Abstract
Cetaceans are well known for their remarkable cognitive abilities including self-recognition, sound imitation and decision making. In other mammals, the prefrontal cortex (PFC) takes a key role in such cognitive feats. In cetaceans, however, a PFC could up to now not be discerned based on its usual topography. Classical in vivo methods like tract tracing are legally not possible to perform in Cetacea, leaving diffusion-weighted imaging (DWI) as the most viable alternative. This is the first investigation focussed on the identification of the cetacean PFC homologue. In our study, we applied the constrained spherical deconvolution (CSD) algorithm on 3 T DWI scans of three formalin-fixed brains of bottlenose dolphins (Tursiops truncatus) and compared the obtained results to human brains, using the same methodology. We first identified fibres related to the medio-dorsal thalamic nuclei (MD) and then seeded the obtained putative PFC in the dolphin as well as the known PFC in humans. Our results outlined the dolphin PFC in areas not previously studied, in the cranio-lateral, ectolateral and opercular gyri, and furthermore demonstrated a similar connectivity pattern between the human and dolphin PFC. The antero-lateral rotation of the PFC, like in other areas, might be the result of the telesco** process which occurred in these animals during evolution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The bottlenose dolphin Tursiops truncatus (Montagu, 1821) is a member of the Delphinidae family often kept in captivity, and consequently frequently studied. Individuals of this species may perform a large variety of complex cognitive tasks including sound imitation, understanding of human syntax, conceptual decision taking, understanding numerosity, and self-recognition (Kilian et al. 2003; Kuczaj et al. 2015, 2016), corrected from Gibb’s ringing artefacts (Kellner et al. 2015), corrected for EPI-distortion (Holland et al. 2010), b0-field inhomogeneity (Andersson et al. 2003; Smith et al. 2004), Eddy-current and movement (Andersson and Sotiropoulos 2016). Subsequently, the dhollander algorithm was applied to compute different response functions for the white matter (WM, anisotropic), cerebrospinal fluid and grey matter (CSF and GM, both isotropic). Finally, the fibre orientation distribution (FOD) was calculated before the elaboration of the tractography (Dhollander et al. 2016a, b, 2017; Tournier et al. 2019). Each brain was then investigated individually.
Humans
Preprocessed data of human brains were already present in the HCP files, but further steps were added in order to create a template atlas, averaging all five subjects. Images were corrected form bias field and group DWI intensity normalisation was performed. In these data, there were 4 shells, therefore, the dhollander algorithm was used to estimate different response functions and then average them to calculate the FOD. Once all subjects’ FOD were complete, we created a population template, a template mask and war** all the FOD images to the template space (for reference, see Tournier et al. 2019).
Tractography
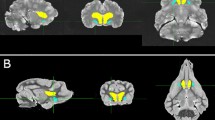
Throughout the following, the procedure was applied to both right and left hemispheres. In dolphins, the MDN mask was manually drawn using the ITK-SNAP software (Yushkevich et al. 2016; www.itksnap.org; version 3.8.0) following the topographical location described by Kruger (1959) and Morgane and Jacobs (1972). The thalamic subdivision is quite evident in the histological data if compared to our MRI data. We first identified the thalamic topographic position based on the histology, and then drew the areas. Since the slides did not include all the thickness of the thalamus, we tried to reconstruct the missing space following its shape (Fig. 2). The final mask was intentionally slightly eroded to avoid the generation of erroneous extra fibres. Since there is no map of the PFC location in Cetacea, a PFC area was first created, based from the tracts originating from the MDN. To verify that the tracts were not artefactual, we restricted the tracking using the designated PFC as the seed image and reaching the MDN nucleus, thus restraining the fibres to the bundle only connecting one with the other.
Representation of the MDN mask based on the panels descripted by Kruger (1959) and Morgane and Jacobs (1972). The directions from a to c are rostro-caudal with a 3D visualisation of the section and the whole mask at the correspondent plane; a section taken based on panel number 6; b section taken based on panel number 7; c section taken based on panel number 3
In the human, MDN and PFC were manually drawn with ITK-SNAP, based on the currently available templates and atlases (Tamraz and Comair 2000; Cho 2010; Ding et al. 2016). For both dolphin and human brains, fibre tracking was performed through a deterministic algorithm based on CSD, with the FOD file as input and the following parameters: FA threshold 0.1 for dolphin brains (as they were fixed) and 0.2 for human brains (since the subjects were alive), step size 0.1 mm, default angle threshold 60°, and default streamline count (5000).
Results
Seeding point: MDN nuclei
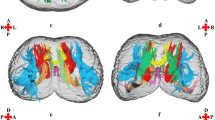
In the dolphin brains, the main fibre bundles exiting the MDN were directed cranially running below the cruciate sulcus (Cr) towards the ventro-cranial pole, passing between the Putamen (PU) and caudate nucleus (CA). Other consistent fibre bundles went dorsally following the internal capsule and terminated in i) the supralimbic cortex around the entolateral sulcus (En), the lateral sulcus (La), and the suprasylvian sulcus (Ss); then ii) laterally in the temporal lobes around the ectolateral sulcus (Es). Some tracts ran caudally to the Edinger–Westphal nucleus (EW), the Interstitial Nucleus of Cajal (INC) and the elliptic nucleus (NE), then moved ventrally towards the crus cerebri (Fig. 2a). Projections from the MDN were also commonly distributed within the thalamus and then ran to other brain areas. Few aberrant fibres passed through the superior colliculi (SC) (Fig. 3a).
Prefrontal pathways in the dolphin and human brains. a Tracts generated from seeding the MDN (blue shape) in dolphin. b Tracts generated from seeding MDN (blue shape) in human. CC corpus callosum, En entolateral sulcus, Es ectolateral sulcus, ES ectolateral gyrus, La lateral sulcus, LG lateral gyrus, Sf sylvian fissure, Ss suprasylvian sulcus, SS suprasylvian gyrus. Red scale bar = 5 cm
In the human brain, fibres ran to the PFC following the anterior thalamic peduncle, then went laterally to the temporal lobe following the inferior thalamic peduncle. Some of the latter fibres split from the inferior thalamic bundle to join the optic radiation directed towards the parietal lobe. We also noted fibres that reached the CC to spread in the contralateral hemisphere (Fig. 3b).
Thanks to our previous results, we were able to estimate a presumptive frontal region based on the extension of the fibres on the WM tracts (Fig. 4).
Seeding point: acquired PFC
Seeding the putative PFC resulted in the presence of several ipsilateral U-shaped fibres within the area. Other fibre tracts were oriented towards the basal ganglia and the cingulate cortex (CI). Other bundles continued through the MDN to end in the pons. A consistent number of fibres joined the superior longitudinal fasciculus (SLF) directed to the temporal lobe (Es, Ss) while others arose to En and La (Fig. 5a). Finally, some tracts reached the CC and crossed contralaterally.
Prefrontal pathways in the dolphin and human brains. a Tracts generated from seeding the PFC (red shape) in dolphin. b Tracts generated from seeding PFC (red shape) in human. The MDN is represented in blue. CA caudate nucleus, CC corpus callosum, CI cingulum, En entolateral sulcus, Es ectolateral sulcus, ES ectolateral gyrus, La lateral sulcus, LG lateral gyrus, PU putamen, SLF superior longitudinal fasciculus, Ss suprasylvian sulcus, SS suprasylvian gyrus. Red scale bar = 5 cm
In the human brain, ipsilateral fibres created from the PFC joined the inferior fronto-occipital fasciculus and reached the V1. Other fibres joined the fornix, then ran caudally towards the MDN, the CI. Some tracts were directed to the temporal lobe apparently through the SLF. Fibres directed towards the mesencephalon reached the periaqueductal grey and the red nucleus in the tegmentum (Fig. 5b). Finally, consistent fibre bundles moved to the CC and then crossed to the contralateral hemisphere.
Specifically constrained thalamo-cortical connections
To delimit the fibres previously generated singularly from the MDN nuclei or from the presumptive PFC, we also selectively constrained the tracking between these two regions.
In general, these fibres were fewer and more limited. In the dolphin, cortically seeded bundles passed below the Cr and between the basal ganglia to reach the MDN. Other streamlines ran laterally to the temporal lobe (Ss, Es), and very few fibres continued dorsally to the ENs and LAs. Finally, some fibre bundles were directed caudally to the red nucleus and the elliptic nucleus (Fig. 6).
Constrained tractography between the putative found PFC (red shape) and the MDN (blue shape). CA caudate nucleus, Cr cruciate sulcus, En entolateral sulcus, Es ectolateral sulcus, ES ectolateral gyrus, La lateral sulcus, LG lateral gyrus, PU putamen, Sf sylvian fissure, Ss suprasylvian sulcus, SS suprasylvian gyrus
Prefrontal pathways in the human brain between PFC and MDN also crossed through the CC and reached the temporal lobe of the contralateral hemisphere through the inferior thalamic peduncle. Few fibres detached from the inferior thalamic peduncle and continued caudally until the VC (Fig. 7).
Discussion
A precise identification of the topography and connectivity of the PFC (or its homologue) in dolphins represents a consistent step forward towards the understanding of their brain architecture and the neural basis for some of the complex behaviours of the species. To the best of our knowledge, DWI technique is currently one of the very few technically feasible and ethically acceptable approaches to identify the PFC in dolphins, and potentially other large ex-vivo brains.
In the present study, we performed CSD-based tractography and aimed at identifying fibre tracts that connect selected thalamic nucleus to their related cortical targets/origin. We then compared the data with those obtained in the human brain. Since the human PFC is one of the key areas assigned to higher brain functions (Tranel et al. 2003; Butler and Hodos 2005; Kandel et al. 2021), we searched for an area in the dolphin brain with the same characteristic connections.
CSD is a HARDI algorithm of DWI that, within the limits of human clinical MRI parameters, can reconstruct crossing fibres within a voxel, thus giving a more plausible biological result (Arrigo et al. 2016; Jeurissen et al. 2017). The algorithm can be applied and extended also to fixed brains, much like with DTI (D’Arceuil and de Crespigny 2007; Rane and Duong 2011; Gerussi et al. 2022). Classical retrograde and anterograde tract-tracing remains the gold standard methodology to accurately study brain connections. However, the technique was seldom performed in cetacean brains, and likely will not be in the future because of the ethical constraints briefly outlined in the Introduction. In this context, opportunistic fixed-brain DWI (and its various algorithmic variations), therefore, constitutes an adequate method to investigate brain connections in these mammals, even considering its biases and limits to interpretation (Jeurissen et al. 2017; Schilling et al. 2020). Fixation produces microstructural changes such as dehydration or tissue degeneration, which in turn may alter some MRI parameters including SNR, FA and apparent diffusion coefficient (ADC) (D’Arceuil and de Crespigny 2007; Rane and Duong 2011). Therefore, the obtained results must consider all the limitations of DWI in general and DWI applied to fixed tissues.
Tracing and tractography in dolphins and porpoises
Literature on evoked potential placed the bottlenose dolphin V1/V2 around En in the LG, extending rostro-caudally and with the La as its lateral boundary. A1/A2 develops from there along the SS, reaching the Ss with a rostro-caudal direction (for review see Supin et al. 2001). According to the literature, the Cr separates rostrally the medial SSC from the lateral MC. Other reports that used tracing in the harbour porpoise (Phocoena phocoena) established the existence of projections from the parvocellular part of the MGN to the suprasylvian and ectosylvian gyri (Krashnoshchekova and Figurina 1980; Voronov et al. 1985), and the involvement of the LGN as target of the optic nerve. Evoked potentials were used also for track-tracing-based injections in various areas of the neocortex (LG, the SS, ES, temporal and orbital) of the harbour porpoise (Revishchin and Garey 1990). Overall, the data obtained showed that medial thalamic projections progressively crossed to a lateral cortical position, therefore, ending contralaterally to their origin (see Revishchin and Garey 1990, Fig. 13). However, no tracing study was ever performed on a putative PFC.
A recent DTI study described for the first time the existence of a direct auditory pathway from IC to MGN to the temporal lobe near the Sylvian fissure in the common (Delphinus delphis) and pantropical spotted dolphin (Stenella attenuata), and an overlap of thalamic visual and auditory pathways (Berns et al. 2015). Such direct pathways, often hypothesised, but never demonstrated before, suggest a direct and profound interconnection of the dolphin visual and auditory system. Nevertheless, given the extremely limited data concerning the functional auditory region in cetaceans, it should be considered a still partially unsolved scientific question.
Topography and characterisation of the PFC in the human and dolphin brain
The human PFC can be divided into four functional areas: orbitofrontal cortex (OFC), dorsolateral PFC (dlPFC), dorsomedial PFC (dmPFC) and ventromedial PFC (vmPFC) (Kolb 2015). The caudolateral boundaries of the PFC roughly correspond to the cranial part of the precentral sulcus (premotor cortex) and medially to the cingulate cortex. The Brodmann areas overlap** the human PFC are: BA8-14, 24, 25, 32, 44–47 (Murray et al. 2017). These are marked by notable cytoarchitectural differences that determine their boundaries (Brodmann’s 2006; Petrides and Pandya 2012). Although the frontal lobe possess its own cytoarchitectural peculiarity (Hof et al. 2005), there are no such characteristic differences in the bottlenose dolphin, and other cetaceans in general (Kern et al. 2011; van Kann et al. 2017). We started by identifying topographical landmarks that may characterise comparatively the dolphin PFC (see orange area in the Fig. 8) and then proceeded with CSD tractography. The average mass of the brain in the bottlenose dolphin is 1.550 g (Huggenberger et al. 2019), vs. the average value of 1300–1400 g for the human (Miller and Corsellis 1977) must be taken in account. Without discussing the body and brain weight correlation, or the Encephalization QuotientFootnote 1 (Jerison 1973; for discussion of its application to cetaceans see Cozzi et al. 2016), we emphasise that the position of the area occupied by the PFC, based on topographically equivalent landmarks, appears more lateral in the dolphin brain comparatively to man, even considering the different brain shape and dimensions.
Prefrontal pathways
In the human DWI scans, seeding the human MDN (Fig. 2b) resulted in fibres that reached most of the PFC through the anterior thalamic peduncle, while some other fibres joined the inferior thalamic peduncle and were then oriented towards the somatosensory association area in the parietal lobe and V1. Our results were consistent with those found by Grodd et al. (2020). When the same method was applied to the brains of the bottlenose dolphins, the resulting fibres joined the internal capsule, passed between the basal ganglia towards the ventro-cranial pole and ended ventrally to the Cr. Other fibres were directed towards either the LGN or the MGN, going through the internal capsule and terminating in the parietal lobe. We also noted that some fibres were directed caudally, possibly to merge in post-thalamic tectal pathways. A putative PFC could be identified in the bottlenose brain by map** fibres that reached the non-parietal neocortex (Fig. 8). The projections arising from this putative PFC then continued (a) within the PFC itself; (b) moved to the contralateral PFC through the CC; (c) to the CI; (d) to the pons passing between the basal ganglia and MDN; or (e) to the temporal lobe following the SLF. The connectivity pattern detected in the bottlenose brain was in fact similar to that found in the human brain.
To check whether these fibres were not just artefactual, we first seeded the MDN to establish where the fibres were going. Then we seeded the designated area to see if some of the fibres projected independently back to the MDN. We noticed that some fibres again connected the PFC with the MDN, but other bundles were oriented towards different areas. The PFC is a multimodal association area which receives and sends inputs to other brain regions, and—to this effect—our results were consistent with what reported in the literature (Fuster 2015). We finally constrained the seeding between the two areas to qualitatively see the amount of fibres only related to these seeds, and exclude other unspecific bundles. We found a more marked asymmetry in the dolphin brains compared to human brains (Figs. 6 and 7), but given the scope of our study and its qualitative approach, we did not emphasise this aspect.
The acquisition parameters used to scan the live human brains (referred to the HCP) were evidently better than those of the (dead) dolphin brains. However, we detected no substantial loss of signal in the formalin-fixed dolphin brains, so that identification of a putative PFC area in their brain gave results that were largely comparable to the human. No former tracking or injection studies based on evoked potential reported a functional identification of a putative PFC in dolphin, hence, this is the first report of a putative PFC in this species, and, to the best of our knowledge, in all cetaceans. Its boundaries start in the orbital lobe, extend laterally in the cranial ES and reach the cranial opercular lobe. However, additional fibres were connected to areas placed below the SSC and MC cortices, in which pyramidal neurons were previously found (Manger 2006).
According to the concept of the “initial brain” (for reference see Glezer et al. 1988, Fig. 7) modern mammals still retain some key topographical features already present in the brains of their early ancestors, notwithstanding the divergent evolutionary path of the taxa. The enlarged human neocortex contains far more and varied cortical modules than in primordial mammals. The cetacean brain obviously followed a different evolutionary route since the brain mantle greatly expanded to the uttermost known limits for mammals in terms of relative cortical size, while the cytoarchitecture is poorly differentiated and mostly homogeneous in the neocortex (Cozzi et al. 2017).
In the human brain, the development of the PFC displaced all the areas caudally, thus modifying the primeval topographical scheme (Fig. 9). In cetaceans, the progressive evolution to life in the water caused a wide range of body structure modifications. In the head specifically, the growth of the melon, the telesco** process, and the nasal shift which caused the change of the cranial axes and shape, might have prevailed over the longitudinal development of the brain (Miller 1923; Cozzi et al. 2017; Roston and Roth 2019). Consequently, the brain folded around the insula (see dotted arrows in Fig. 9) and expanded more laterally: what in terrestrial mammals is “caudal” becomes “dorsal” in the dolphin and so forth (Morgane and Jacobs 1972). Concomitant space limits within the cerebral cavity prompted a striking cortical gyrification and induced the topographical shift of neocortical areas. Our data based on CSD tractography confirmed that the (evolutionary) process that modified the cetacean brain was compatible with the persistence of a very large and richly connected PFC area.
Schematic picture of the brain evolution from its “initial” form (top, Glezer et al. 1988) to the dolphin (Jacobs et al. 1984; Cozzi et al. 2017) and human (Martin 2021) brains (bottom). Orange, PFC; Blue, SSC; Red, MC; Green, A1/A2; Yellow, V1/V2. Sf, Sylvian fissure. Dashed purple line delimits the border of the insular cortex. The dotted arrow in the human brain indicates the displacement of the other areas due to the expansion of the PFC. The dotted arrows in the dolphin brain indicate the rotation of the organ around the insular cortex and the probable cranio-lateral shift of the PFC and other cortical areas on the surface. X, Y body axes
The PFC of terrestrial mammals, and the correspondent areas in birds, are highly innervated by dopaminergic neurons (Sawaguchi and Goldman-Rakic 1991; Gaspar et al. 1992; Güntürkün 2005). Based on this neurochemical concept, further studies are needed to investigate the actual presence of dopaminergic neurons in the PFC of dolphins. This is particularly important in the fronto-ventral area, where one should not expect to find an associative cortex (Manger 2006, referring to Kojima 1951), and in the lateral (opercular) area. Additional functional MRI (fMRI) or recently developed functional near-infrared spectroscopy (fNIRS) techniques (Scholkmann et al. 2014) might also become useful for the study of brain functions through the proxy of increased blood flow during local stimulation, very similar to blood-oxygen-level-dependent contrast in fMRI. A composite multidisciplinary approach may eventually shed light on the unmapped cortical fields of these mammals with remarkable cognitive abilities.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Notes
EQ [EQ = Ei / 0.12 × P2/3; E.i = mean brain weight, P = mean body weight] is 3.79 in the bottlenose dolphin vs. 6.62 in man, according to Huggenberger et al. (2019)
Abbreviations
- En:
-
Entolateral sulcus
- Es:
-
Ectolateral sulcus
- La:
-
Lateral sulcus
- Sf:
-
Sylvian fissure
- Ss:
-
Suprasylvian sulcus
- AC:
-
Anterior commissure
- CC:
-
Corpus callosum
- Cl:
-
Cingulate cortex
- CSD:
-
Constrained spherical deconvolution
- DTI:
-
Diffusion tensor imaging
- DWI:
-
Diffusion-weighted imaging
- ES:
-
Ectolateral gyrus
- MC:
-
Motor cortex
- MDN:
-
Medio-dorsal thalamic nucleus
- MGN:
-
Medial geniculate nucleus
- LG:
-
Lateral gyrus
- LGN:
-
Lateral geniculate nucleus
- OT:
-
Optic tract
- PFC:
-
Prefrontal cortex
- SS:
-
Suprasylvian gyrus
- SSC:
-
Somatosensory cortex
References
Andersson JLR, Sotiropoulos SN (2016) An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage 125:1063–1078. https://doi.org/10.1016/j.neuroimage.2015.10.019
Andersson JLR, Skare S, Ashburner J (2003) How to correct susceptibility distortions in spin-echo echo-planar images: application to diffusion tensor imaging. Neuroimage 20:870–888. https://doi.org/10.1016/S1053-8119(03)00336-7
Arrigo A, Calamuneri A, Mormina E (2016) Diffusion MRI: from principles to modelling and clinical applications. Biomedical imaging. Dover, SmGroup, pp 1–18
Basser PJ (1995) Inferring microstructural features and the physiological state of tissues from diffusion-weighted images. NMR Biomed 8:333–344. https://doi.org/10.1002/nbm.1940080707
Berns GS, Cook PF, Foxley S, Jbabdi S, Miller KL, Marino L (2015) Diffusion tensor imaging of dolphin brains reveals direct auditory pathway to temporal lobe. Proc R Soc B 282:20151203. https://doi.org/10.1098/rspb.2015.1203
Brodmann K (2006) Localisation in the cerebral cortex. Springer, New York. https://doi.org/10.1007/b138298
Bullock TH, Gurevich VS (1979) Soviet literature on the nervous system and psychobiology of cetacea. Int Rev Neurobiol. https://doi.org/10.1016/s0074-7742(08)60637-6
Butler AB, Hodos W (2005) Comparative vertebrate neuroanatomy: evolution and adaptation. John Wiley & Sons, Hoboken
Calamuneri A, Arrigo A, Mormina E et al (2018) White matter tissue quantification at low b-values within constrained spherical deconvolution framework. Front Neurol. https://doi.org/10.3389/fneur.2018.00716
Cho Z-H (2010) 7.0 Tesla MRI brain atlas. Springer, New York. https://doi.org/10.1007/978-1-60761-154-7
Cozzi B, Mazzariol S, Podestà M et al (2016) An unparalleled sexual dimorphism of sperm whale encephalization. Int J Comp Psychol. https://doi.org/10.46867/ijcp.2016.29.00.01
Cozzi B, Huggenberger S, Oelschläger H (2017) Brain, spinal cord, and cranial nerves. In: Cozzi B, Huggenberger S, Oelschläger H (eds) Anatomy of dolphins. Elsevier, Amsterdam, pp 197–304. https://doi.org/10.1016/b978-0-12-407229-9.00006-3
D’Arceuil H, de Crespigny A (2007) The effects of brain tissue decomposition on diffusion tensor imaging and tractography. Neuroimage 36:64–68. https://doi.org/10.1016/j.neuroimage.2007.02.039
Dhollander T, Connelly A (2016a) Generating a T1-like contrast using 3-tissue constrained spherical deconvolution results from single-shell (or multi-shell) diffusion MR data. Conf ISMRM Work Break Barriers Diffus. MRI
Dhollander T, Raffelt D, Connelly A (2016b) Unsupervised 3-tissue response function estimation from single-shell or multi-shell diffusion MR data without a co-registered T1 image. In: Conference: ISMRM Works Break Barriers Diffus. MRI. Lisbon, Portugal, p 5
Dhollander T, Raffelt D, Connelly A (2017) Towards interpretation of 3-tissue constrained spherical deconvolution results in pathology. Proc Intl Soc Mag Reson Med 25:1815
Ding S, Royall JJ, Sunkin SM et al (2016) Comprehensive cellular-resolution atlas of the adult human brain. J Comp Neurol 524:3127–3481. https://doi.org/10.1002/cne.24080
Dinopoulos A, Karamanlidis AN, Papadopoulos G et al (1985) Thalamic projections to motor, prefrontal, and somatosensory cortex in the sheep studied by means of the horseradish peroxidase retrograde transport method. J Comp Neurol 241:63–81. https://doi.org/10.1002/cne.902410106
Fuster JM (2015) Anatomy of the prefrontal cortex. Prefrontal Cortex. https://doi.org/10.1016/b978-0-12-407815-4.00002-7
Garey LJ, Revishchin AV (1990) Structure and thalamocortical relations of the cetacean sensory cortex: histological, tracer and immunocytochemical studies. In: Thomas JA, Kastelein RA (eds) Sensory abilities of cetaceans. Springer, New York, pp 19–30. https://doi.org/10.1007/978-1-4899-0858-2_2
Gaspar P, Stepniewska I, Kaas JH (1992) Topography and collateralization of the dopaminergic projections to motor and lateral prefrontal cortex in owl monkeys. J Comp Neurol 325:1–21. https://doi.org/10.1002/cne.903250102
Gerussi T, Graïc J-M, Grandis A et al (2022) The orbitofrontal cortex of the sheep. Topography, organization, neurochemistry, digital tensor imaging and comparison with the chimpanzee and human. Brain Struct Funct 227:1871–1891. https://doi.org/10.1007/s00429-022-02479-w
Glezer II, Jacobs MS, Morgane PJ (1988) Implications of the “initial brain” concept for brain evolution in Cetacea. Behav Brain Sci 11:75–89. https://doi.org/10.1017/s0140525x0005281x
Grodd W, Kumar VJ, Schüz A et al (2020) The anterior and medial thalamic nuclei and the human limbic system: tracing the structural connectivity using diffusion-weighted imaging. Sci Rep. https://doi.org/10.1038/s41598-020-67770-4
Güntürkün O (2005) The avian ‘prefrontal cortex’ and cognition. Curr Opin Neurobiol 15:686–693. https://doi.org/10.1016/j.conb.2005.10.003
Güntürkün O (2014) Is dolphin cognition special? Brain Behav Evol 83:177–180. https://doi.org/10.1159/000357551
Herman LM (2012) Body and self in dolphins. Conscious Cogn 21(1):526–545. https://doi.org/10.1016/j.concog.2011.10.005
Hof PR, Chanis R, Marino L (2005) Cortical complexity in cetacean brains. Anat Rec 287A:1142–1152. https://doi.org/10.1002/ar.a.20258
Hofman MA (1985) Size and shape of the cerebral cortex in mammals. Brain Behav Evol 27:28–40. https://doi.org/10.1159/000118718
Holland D, Kuperman JM, Dale AM (2010) Efficient correction of inhomogeneous static magnetic field-induced distortion in echo planar imaging. Neuroimage 50:175–183. https://doi.org/10.1016/j.neuroimage.2009.11.044
Huggenberger S, Oelschläger H, Cozzi B (2019) The nervous system. In: Huggenberger S, Oelschläger H, Cozzi B (eds) Atlas of the anatomy of dolphins and whales. Elsevier, Amsterdam, pp 323–362. https://doi.org/10.1016/b978-0-12-802446-1.00005-9
Jacobs MS, Galaburda AM, McFarland WL, Morgane PJ (1984) The insular formations of the dolphin brain: quantitative cytoarchitectonic studies of the insular component of the limbic lobe. J Comp Neurol 225:396–432
Jefferson TA, Webber MA, Pitman RL (2015) Cetaceans. In: Jefferson TA, Webber MA, Pitman RL (eds) Marine mammals of the world. Elsevier, Amsterdam, pp 24–357. https://doi.org/10.1016/b978-0-12-409542-7.50004-4
Jenkinson M, Beckmann CF, Behrens TEJ et al (2012) FSL Neuroimage 62:782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015
Jerison HJ (1973) Evolution of the brain and intelligence. Academic Press, New York
Jeurissen B, Descoteaux M, Mori S, Leemans A (2017) Diffusion MRI fiber tractography of the brain. NMR Biomed. https://doi.org/10.1002/nbm.3785
Kandel ER, Koester JD, Mack SH, Siegelbaum SA (2021) Principles of neural science, 6th edn. McGraw-hill, New York
Kellner E, Dhital B, Kiselev VG, Reisert M (2015) Gibbs-ringing artifact removal based on local subvoxel-shifts. Magn Reson Med 76:1574–1581. https://doi.org/10.1002/mrm.26054
Kern A, Siebert U, Cozzi B et al (2011) Stereology of the neocortex in odontocetes: qualitative, quantitative, and functional implications. Brain Behav Evol 77:79–90. https://doi.org/10.1159/000323674
Kilian A, Yaman S, von Fersen L, Güntürkün O (2003) A bottlenose dolphin discriminates visual stimuli differing in numerosity. Anim Learn Behav 31:133–142. https://doi.org/10.3758/bf03195976
Kojima T (1951) On the brain of the sperm whale (Physeter catodon L.). Sci Rep Whales Res Inst Tokyo 6:49–72
Kolb B (2015) Prefrontal cortex. Int Encycl Soc Behav Sci. https://doi.org/10.1016/b978-0-08-097086-8.55045-4
Krasnoshchekova EI, Figurina II (1980) Korkovye proektsii medial’nogo kolenchatogo tela mozga del’fina. Arkh Anat Gistol Embriol 4:19–24
Kruger L (1959) The thalamus of the dolphin (Tursiops truncatus) and comparison with other mammals. J Comp Neurol 111:133–194. https://doi.org/10.1002/cne.901110107
Kruger L (1966) Specialized features of the cetacean brain. In: Norris KS (ed) Whales, dolphins and porpoises. University of California Press, University, pp 232–254
Kuczaj Il SA, Gory JD, **tco MJ Jr (2009) How intelligent are dolphins? A partial answer based on their ability to plan their behavior when confronted with novel problems. Jpn J Anim Psychol 59:99–115. https://doi.org/10.2502/janip.59.1.9
Ladygina TF, Supin AI (1970) Acoustic projection in the dolphin cerebral cortex. Fiziologicheskii Zhurnal SSSR Imeni IM Sechenova 56(11):1554–1560
Ladygina TF, Supin AY (1974) Morphology, physiology and acoustics of marine mammals. Nauka, Moskow
Lende RA, Akdikmen S (1968) Motor field in cerebral cortex of the bottlenose dolphin. J Neurosurg 29:495–499. https://doi.org/10.3171/jns.1968.29.5.0495
Lende RA, Welker WI (1972) An unusual sensory area in the cerebral neocortex of the bottlenose dolphin, Tursiops truncatus. Brain Res 45:555–560. https://doi.org/10.1016/0006-8993(72)90482-9
Loth A, Güntürkün O, von Fersen L, Janik VM (2022) Through the looking glass: how do marked dolphins use mirrors and what does it mean? Anim Cogn 25:1151–1160. https://doi.org/10.1007/s10071-022-01680-y
Mai JK, Forutan F (2012) Thalamus. In: Mai JK, Paxinos G (eds) The human nervous system. Elsevier, Amsterdam, pp 618–677. https://doi.org/10.1016/B978-0-12-374236-0.10019-7
Manger PR (2006) An examination of cetacean brain structure with a novel hypothesis correlating thermogenesis to the evolution of a big brain. Biol Rev 81:293. https://doi.org/10.1017/s1464793106007019
Martin JH (2021) Neuroanatomy, 5th edn. McGraw-hill, New York
Miller GS (1923) The telesco** of the cetacean skull (with eight plates). Smithso Misc Collect 76:1
Miller AKH, Corsellis JAN (1977) Evidence for a secular increase in human brain weight during the past century. Ann Hum Biol 4:253–257. https://doi.org/10.1080/03014467700007142
Morgane PJ, Jacobs MS (1972) Comparative anatomy of the cetacean nervous system. In: Harrison RJ (ed) Functional anatomy of marine mammals, vol 1. Academic Press, Cambridge, pp 117–244
Murray EA, Wise SP, Graham KS (2017) The evolution of memory systems: ancestors, anatomy, and adaptations. Oxford University Press, Oxford
Petrides M, Pandya DN (2012) The frontal cortex. In: Mai JK, Paxinos G (eds) The human nervous system. Elsevier, Amstredam, pp 988–1011. https://doi.org/10.1016/b978-0-12-374236-0.10026-4
Rane S, Duong TQ (2011) Comparison of in vivo and ex vivo diffusion tensor imaging in rhesus macaques at short and long diffusion times. Open Neuroimag J 5:172–178. https://doi.org/10.2174/1874440001105010172
Revishchin AV, Garey LJ (1990) The thalamic projection to the sensory neocortex of the porpoise, Phocoena phocoena. J Anat 169:85–102
Ridgway SH, Brownson RH (1984) Relative brain sizes and cortical surface areas in odontocetes. Acta Zool Fenn 172:149–152
Rose JE, Woolsey CN (1948) Structure and relations of limbic cortex and anterior thalamic nuclei in rabbit and cat. J Comp Neurol 89(3):279–347. https://doi.org/10.1002/cne.900890307
Roston RA, Roth VL (2019) Cetacean skull telesco** brings evolution of cranial sutures into focus. Anat Rec 302:1055–1073. https://doi.org/10.1002/ar.24079
Sawaguchi T, Goldman-Rakic PS (1991) D1 dopamine receptors in prefrontal cortex: involvement in working memory. Science 251:947–950. https://doi.org/10.1126/science.1825731
Schilling KG, Petit L, Rheault F et al (2020) Brain connections derived from diffusion MRI tractography can be highly anatomically accurate—if we know where white matter pathways start, where they end, and where they do not go. Brain Struct Funct 225:2387–2402. https://doi.org/10.1007/s00429-020-02129-z
Scholkmann F, Kleiser S, Metz AJ et al (2014) A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. Neuroimage 85:6–27. https://doi.org/10.1016/j.neuroimage.2013.05.004
Smith SM, Jenkinson M, Woolrich MW et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23:S208–S219. https://doi.org/10.1016/j.neuroimage.2004.07.051
Sokolov VE, Ladygina TF, Supin AI (1972) Localization of sensory zones in the dolphin cerebral cortex. Doklady Akademii Nauk SSSR 202(2):490–493. http://www.ncbi.nlm.nih.gov/pubmed/4333815
Supin AY, Popov VV, Mass AM (2001) The Sensory Physiology of Aquatic Mammals. Springer, US. https://doi.org/10.1007/978-1-4615-1647-7
Tamraz JC, Comair YG (2000) Atlas of regional anatomy of the brain using MRI. Springer-Verlag. https://doi.org/10.1007/3-540-30672-2
Tournier J-D, Calamante F, Connelly A (2007) Robust determination of the fibre orientation distribution in diffusion MRI: non-negativity constrained super-resolved spherical deconvolution. Neuroimage 35:1459–1472. https://doi.org/10.1016/j.neuroimage.2007.02.016
Tournier J-D, Calamante F, Connelly A (2012) MRtrix: diffusion tractography in crossing fiber regions. Int J Imaging Syst Technol 22:53–66. https://doi.org/10.1002/ima.22005
Tournier J-D, Smith R, Raffelt D et al (2019) MRtrix3: a fast, flexible and open software framework for medical image processing and visualisation. Neuroimage 202:116137. https://doi.org/10.1016/j.neuroimage.2019.116137
Tranel D, Cooper G, Rodnitzky RL (2003) Higher brain functions. In: Conn PM (ed) Neuroscience in medicine. Humana Press, New Jersey, pp 621–639. https://doi.org/10.1007/978-1-59259-371-2_29
Tyack PL (2009) Behavior, overview. In: Perrin WF, Würsig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Elsevier, Amsterdam, pp 101–108. https://doi.org/10.1016/b978-0-12-373553-9.00029-8
van Kann E, Cozzi B, Hof PR, Oelschläger HHA (2017) Qualitative and quantitative analysis of primary neocortical areas in selected mammals. Brain Behav Evol 90:193–210. https://doi.org/10.1159/000477431
Veraart J, Fieremans E, Novikov DS (2015) Diffusion MRI noise map** using random matrix theory. Magn Reson Med 76:1582–1593. https://doi.org/10.1002/mrm.26059
Veraart J, Novikov DS, Christiaens D et al (2016) Denoising of diffusion MRI using random matrix theory. Neuroimage 142:394–406. https://doi.org/10.1016/j.neuroimage.2016.08.016
Voronov VA, Krasnoshchekova EI, Stosman IM, Figurina II (1985) Morphofunctioinal organization and cortical projections of the medial geniculate body in the harbour porpoise Phocaena phocaena. J Evol Biochem Phys 21(1):45–51
Woolrich MW, Jbabdi S, Patenaude B et al (2009) Bayesian analysis of neuroimaging data in FSL. Neuroimage 45:S173–S186. https://doi.org/10.1016/j.neuroimage.2008.10.055
Yaman S, Kilian A, von Fersen L, Güntürkün O (2012) Evidence for a numerosity category that is based on abstract qualities of “few” vs “many” in the bottlenose dolphin (Tursiops truncatus). Front Psychol. https://doi.org/10.3389/fpsyg.2012.00473
Yushkevich PA, Gao Y, Gerig G (2016) ITK-SNAP: An interactive tool for semi-automatic segmentation of multi-modality biomedical images. In 2016 38th annual international conference of the IEEE engineering in medicine and biology society (EMBC). IEEE. pp. 3342–3345
Acknowledgements
The authors thank the Human Connectom Project for the available data used in this manuscript. (Principal Investigators: Bruce Rosen, M.D., Ph.D., Arthur W. Toga, Ph.D., Van J. Weeden, M.D.). HCP funding was provided by the National Institute of Dental and Craniofacial Research (NIDCR), the National Institute of Mental Health (NIMH) and the National Institute of Neurological Disorders and Stroke (NINDS). HCP data are disseminated by the Laboratory of Neuro Imaging at the University of Southern California).
Funding
Open access funding provided by Università degli Studi di Padova within the CRUI-CARE Agreement. This work was supported by a grant from the German Research Council to OG (Gu 227/16-1), and from the University of Padova D.O.R. Bruno Cozzi.
Author information
Authors and Affiliations
Contributions
Conceptualisation: TG, OG, BC; methodology: TG, MB, LS; formal analysis and investigation: TG, JMG, AP, SH, BC; writing—original draft preparation: TG; writing—review and editing: JMG, MB, LS, SH, OG, BC; funding acquisition: OG, BC; supervision: JMG, MB, LS, SH, OG, BC.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The dolphin brains used in this study were sampled during routine necropsies at the University. Therefore, no animals were deliberately killed for the purpose of this study. The human scans were freely accessible on the Human Connectom Project website, under the condition of adding them in the acknowledgments.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gerussi, T., Graïc, JM., Peruffo, A. et al. The prefrontal cortex of the bottlenose dolphin (Tursiops truncatus Montagu, 1821): a tractography study and comparison with the human. Brain Struct Funct 228, 1963–1976 (2023). https://doi.org/10.1007/s00429-023-02699-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02699-8