Abstract
Background
Furosemide stress test (FST) is a novel functional biomarker for predicting severe acute kidney injury (AKI); however, pediatric studies are limited.
Methods
Children 3 months to 18 years of age admitted to the intensive care unit (ICU) of a tertiary care hospital from Nov 2019 to July 2021 were screened and those who developed AKI stage 1 or 2 within 7 days of admission underwent FST (intravenous furosemide 1 mg/kg). Urine output was measured hourly for the next 6 h; a value > 2 ml/kg within the first 2 h was deemed furosemide responsive. Other biomarkers like plasma neutrophil gelatinase-associated lipocalin (NGAL) and proenkephalin (PENK) were also evaluated.
Results
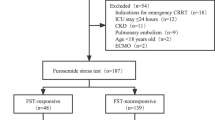
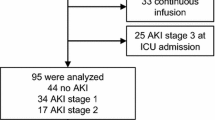
Of the 480 admitted patients, 51 developed AKI stage 1 or 2 within 7 days of admission and underwent FST. Nine of these patients were furosemide non-responsive. Thirteen (25.5%) patients (eight of nine from FST non-responsive group) developed stage 3 AKI within 7 days of FST, nine (17.6%) of whom (seven from non-responsive group) required kidney support therapy (KST). FST emerged as a good biomarker for predicting stage 3 AKI and need for KST with area-under-the-curve (AUC) being 0.93 ± 0.05 (95% CI 0.84–1.0) and 0.96 ± 0.03 (95% CI 0.9–1.0), respectively. FST outperformed NGAL and PENK in predicting AKI stage 3 and KST; however, the combination did not improve the diagnostic accuracy.
Conclusions
Furosemide stress test is a simple, inexpensive, and robust biomarker for predicting stage 3 AKI and KST need in critically ill children. Further research is required to identify the best FST cut-off in children.
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable. Data subsets are available with the corresponding author.
References
Kaddourah A, Basu RK, Bagshaw SM, Goldstein SL, AWARE Investigators (2017) Epidemiology of acute kidney injury in critically ill children and young adults. N Engl J Med 376:11–20
Sutherland SM, Ji J, Sheikhi FH, Widen E, Tian L, Alexander SR, Ling XB (2013) AKI in hospitalized children. Clin J Am Soc Nephrol 8:1661–1669
Sanchez-Pinto LN, Goldstein SL, Schneider JB, Khemani RG (2015) Association between progression and improvement of acute kidney injury and mortality in critically ill children. Pediatr Crit Care Med 16:703–710
Alkandari O, Eddington KA, Hyder A, Gauvin F, Ducruet T, Gottesman R, Phan V, Zappitelli M (2011) Acute kidney injury is an independent risk factor for pediatric intensive care unit mortality, longer length of stay and prolonged mechanical ventilation in critically ill children: a two-center retrospective cohort study. Crit Care 15:R146
Greenberg JH, Coca S, Parikh CR (2014) Long-term risk of chronic kidney disease and mortality in children after acute kidney injury: a systematic review. BMC Nephrol 15:184
Greenberg JH, Zappitelli M, Devarajan P, Thiessen-Philbrook HR, Krawczeski C, Li S, Garg AX, Coca S, Parikh CR, TRIBE-AKI Consortium (2016) Kidney outcomes 5 years after pediatric cardiac surgery: the TRIBE-AKI study. JAMA Pediatr 170:1071–1078
Hessey E, Perreault S, Dorais M, Roy L, Zappitelli M (2019) Acute kidney injury in critically ill children and subsequent chronic kidney disease. Can J Kidney Health Dis 6:2054358119880188
Hessey E, Perreault S, Roy L, Dorais M, Samuel S, Phan V, Lafrance JP, Zappitelli M (2020) Acute kidney injury in critically ill children and 5-year hypertension. Pediatr Nephrol 35:1097–1107
Robinson CH, Jeyakumar N, Luo B, Wald R, Garg AX, Nash DM, McArthur E, Greenberg JH, Askenazi D, Mammen C, Thabane L, Goldstein S, Parekh RS, Zappitelli M, Chanchlani R (2021) Long-term kidney outcomes following dialysis-treated childhood acute kidney injury: a population-based cohort study. J Am Soc Nephrol 32:2005–2019
Kulvichit W, Kellum J, Srisawat N (2021) Biomarkers in acute kidney injury. Crit Care Clin 37:385–398
Mishra J, Dent C, Tarabishi R, Mitsnefes MM, Ma Q, Kelly C, Ruff SM, Zahedi K, Shao M, Bean J, Mori K, Barasch J, Devarajan P (2005) Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365:1231–1238
Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A, NGAL Meta-analysis Investigator Group (2009) Accuracy of neutrophil gelatinase associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta analysis. Am J Kidney Dis 54:1012–1024
Mårtensson J, Bellomo R (2014) The rise and fall of NGAL in acute kidney injury. Blood Purif 37:304–310
Krzych ŁJ, Czempik PF (2019) Impact of furosemide on mortality and the requirement for renal replacement therapy in acute kidney injury: a systematic review and meta-analysis of randomised trials. Ann Intensive Care 9:85
**e CM, Yao YT, Yang K, Shen MQ, He LX, Dai Z, Evidence in Cardiovascular Anesthesia (EICA) Group (2022) Furosemide does not reduce the incidence of postoperative acute kidney injury in adult patients undergoing cardiac surgery: a PRISMA-compliant systematic review and meta-analysis. J Card Surg 37:4850–4860
Chawla LS, Davison DL, Brasha-Mitchell E, Koyner JL, Arthur JM, Shaw AD, Tumlin JA, Trevino SA, Kimmel PL, Seneff MG (2013) Development and standardization of a furosemide stress test to predict the severity of acute kidney injury. Crit Care 17:R207
Lumlertgul N, Peerapornratana S, Trakarnvanich T, Pongsittisak W, Surasit K, Chuasuwan A, Tankee P, Tiranathanagul K, Praditpornsilpa K, Tungsanga K, Eiam-Ong S, Kellum JA, Srisawat N, FST Study Group (2018) Early versus standard initiation of renal replacement therapy in furosemide stress test non-responsive acute kidney injury patients (the FST trial). Crit Care 22:101
McMahon BA, Chawla LS (2021) The furosemide stress test: current use and future potential. Ren Fail 43:830–839
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group (2012) KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl 2:1–138
Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, De Zeeuw D, Hostetter TH, Lameire N, Eknoyan G (2005) Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 67:2089–2100
Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637
Pottel H, Vrydags N, Mahieu B, Vandewynckele E, Croes K, Martens F (2008) Establishing age/sex related serum creatinine reference intervals from hospital laboratory data based on different statistical methods. Clin Chim Acta 396:49–55
Kashani K, Cheungpasitporn W, Ronco C (2017) Biomarkers of acute kidney injury: the pathway from discovery to clinical adoption. Clin Chem Lab Med 55:1074–1089
Khorashadi M, Beunders R, Pickkers P, Legrand M (2020) Proenkephalin: a new biomarker for glomerular filtration rate and acute kidney injury. Nephron 144:655–661
Chen JJ, Chang CH, Huang YT, Kuo G (2020) Furosemide stress test as a predictive marker of acute kidney injury progression or renal replacement therapy: a systemic review and meta-analysis. Crit Care 24:202
Kakajiwala A, Kim JY, Hughes JZ, Costarino A, Ferguson J, Gaynor JW, Furth SL, Blinder JJ (2017) Lack of furosemide responsiveness predicts acute kidney injury in infants after cardiac surgery. Ann Thorac Surg 104:1388–1394
Borasino S, Wall KM, Crawford JH, Hock KM, Cleveland DC, Rahman F, Martin KD, Alten JA (2018) Furosemide response predicts acute kidney injury after cardiac surgery in infants and neonates. Pediatr Crit Care Med 19:310–317
Penk J, Gist KM, Wald EL, Kitzmiller L, Webb TN, Li Y, Cooper DS, Goldstein SL, Basu RK (2019) Furosemide response predicts acute kidney injury in children after cardiac surgery. J Thorac Cardiovasc Surg 157:2444–2451
Hasson DC, Zhang B, Krallman K, Rose JE, Kempton KM, Steele P, Devarajan P, Goldstein SL, Alder MN (2023) Acute kidney injury biomarker olfactomedin 4 predicts furosemide responsiveness. Pediatr Nephrol 38:3153–3161
McGalliard RJ, McWilliam SJ, Maguire S, Jones CA, Jennings RJ, Siner S, Newland P, Peak M, Chesters C, Jeffers G, Broughton C, McColl L, Lane S, Paulus S, Cunliffe NA, Baines P, Carrol ED (2020) Identifying critically ill children at high risk of acute kidney injury and renal replacement therapy. PLoS One 15:e0240360
Kari JA, Shalaby MA, Sofyani K, Sanad AS, Ossra AF, Halabi RS, Aljuhani MH, Toffaha WM, Moria FA, Sabry S, Ahmed HA, Alhasan KA, Sharief S, Safdar O (2018) Urinary neutrophil gelatinase-associated lipocalin (NGAL) and serum cystatin C measurements for early diagnosis of acute kidney injury in children admitted to PICU. World J Pediatr 14:134–142
Parikh CR, Devarajan P, Zappitelli M, Sint K, Thiessen-Philbrook H, Li S, Kim RW, Koyner JL, Coca SG, Edelstein CL, Shlipak MG, Garg AX, Krawczeski CD, TRIBE-AKI Consortium (2011) Postoperative biomarkers predict acute kidney injury and poor outcomes after pediatric cardiac surgery. J Am Soc Nephrol 22:1737–47
Hollinger A, Wittebole X, François B, Pickkers P, Antonelli M, Gayat E, Chousterman BG, Lascarrou JB, Dugernier T, Di Somma S, Struck J, Bergmann A, Beishuizen A, Constantin JM, Damoisel C, Deye N, Gaudry S, Huberlant V, Marx G, Mercier E, Oueslati H, Hartmann O, Sonneville R, Laterre PF, Mebazaa A, Legrand M (2018) Proenkephalin A 119–159 (Penkid) is an early biomarker of septic acute kidney injury: the kidney in sepsis and septic shock (Kid-SSS) study. Kidney Int Rep 3:1424–1433
Jäntti T, Tarvasmäki T, Harjola VP, Pulkki K, Turkia H, Sabell T, Tolppanen H, Jurkko R, Hongisto M, Kataja A, Sionis A, Silva-Cardoso J, Banaszewski M, DiSomma S, Mebazaa A, Haapio M, Lassus J, Investigators CardShock (2021) Predictive value of plasma proenkephalin and neutrophil gelatinase-associated lipocalin in acute kidney injury and mortality in cardiogenic shock. Ann Intensive Care 11:25
Kieneker LM, Hartmann O, Bergmann A, de Boer RA, Gansevoort RT, Joosten MM, Struck J, Bakker SJL (2018) Proenkephalin and risk of develo** chronic kidney disease: the Prevention of Renal and Vascular End-stage Disease study. Biomarkers 23:474–482
Hartman SJF, Zwiers AJM, van de Water NEC, van Rosmalen J, Struck J, Schulte J, Hartmann O, Pickkers P, Beunders R, Tibboel D, Schreuder MF, de Wildt SN (2020) Proenkephalin as a new biomarker for pediatric acute kidney injury - reference values and performance in children under one year of age. Clin Chem Lab Med 58:1911–1919
Koyner JL, Davison DL, Brasha-Mitchell E, Chalikonda DM, Arthur JM, Shaw AD, Tumlin JA, Trevino SA, Bennett MR, Kimmel PL, Seneff MG, Chawla LS (2015) Furosemide stress test and biomarkers for the prediction of AKI severity. J Am Soc Nephrol 26:2023–2031
Matsuura R, Komaru Y, Miyamoto Y, Yoshida T, Yoshimoto K, Isshiki R, Mayumi K, Yamashita T, Hamasaki Y, Nangaku M, Noiri E, Morimura N, Doi K (2018) Response to different furosemide doses predicts AKI progression in ICU patients with elevated plasma NGAL levels. Ann Intensive Care 8:8
Goldstein SL, Krallman KA, Roy JP, Collins M, Chima RS, Basu RK, Chawla L, Fei L (2023) Real-time acute kidney injury risk stratification–biomarker directed fluid management improves outcomes in critically ill children and young adults. Kidney Int Rep 8:2690–2700
Author information
Authors and Affiliations
Contributions
SK, AS, JS, RL, AB, and PH managed the patients. SK participated in study protocol preparation, recruited patients, participated in data analysis, and drafted the first version of the manuscript. AS, AB, and PH conceptualized the study design, interpreted the data, and critically revised the manuscript. JS and RL assisted in protocol preparation, recruitment of patients, and drafting the manuscript. MT and LR helped in performing biomarkers for the study. All authors contributed to protocol preparation, drafting of the manuscript, and approved the final version of the manuscript. PH shall act as the corresponding author and guarantor of the paper.
Corresponding author
Ethics declarations
Consent to participate and publish the data
The authors affirm that human research participants provided informed consent to participate in the study and publish the data.
Ethics approval
The study was approved by the Institute Ethics Committee (IECPG-590/24.10.2019, RT-15/28.11.2019, titled “Furosemide stress test in predicting progression to AKI stage III in children admitted to the ICU with early stages of AKI”).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Krishnasamy, S., Sinha, A., Lodha, R. et al. Furosemide stress test to predict acute kidney injury progression in critically ill children. Pediatr Nephrol (2024). https://doi.org/10.1007/s00467-024-06387-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00467-024-06387-5