Abstract
To explore the microbial community structure and ecological function of mulberry and their potential relationship with the resistance of mulberry, the community structure and function of endophytic fungi in 18 mulberry cultivars were analyzed and predicted by using high-throughput sequencing technology and the FUNGuild database. A total of 352 operational taxonomic units of fungi were observed at a 97% similarity level, representing six phyla of fungi, Fungi_unclassified, Ascomycota, Basidiomycota, Zygomycota, Rozellomycota, and Chytridiomycota. Fungi_unclassified was dominant, and Ascomycota was relatively dominant in all cultivars. At the genus level, Ascomycota_unclassified was dominant, and Ampelomyces was relatively dominant, with a richness in TAIWANCHANGGUOSANG 16.47–8975.69 times that in the other cultivars. Classified Ascomycota_unclassified was 4.75–296.65 times more common in NANYUANSIJI than in the other cultivars. Based on the FUNGuild analysis method, we successfully annotated six nutrient types, namely, pathotroph, pathotroph–saprotroph, pathotroph–saprotroph–symbiotroph, saprotroph, saprotroph–symbiotroph, and symbiotroph, among which saprophytic–symbiotic accounted for the largest proportion and was absolutely dominant in TWC. This research suggests that community composition differs among cultivars and that the diversity and richness of endophytic fungi in resistant cultivars are higher than those in susceptible cultivars. The ecological functions of cultivars with different resistances are quite different.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endophytes widely exist in plants and are an important part of plant microecosystems. In the long evolutionary process, they have established a special symbiotic relationship with parasitic plants, which can promote the growth of host plants, enhance their adaptability, enhance their tolerance to abiotic and biological threats, and promote the accumulation of secondary metabolites [30]. According to relevant reports [23, 24, 31] and observations on the incidence of sclerotiniosis at the mulberry fruit base of the Chongqing Academy of Agricultural Sciences, TWC is an immune cultivar against sclerotiniosis, and JZS, DBE, NYSJ, and YK display good disease resistance, indicating that a higher microbial community diversity index means higher resistance, which is in line with the results of Allison [32] and Wu et al. [17].
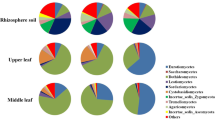
Plant endophytic fungi mainly include Ascomycota, Basidiomycota, and Zygomycota [33]. In this study, the endophytic fungi in the mulberry stems included 5 phyla in addition to Fungi_unclassified, namely, Ascomycota, Basidiomycota, Zygomycota, and a very small amount of Rozellomycota and Chytridomycota. Ascomycota is the dominant phylum, which is similar to the case of mulberry fruit [34]. The study indicates that there are significant differences among the community structures of different cultivars. Ascomycota and Basidiomycota are common phyla, and Zygomycota was found only in four typical cultivars, namely, NYSJ, JZS, DBE, and TWC. A small number of Chytridiomycota were found in A7 (0.03%) and T6 (0.13%), the two cultivars collected in **njiang. Rozellomycota was found in YK. It is speculated that these differences may be closely related to the mulberry cultivar or resistance. As shown in the heatmap graph, specific genera or dominant genera with significantly high abundances exist in several typical mulberry cultivars. The most noticeable genus was Ampelomyces, of which the abundance in TWC was 16.47–8975.69 times that in the other cultivars. It is considered to be a biocontrol bacterium of epiparasitism. The secretion of antibacterial quinone compounds [35] directly affects the health of plants during the interaction between microorganisms and plants, thus improving plant disease resistance [36]. Some strains were later registered as commercial fungicide products after being used as biocontrol bacteria for the prevention and control of powdery mildew in important crops such as grapes and cucumbers [37]. Importantly, they are not just the host strains of powdery mildew. They are also speculated to be parasitic on other pathogenic fungi, including Botrytis cinerea, Alternaria solani, Colletotrichum coccodes, and Cladosporium cucumerinum [38]. Such findings provide a new approach for the biological control of mulberry fruit sclerotiniosis. In addition, a large number of Ascomycota_unclassified existing in NYSJ are likely to fall within a new species that is speculated to be capable of playing an important role in biological control.
Although sequencing technology allows researchers to obtain large microorganism resources, many of the microorganism cultivars may play only a minor role in ecological functions [39]. In this study, relevant functional information for 77.00%–98.74% of OTUs was not obtained, which is speculated to be because access to functional prediction information is limited owing to the incomplete information in the fungal function prediction database [22] or because the current investigations into mulberry endophytic fungi are still too scarce, which means that microbial resources and ecological functions need to be further developed. The study indicates that there are certain differences in the nutritional types and ecological flora among the cultivars. Overall, saprotroph–symbiotroph, pathotroph, and saprotroph were the main types. According to the clustering results, the typical species are TWC, NYSJ, BYW, DBE, and YK, which is in line with the results of the endophytic diversity analysis. Therefore, combining the results of endophytic fungal diversity and ecological function prediction, it can be inferred that the endophytic fungal diversity and richness of the cultivars with strong disease resistance are higher, and the ecological function is significantly different from that of the susceptible cultivars. The following points need to be emphasized: the abundance of plant pathogens in TWC was the lowest, and other fungi were highly abundant. Ampelomyces was the absolutely dominant genus (98.69%) among the other fungi. According to the analysis above, the high resistance of TWC to sclerotiniosis may be correlated with Ampelomyces. BYW has a low abundance of plant pathogens and an absolute predominance of wood saprotrophs. As the dominant genus that grows in BYW only, Ceriporia (91.47%) has many functions in agriculture, and it can improve the utilization efficiency of soil nutrients by crops [40, 41]. Notably, Ceriporia can also synthesize a series of secondary metabolites and has gained attention as a microbial source of biocatalysts for the biotransformation of natural terpenoid products [42]. The in vitro antifungal activities of terpenoids against a variety of plant pathogenic fungi were evaluated by the mycelial growth rate method, and the results showed that terpenoids exhibited broad-spectrum antifungal activity [43]; thus, the fungi can be considered potential biocontrol fungi [44]. Although the relative abundance of pathotrophs in LS is high, the dominant fungus (Hirsutella) is a parasitic fungus that has a good biological control effect on plant pathogenic nematodes such as mites, stem nematodes, root-knot nematodes, and some lepidopteran insects [45].
Conclusion
In this study, high-throughput sequencing technology was applied to obtain information on the endophyte communities in the stems of different mulberry cultivars. By comparative analyses, we found that there are certain differences in terms of the diverse structures of endophyte communities of different cultivars and that the higher the microbial diversity is, the stronger the plant resistance is. It was also found that the resistant endophytic fungi demonstrate particularity in quantity or strains, which may be of certain value in investigations into plant resistance. Furthermore, cluster analysis also showed differences between mulberries with high resistance and other species in terms of ecological function. The research results can provide theoretical references for the construction of mulberry microbial information systems, the screening of antagonistic strains against sclerotinia, the ecological utilization of mulberry microorganisms and the interactions between mulberry and microbes.
References
Jia M, Chen L, **n HL, Zheng CJ, Rahman K, Han T, Qin LP (2016) A friendly relationship between endophytic fungi and medicinal plants: a systematic review. Front Microbiol 7:906–912. https://doi.org/10.3389/fmicb.2016.00906
Foyer CH, Noctor G (2011) Ascorbate and glutathione: the heart of the redox hub. Plant Physiol 155(1):2–18. https://doi.org/10.1104/pp.110.167569
Cosme M, Lu J, Erb M, Stout MJ, Franken P, Wurst S (2016) A fungal endophyte helps plants to tolerate root herbivory through changes in gibberellin and jasmonate signaling. New Phytol 211(3):1065–1076. https://doi.org/10.1111/nph.13957
Mishra A, Singh SP, Mahfooz S, Singh SP, Bhattacharya A, Mishra N, et al (2018) Endophyte mediated modulation of defense-responsive genes and systemic resistance in Withania somnifera (L.) Dunal under Alternaria alternata stress. Appl Environ Microbiol 84(8):e02845. https://doi.org/10.1128/AEM.02845-17
Pappas ML, Liapoura M, Papantoniou D, Avramidou M, Kavroulakis N, Weinhold A et al (2018) The beneficial endophytic fungus Fusarium solani strain k alters tomato responses against spider mites to the benefit of the plant. Front Plant Sci 9:1603–1621. https://doi.org/10.3389/fpls.2018.01603
Jha P, Kaur T, Chhabra I, Pania A, Paul S, Kumar V (2023) Endophytic fungi: hidden treasure chest of antimicrobial metabolites interrelationship of endophytes and metabolites. Front Microbiol. https://doi.org/10.3389/fmicb.2023.1227830
Finkel OM, Burch AY, Lindow SE, Post AF, Belkin S (2011) Geographical location determines the population structure in phyllosphere microbial communities of a salt-excreting desert tree. Appl Environ Microbiol 77(21):7647–7655. https://doi.org/10.1128/AEM.05565-11
Hunter PJ, Pink DAC, Bending GD (2015) Cultivar-level genotype differences influence diversity and composition of lettuce (Lactuca sp.) phyllosphere fungal communities. Fungal Ecol 17:183–186. https://doi.org/10.1016/j.funeco.2015.05.007
Huang ZY, Liu WJ, Qin RL, Pang SC, **ao J, Yang SD (2021) Endophytic bacterial community composition and PICRUSt gene functions in different pumpkin varieties. Sci Agric Sin 54(18):4018–4032. https://doi.org/10.3864/j.issn.0578-1752.2021.18.019
Ashkezari SJ, Fotouhifar KB (2017) Diversity of endophytic fungi of common yew (Taxus baccata L.) in Iran. Mycol Progr 16(3):247–256. https://doi.org/10.1007/s11557-017-1274-4
Paunović SM, Mašković P, Milinković M (2020) Determination of primary metabolites, vitamins and minerals in black mulberry (Morus nigra) berries depending on altitude. Erwerbs-obstbau 62:355–360. https://doi.org/10.1007/s10341-020-00509-7
Ou T, Xu WF, Wang F, Strobel G, Zhou ZY, **ang ZH et al (2019) A microbiome study reveals seasonal variation in endophytic bacteria among different mulberry cultivars. Comput Struct Biotechnol J 17:1091–1100. https://doi.org/10.1016/j.csbj.2019.07.018
Huang ET, Tian LD, **ao LZ (1992) Practical mulberry conservation. ChengDu: Sichuan Science and Technology Press, pp 114–117
Ju WT, Kim HB, Sung GB, Park KY, Kim YS (2016) Mulberry popcorn disease occurrence in Korea region and development of integrative control method. Int J Ind Entomol 33(1):36–40. https://doi.org/10.7852/ijie.2016.33.1.36
Dessalegn Y, Ayalew A, Woldetsadik K (2013) Integrating plant defense inducing chemical, inorganic salt and hot water treatments for the management of postharvest mango anthracnose. Postharvest Biol Technol 85:83–88. https://doi.org/10.1016/j.postharvbio.2013.05.003
Zheng ZY, Yang Y, Zhang MH, Ren JQ, Zeng XL (2015) Field resistance evaluation of 21 fruit mulberry germplasm resources against mulberry fruit hypertrophic sclerote disease. Acta Sericol Sin 41(6):1011–1016. https://doi.org/10.13441/j.cnki.cykx.2015.06.006
Wu YY, Xu WF, Luo Q, Wang LL, Liu WY, Chen K et al (2018) Diversity analysis of endophytic bacteria of different mulberry cultivars by Illumina MiSeq high-throughput sequencing. Newslett Sericult Sci 38(3):1–10
Liu B, Lai JH, Wu SM, Jiang JX, Kuang WG (2021) Endophytic bacterial community diversity in two citrus cultivars with different citrus canker disease resistance. Arch Microbiol 203(9):5453–5462. https://doi.org/10.1007/s00203-021-02530-0
Xu L, Ruan XL, Li DL, Li HP (2012) Endophytic bacterial diversity and communities of banana cultivars with different resistance to fusarium wilt disease. Microbiol China 39(9):1250–1259. https://doi.org/10.13344/j.microbiol.china.2012.09.003
Kiani T, Khan SA, Noureen N, Yasmin T, Zakria M, Ahmed H et al (2019) Isolation and characterization of culturable endophytic bacterial community of stripe rust-resistant and stripe rust-susceptible Pakistani wheat cultivars. Int Microbiol 22(2):191–201. https://doi.org/10.1007/s10123-018-00039-z
Tian XL, Xu D, Sun TT, Zhao SY, Li Y, Wang DD (2020) Plant resistance and leaf chemical characteristic jointly shape phyllosphere bacterial community. World J Microbiol Biotechnol 36(9):139. https://doi.org/10.1007/s11274-020-02908-0
Nguyen NH, Song ZW, Bates ST, Branco S, Tedersoo L, Menke J et al (2015) FUNGuild: An open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol 20:241–248. https://doi.org/10.1016/j.funeco.2015.06.006
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73(16):5261–5267. https://doi.org/10.1128/AEM.00062-07
Jami E, Israel A, Kotser A, Mizrahi I (2013) Exploring the bovine rumen bacterial community from birth to adulthood. ISME J 7(6):1069–1079. https://doi.org/10.1038/ismej.2013.2
Wang Y, Sheng HF, He Y, Wu JY, Jiang YX, Tam NF-Y et al (2012) Comparison of the levels of bacterial diversity in freshwater, intertidal wetland, and marine sediments by using millions of illumina tags. Appl Environ Microbiol 78(23):8264–8271. https://doi.org/10.1128/aem.01821-12
Toju H, Kishida O, Katayama N, Takagi K (2016) Networks depicting the fine-scale cooccurrences of fungi in soil horizons. PLoS ONE 11(11):e0165987. https://doi.org/10.1371/journal.pone.0165987
Zhang AM, Guo BM, Han XY, Li RX (2020) Diversity of endophytic bacteria in seeds of Hippophae rhamnoides subsp. sinnensis in two different habitats. Acta Ecol Sin 40(15):5247–5257.
Zheng YK, Qiao XG, Miao CP, Liu K, Chen YW, Xu LH et al (2016) Diversity, distribution and biotechnological potential of endophytic fungi. Ann Microbiol 66:529–542. https://doi.org/10.1007/s13213-015-1153-7
Tan RX, Zou WX (2001) Endophytes:a rich source of functional metabolites. Nat Prod Rep 18(4):448–459. https://doi.org/10.1039/b100918o
Yang P, Zhai YP, Zhao X, Wang SM, Liu HL, Zhang X (2020) Effect of arbuscular mycorrhizal fungi and rhizobium inoculation on soil fungal community structure and function in the rhizosphere of Medicago sativa. Pratacult Sci 37(9):1669–1680. https://doi.org/10.11829/j.issn.1001-0629.2019-0622
Qin HS, Zhang ZL, Liang Y, **e ZJ, Huang ZQ, He MX (2020) Introduction and comprehensive evaluation of new fruit mulberry varieties, South China. Fruits 49(2):124–126+130. https://doi.org/10.13938/j.issn.1007-1431.20190362
Allison SD, Martiny JB (2008) Resistance, resilience, and redundancy and redundancy in microbial communities. Proc Natl Acad Sci 105(32):11512–11519. https://doi.org/10.1073/pnas.0801925105
Carvalho CR, Gonçalves VN, Pereira CB, Johann S, Galliza IV, Alves TMA et al (2012) The diversity, antimicrobial and anticancer activity of endophytic fungi associated with the medicinal plant Stryphnodendron adstringens (Mart.) Coville (Fabaceae) from the Brazilian savannah. Symbiosis 57(2):95–107. https://doi.org/10.1007/s13199-012-0182-2
Peng FF, Wei ZX, Li XL, Li YJ, Han GH et al (2021) Analysis of diversity and community structure of endophytes in infected sclerotia and healthy mulberry fruit by high-throughput sequencing. Food Sci 42(20):61–68. https://doi.org/10.7506/spkx1002-6630-20200729-362
Aly AH, Edrada-Ebel RA, Wray V, Müller WEG, Kozytska S, Hentschel U et al (2008) Bioactive metabolites from the endophytic fungus Ampelomyces sp. isolated from the medicinal plant Urospermum picroides. Phytochemistry 69(8):1716–1725. https://doi.org/10.1016/j.phytochem.2008.02.013
Berg G (2009) Plant–microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agricultur. Appl Microbiol Biotechnol 84(1):11–18. https://doi.org/10.1046/j.1439-037x.1999.00294.x
Kiss L, Russell JC, Szentiványi O, Xu XM, Jeffries P (2004) Biology and biocontrol potential of Ampelomyces mycoparasites, natural antagonists of powdery mildew fungi. Biocontrol Sci Technol 14(7):635–651. https://doi.org/10.1080/09583150410001683600
Jarvis WR, Slingsby K (1997) The control of powdery mildew of green-house cucumber by water sprays and Ampelomyces quisqualis. Plant Dis Report 61(9):728–730
Wagg C, Schlaeppi K, Banerjee S, Kuramae EE, van der Heijden MGA (2019) Fungal-bacterial diversity and microbiome complexity predict ecosystem functioning. Nat Commun 10(1):4841–4850. https://doi.org/10.1038/s41467-019-12798-y
Sui Z, Huang J, Yuan L (2022) Ceriporia lacerata HG2011 enhances P mobilization and wheat agronomic performance irrespective of P fertilization levels. J Appl Microbiol 133(3):1322–1332. https://doi.org/10.1111/jam.15626
Yin J, Yuan L, Huang J (2021) New functions of Ceriporia lacerata HG2011: mobilization of soil nitrogen and phosphorus and enhancement of yield and quality of ketchup-processing tomato. J Agric Food Chem 69(14):4056–4063. https://doi.org/10.1021/acs.jafc.0c06783
Mao Z, Yang P, Liu H, Mao YF, Lei Y, Hou DW et al (2022) Whole-genome sequencing and analysis of the white-rot fungus Ceriporia lacerata reveals its phylogenetic status and the genetic basis of lignocellulose degradation and terpenoid synthesis. Front Microbiol l24(13):880946. https://doi.org/10.3389/fmicb.2022.880946
Wang K, Jiang S, Pu T, Fan LM, Su FW, Ye M (2019) Antifungal activity of phenolic monoterpenes and structure-related compounds against plant pathogenic fungi. Nat Prod Res 33(10):1423–1430. https://doi.org/10.1080/14786419.2017.1419232
Bai RX, Zeng HW, Fan Q, Yin J, Sui ZM, Yuan L (2019) Effects of Ceriporia lacerata on gummy stem blight control, growth promotion and yield increase of cucumbers. Sci Agricult Sin 52(15):2604–2615. https://doi.org/10.3864/j.issn.0578-1752.2019.15.005
Qu J Ji, Zou X, Yu J P, Zhou JX, Zhou YM, Liang ZQ (2017) Re-description of two entomopathogenic fungi in Hirsutella Pat. Microbiol China 44(11): 2615−2623. https://doi.org/10.13938/j.issn.1007-1431.20190362
Acknowledgements
The author(s) would like to thank the staff of the lemon and characteristic fruit tree team of Chongqing Academy of Agricultural Sciences.
Funding
This study was funded by the Chongqing Research Institutions Performance Incentive And Guidance Special Project (CSTC2018JXJL0118); Natural Science Foundation Project of Chongqing (2023NCSQ-MSX4070); Chongqing Agricultural Development Fund—Improved Seed Innovation Project (NKY-2021AB019); Young Innovation Team Project of Chongqing Agriculture Science Academy (NKY-2019QC08); and Chongqing Research Institutions Performance Incentive And Guidance Special Project (cqaas2021.jx.j108).
Author information
Authors and Affiliations
Contributions
Guohui Han conceived the idea, Fangfang Peng and Xunlan Li wrote and drafted the paper, and the others created the figures and tables. All the authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
No potential conflicts of interest are declared by the author(s).
Consent for Publication
Not applicable.
Ethics Approval and Consent to Participate
Not applicable.
Human and Animal Rights
No human subjects or livestock were included in this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Peng, F., Li, X., Wei, Z. et al. Structure and Ecological Function of Fungal Endophytes from Stems of Different Mulberry Cultivars. Curr Microbiol 80, 401 (2023). https://doi.org/10.1007/s00284-023-03504-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03504-9