical, swelling and dielectric properties of Al2O3/EPDM/SBR blend composites. Silicon 15:3609–3621
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is not any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zidan, T.A., Yehia, A.A. Novel antioxidants based on polymerized 2,2,4-trimethyl-1,2-dihydroquinoline for styrene-butadiene rubber composites. J Polym Res 31, 170 (2024). https://doi.org/10.1007/s10965-024-04006-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-024-04006-3