Abstract
The perplexing issues related to positive surgical margins and the considerable negative consequences associated with systemic chemotherapy have posed ongoing challenges for clinicians, especially when it comes to addressing bladder cancer treatment. The current investigation describes the production of nanocomposites loaded with gemcitabine (GEM) and cisplatin (CDDP) through the utilization of electrospinning technology. In vitro and in vivo studies have provided evidence of the strong effectiveness in suppressing tumor advancement while simultaneously reducing the accumulation of chemotherapy drugs within liver and kidney tissues. Mechanically, the GEM and CDDP-loaded electrospun nanocomposites could effectively eliminate myeloid-derived suppressor cells (MDSCs) in tumor tissues, and recruit CD8+ T cells and NKp46+ NK cells to kill tumor cells, which can also effectively inhibit tumor microvascular formation. Our investigation into the impact of localized administration of chemotherapy through GEM and CDDP-loaded electrospun nanocomposites on the tumor microenvironment will offer novel insights for tackling tumors.
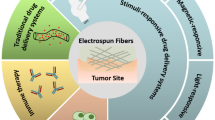
Graphical abstract

Drug-loaded electrospun nanofibrous mats suppress bladder tumor growth by improving the tumor immune microenvironment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The incidence of tumors in various systems and organs has exhibited a substantial increase in recent years [1]. Recurrent growths in the form of distinct masses are commonly observed in solid tumors, and surgical intervention continues to be the foremost treatment approach employed in clinical settings [2,3,4]. Moreover, the challenges associated with positive surgical margins and postoperative tumor recurrence have long perplexed clinicians, particularly in the context of bladder cancer treatment [5, 6]. Bladder cancer is a prevalent malignancy in the genitourinary system, with non-muscle invasive bladder cancer accounting for 75–85% of all cases [7]. Transurethral resection of bladder tumor (TURBT) represents the cornerstone in the management of these patients. However, the recurrence rate after TURBT is significantly high, varying between 40 and 60% [8]. Hence, it is frequently necessary to administer regular intravesical chemotherapy in order to minimize the occurrence of tumor relapse following TURBT.
However, traditional intravesical chemotherapy is frequently administered with a short infusion time and low timeliness, which may predispose to urinary tract infection [9]. The development of a slow-release system for anti-tumor drugs introduces novel perspectives on postoperative chemotherapy, offering potential solutions to the challenges encountered in tumor chemotherapy after surgery [10, 11]. In the past decade, drug-controlled delivery systems have garnered significant attention due to their numerous advantages over conventional dosage forms, including enhanced drug delivery efficiency and reduced drug-related side effects [26], however, the majority of scholars substantiate the efficacy of novel anti-tumor materials through in vivo and in vitro experiments, while neglecting to delve into the underlying molecular mechanisms [27, 28].
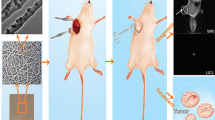
Why does the drug sustained-release material have a more significant anti-tumor effect than intraperitoneal chemotherapy? On one hand, it is hypothesized that this phenomenon may be attributed to the distribution and metabolism of drugs [29]; on the other hand, it could potentially be ascribed to the substantial impact of slow-release materials on drug delivery within the tumor microenvironment [26]. Previous studies have demonstrated that local administration of sustained-release materials exhibits superior hepatoprotective, nephroprotective, and hematopoietic-preserving effects compared to intravenous administration [30, 31]. The local drug concentration within the tumor and the cumulative drug concentration in normal tissues (liver, kidney, and blood) were quantified in this study. The findings substantiate that localized administration of slow-release materials can significantly enhance drug delivery efficacy while mitigating chemotherapy drug accumulation in normal tissue. The findings of our study make a significant contribution to the existing body of research.
Through the immunofluorescence histochemistry, we observed that the distribution of MDSCs in the PLA-drug group was significantly reduced in the tumor tissues. At the same time, as the effector cells of MDSCs, CD8+ T cells and NKp46+ NK cells were significantly increased in the PLA-drug group. The utilization of T cells is rapidly gaining prominence as a promising therapeutic approach for the treatment of cancer and various other diseases. Alex et al. [32] proposed a materials-based strategy to enhance the activation and expansion of T cells, employing electrospinning techniques to fabricate a poly(ε-caprolactone) fiber mesh that serves as a platform for presenting activating ligands targeting CD3 and CD28 receptors, thereby promoting T cell activation and subsequent expansion. Our results suggested that MDSCs were effectively cleared from the tumor tissue due to the improvement of local chemotherapy delivery efficiency of the sustained-release materials, thereby restoring the normal recruitment and differentiation ability of CD8+ T cells and NKp46+ NK cells in the tumor tissue, enhancing the anti-tumor effect of the autoimmune system. Moreover, based on the study of angiogenesis, slow-release materials can also effectively inhibit tumor microvascular formation within the tissue, thereby further restricting tumor growth [33, 34].
Although our experiments have confirmed the chemotherapeutic effect of locally delivered nano-sustained-release materials and their impact on the immune microenvironment, there are still certain limitations in our study. Firstly, there is a lack of comprehensive research on the in vivo decomposition and release process of nano-sustained-release materials. Despite conducting relevant in vitro simulation experiments, they may not fully represent real-life scenarios. Secondly, due to the limited number of experimental animals and the relatively short duration of observation, it remains unclear whether nano-sustained-release materials effectively mitigate systemic chemotherapy side effects. Lastly, while we observed changes in the tumor microenvironment, further investigation is required to elucidate the role of immune cells within this context and explore their interrelationships.
Despite the aforementioned limitations, we have preliminarily demonstrated the anti-tumor efficacy of the GEM and CDDP-loaded mats, thereby establishing a robust foundation for sustained drug delivery to address positive surgical margins and mitigate systemic chemotherapy-induced side effects. Furthermore, exploring the impact of localized chemotherapy delivery using sustained-release materials on the tumor microenvironment will provide novel insights into anti-tumor strategies.
5 Conclusions
In summary, we successfully fabricated gemcitabine-cisplatin-loaded nanocomposites from PLA using classical electrospinning technology. By UPLC-MS/MS, we confirmed that the electrospun nanocomposites could significantly improve the drug delivery efficiency and reduce the accumulation of chemotherapeutic drugs in the liver, kidney, and other organs. A mouse bladder cancer model was used to verify the significant anti-tumor effect in vivo and in vitro. Local delivery of chemotherapeutic drugs via slow-release materials can significantly inhibit bone marrow-derived cells and tumor microvascular formation while refinancing CD8+ T cells, and NKp46+ NK cells into the tumor tissue.
Data availability
All data generated or analyzed during this study are included in this article and its Supplementary Material files. Further inquiries can be directed to the corresponding author (YPZ, zhuyunpeng2016@163.com).
References
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48.
Younan G. Pancreas solid tumors. Surg Clin North Am. 2020;100:565–80.
El LM, Gianakou A, Nockel P, Wiseman D, Tirosh A, Quezado MA, et al. Radioguided surgery with gallium 68 dotatate for patients with neuroendocrine tumors. JAMA Surg. 2019;154:40–5.
Poveda A, Garcia DMX, Lopez-Guerrero JA, Cubedo R, Martinez V, Romero I, et al. GEIS guidelines for gastrointestinal sarcomas (GIST). Cancer Treat Rev. 2017;55:107–19.
Laukhtina E, Rajwa P, Mori K, Moschini M, D’Andrea D, Abufaraj M, et al. Accuracy of frozen section analysis of urethral and ureteral margins during radical cystectomy for bladder cancer: a systematic review and diagnostic meta-analysis. Eur Urol Focus. 2022;8:752–60.
Pak S, Park S, Kim M, Go H, Cho YM, Ahn H. The impact on oncological outcomes after radical prostatectomy for prostate cancer of converting soft tissue margins at the apex and bladder neck from tumour-positive to -negative. BJU Int. 2019;123:811–7.
Jubber I, Ong S, Bukavina L, Black PC, Comperat E, Kamat AM, et al. Epidemiology of bladder cancer in 2023: a systematic review of risk factors. Eur Urol. 2023;84:176–90.
Dyrskjot L, Hansel DE, Efstathiou JA, Knowles MA, Galsky MD, Teoh J, et al. Bladder cancer. Nat Rev Dis Prim. 2023;9:58.
Joice GA, Bivalacqua TJ, Kates M. Optimizing pharmacokinetics of intravesical chemotherapy for bladder cancer. Nat Rev Urol. 2019;16:599–612.
Pei B, Hu M, Wu X, Lu D, Zhang S, Zhang L, et al. Investigations into the effects of scaffold microstructure on slow-release system with bioactive factors for bone repair. Front Bioeng Biotechnol. 2023;11:1230682.
Khan H, Mirzaei HR, Amiri A, Kupeli AE, Ashhad HS, Mirzaei H. Glyco-nanoparticles: new drug delivery systems in cancer therapy. Semin Cancer Biol. 2021;69:24–42.
Xue J, Wu T, Dai Y, **a Y. Electrospinning and electrospun nanofibers: methods, materials, and applications. Chem Rev. 2019;119:5298–415.
Farazin A, Torkpour Z, Dehghani S, Mohammadi R, Fahmy MD, Saber-Samandari S, et al. A review on polymeric wound dress for the treatment of burns and diabetic wounds. Int J Basic Sci Med. 2021;6:44–50.
Karamian E, Khandan A, Motamedi MR, Mirmohammadi H. Surface characteristics and bioactivity of a novel natural HA/zircon nanocomposite coated on dental implants. Biomed Res Int. 2014;2014:410627.
Karamian E, Motamedi MRK, Khandan A, Soltani P, Maghsoudi S. An in vitro evaluation of novel NHA/zircon plasma coating on 316L stainless steel dental implant. Prog Nat Sci Mater. 2014;24:150–6.
Tan H, Zhang Y, Sun L, Sun Y, Dang H, Yang Y, et al. Preparation of nano sustained-release fertilizer using natural degradable polymer polylactic acid by coaxial electrospinning. Int J Biol Macromol. 2021;193:903–14.
Salami MA, Kaveian F, Rafienia M, Saber-Samandari S, Khandan A, Naeimi M. Electrospun polycaprolactone/lignin-based nanocomposite as a novel tissue scaffold for biomedical applications. J Med Signals Sens. 2017;7:228–38.
Heydary HA, Karamian E, Poorazizi E, Heydaripour J, Khandan A. Electrospun of polymer/bioceramic nanocomposite as a new soft tissue for biomedical applications. J Asian Ceram Soc. 2015;3:417–25.
**ao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer. Pharm Ther. 2021;221:107753.
Wu Y, Yi M, Niu M, Mei Q, Wu K. Myeloid-derived suppressor cells: an emerging target for anticancer immunotherapy. Mol Cancer. 2022;21:184.
Gabrilovich DI. Myeloid-derived suppressor cells. Cancer Immunol Res. 2017;5:3–8.
Li G, Zhao M, Zhao L. Ultra-performance liquid chromatography-tandem mass spectrometry for simultaneous determination of 12 anti-tumor drugs in human plasma and its application in therapeutic drug monitoring. J Pharm Biomed Anal. 2021;206:114380.
Wopat H, Harrod T, Brem RF, Kaltman R, Anderson K, Robien K. Body composition and chemotherapy toxicity among women treated for breast cancer: a systematic review. J Cancer Surviv. 2023. Epub ahead of print. https://doi.org/10.1007/s11764-023-01380-7.
Pan Q, Fan X, **e L, Wu D, Liu R, Gao W, et al. Nano-enabled colorectal cancer therapy. J Control Release. 2023;362:548–64.
Li L, Li F, Zhao Z, **e R, Xu D, Ding M, et al. An exploratory research on antitumor effect of drug-eluting slow-releasing electrospinning membranes. Heliyon. 2023;9:e20295.
Luo Z, He Y, Li M, Ge Y, Huang Y, Liu X, et al. Tumor microenvironment-inspired glutathione-responsive three-dimensional fibrous network for efficient trap** and gentle release of circulating tumor cells. ACS Appl Mater Interfaces. 2023;15:24013–22.
Erickson A, Chiarelli PA, Huang J, Levengood SL, Zhang M. Electrospun nanofibers for 3-D cancer models, diagnostics, and therapy. Nanoscale Horiz. 2022;7:1279–98.
Mane PP, Ambekar RS, Kandasubramanian B. Electrospun nanofiber-based cancer sensors: a review. Int J Pharm. 2020;583:119364.
Li J, Liu Y, Abdelhakim HE. Drug delivery applications of coaxial electrospun nanofibres in cancer therapy. Molecules. 2022;27:1803. https://doi.org/10.3390/molecules27061803.
Wang J, Wu M, Zhu Y, Wang Z, Cao H, Li X, et al. A multilayer nanofibrous mat for the topical chemotherapy of the positive margin in bladder cancer. Tissue Eng Part A. 2022;28:958–67.
Zhu Y, Liu S, Feng C, Liu C, Wang Z, Yu K, et al. The delivery materials with chemotherapy drugs for treatment of the positive margin in solid tumors. Tissue Eng Part A. 2021;27:536–48.
Dang A, De Leo S, Bogdanowicz DR, Yuan DJ, Fernandes SM, Brown JR, et al. Enhanced activation and expansion of T cells using mechanically soft elastomer fibers. Adv Biosyst. 2018;2:1700167.
He X, Zhou M, Chen X, Wang J, Zhao X, Zhu Y, et al. Development and characterization of multifunctional wound dressing with the property of anti-bacteria and angiogenesis. Probiotics Antimicrob Proteins. 2023;15:941–54.
Oh YS, Choi MH, Shin JI, Maza P, Kwak JY. Co-culturing of endothelial and cancer cells in a nanofibrous scaffold-based two-layer system. Int J Mol Sci. 2020;21:4128.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (NSFC #82070715).
Author information
Authors and Affiliations
Contributions
Methodology, investigation, formal analysis, writing—original draft, and writing—review, and editing by JW. Conceptualization, investigation, formal analysis, and writing—original draft by YPZ. Methodology, investigation, and writing—original draft by YSY. Formal analysis and writing—review and editing by XR. Supervision, project administration, and writing—review and editing by YPZ and SGW.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All procedures were approved by the Ethics Committee of Tongji Hospital and complied with the Guide for the Care and Use of Laboratory Animals (No. TJ-C20200155).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Yin, Y., Ren, X. et al. Electrospun nanofibrous mats loaded with gemcitabine and cisplatin suppress bladder tumor growth by improving the tumor immune microenvironment. J Mater Sci: Mater Med 35, 21 (2024). https://doi.org/10.1007/s10856-024-06786-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-024-06786-y