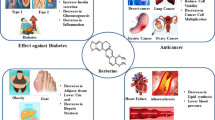
Abstract
This review aims to provide an in-depth analysis of the pharmacological properties of mangiferin, focusing primarily on its bioavailability and mechanisms of action, and its potential therapeutic applications, especially in the context of chronic diseases. We conducted a comprehensive examination of in vitro and in vivo studies, as well as clinical trials involving mangiferin or plant extracts containing mangiferin. The primary source of mangiferin is Mangifera indica, but it’s also found in other plant species from the families Anacardiaceae, Gentianaceae, and Iridaceae. Mangiferin has exhibited a myriad of therapeutic properties, presenting itself as a promising candidate for treating various chronic conditions including neurodegenerative disorders, cardiovascular diseases, renal and pulmonary diseases, diabetes, and obesity. Despite the promising results showcased in many in vitro studies and certain animal studies, the application of mangiferin has been limited due to its poor solubility, absorption, and overall bioavailability. Mangiferin offers significant therapeutic potential in treating a spectrum of chronic diseases, as evidenced by both in vitro and clinical trials. However, the challenges concerning its bioavailability necessitate further research, particularly in optimizing its delivery and absorption, to harness its full medicinal potential. This review serves as a comprehensive update on the health-promoting and therapeutic activities of mangiferin.


Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- α-SMA:
-
alpha smooth muscle actin
- A1/G1 (ABCA1/G1):
-
ATP binding cassette
- AChE:
-
acetylcholinesterase
- Ab1-40:
-
amyloid beta peptide 1–40
- Ab1-42:
-
amyloid beta peptide 1–42
- ABCG2:
-
ATP-binding cassette superfamily G member 2
- ADPS:
-
average daily pain diary score
- AKT:
-
protein kinase B
- AMPK:
-
AMP-activated protein kinase
- APAF-1:
-
apoptosis protease-activating factor-1
- APP:
-
amyloid precursor protein
- APP/PS1:
-
mouse model of Alzheimer’s disease
- AQP-2:
-
aquaporin-2
- AR-29:
-
Anoxybacillus sp. AR-29 strain
- ASC:
-
artificial cerebrospinal fluid
- ASK1:
-
apoptosis signal-regulating kinase 1
- ATP:
-
adenosine triphosphate
- β-LG:
-
β-LACTOGLOBULIN
- Bcl-2:
-
B-cell lymphoma 2
- BCS:
-
biopharmaceutics Classification System
- BDNF:
-
brain-derived neurotrophic factor
- BV-2:
-
immortalized murine microglial cell line
- CAT:
-
catalase
- CLP:
-
cecal ligation and puncture
- Col I:
-
collagen type I alpha 1
- COMT:
-
catechol-O-methyltransferase
- COX-2:
-
cyclooxygenase
- DCM:
-
diabetic cardiomyopathy
- DSS:
-
dextran sulphate sodium
- E2(PGE2):
-
prostaglandin E2
- EEG:
-
electroencephalogram
- EPM:
-
elevated plus maze
- ESI-MSn:
-
electrospray ionization mass spectroscopy
- F2a(8–150-PGF2a):
-
8–150-prostaglandin F2α
- Fas:
-
apoptosis antigen 1
- FFA:
-
free fatty acids
- FN:
-
formononetin
- FST:
-
forced swimming test
- GIT:
-
gastrointestinal regulation
- GLUT4:
-
glucose transporter 4
- GLUT9:
-
glucose transporter 9
- GPX:
-
glutathione peroxidase
- GRAS:
-
generally recognized as safe
- GSH:
-
hippocampal glutathione
- HDL:
-
high-density lipoprotein cholesterol
- HO-1:
-
hemeoxygenase-1
- HPLC:
-
high-performance liquid chromatography
- HSV-1:
-
herpes simplex virus
- IBs:
-
inflammatory bowel syndrome
- IC50:
-
inhibitory concentration
- ICAM-1:
-
intercellular adhesion molecule 1
- IκBα:
-
nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha
- IL-1b:
-
interleukin 1b
- IL-6:
-
interleukin 6
- IL-18:
-
interleukin 18
- iNOS:
-
inducible nitric oxide synthase
- IUPAC:
-
International Union of Pure and Applied Chemistry
- KATP-channels:
-
ATP-sensitive potassium channel
- KOS:
-
herpes simplex virus type 1 (HSV-1) strain KOS
- LD50:
-
lethal dose
- LDL:
-
low-density lipoprotein cholesterol
- LPS:
-
lipopolysaccharide
- LXRα:
-
liver X receptor-α
- MANG:
-
bacteroides
- MAOA:
-
monoamine oxidase A
- MAOB:
-
monoamine oxidase B
- MAP:
-
mitogen-activated protein
- MCP-1:
-
monocyte chemoattractant protein 1
- MeSH:
-
medical subject headings
- MGF:
-
mangiferin
- MMP-2:
-
matrix metallopeptidase 2
- MMP-9:
-
matrix metallopeptidase 9
- mRNA:
-
messenger ribonucleic acid
- MWM:
-
Morris water maze
- NF-kB:
-
nuclear factor kappa-light-chain-enhancer of activated B cells
- NGF:
-
nerve growth factor
- NLRP3:
-
NLR family pyrin domain containing 3
- NO:
-
nitric oxide
- NOR:
-
novel object recognition
- NOS-II:
-
nitric oxide synthase II
- Nrf-2:
-
nuclear factor erythroid 2-related factor 2
- OAT1:
-
organic anion transporter 1
- OVA:
-
ovalbumin
- p38 MAPK:
-
p38 mitogen-activated protein kinases
- PGE2:
-
prostaglandin E2
- PAT:
-
passive avoidance test
- PI3K:
-
phosphoinositide 3-kinase
- PPAR-γ:
-
peroxisome proliferator-activated receptor gamma
- PTEN:
-
fosfatidilinositol-3,4,5-trisfosfato 3-fosfatasa
- ROS:
-
reactive oxygen species
- rRNA:
-
ribosomal ribonucleic acid
- SAMP 8 :
-
senescence-accelerated mouse
- SCA-2:
-
spinocerebellar ataxia type 2
- SD:
-
sleep deprivation
- SKIP:
-
sphingosine kinase interacting protein
- Smad2/3:
-
mothers against decapentaplegic homolog 3
- SOD:
-
superoxide dismutase
- STAT:
-
signal transducer and the activator of transcription
- STZ:
-
streptozotocin
- TAU:
-
tubulin-associated unit
- tBHP:
-
t-butyl hydroperoxide
- tBid:
-
truncated bid protein
- TCM:
-
traditional Chinese medicine
- TG:
-
serum thyroglobulin
- TGF-β:
-
transforming growth factor beta
- TGF-β1:
-
transforming growth factor beta 1
- Th9:
-
T helper type 9 cells
- Th17 :
-
T helper type 17 cells
- TLR4:
-
toll-like receptor 4
- TNF-a:
-
tumor necrosis factor α
- TRIP :
-
turning research into practice
- TXNIP:
-
thioredoxin-interacting protein
- U138-MG:
-
glioblastoma cells
- URAT1:
-
solute carrier family 22 (organic anion/cation transporter), member 12S
- WOMAC:
-
Western Ontario and Mc Master Universities
References
Acevedo LM, Raya AI, Martínez-Moreno JM, Aguilera-Tejero E, Rivero JL (2017) Mangiferin protects against adverse skeletal muscle changes and enhances muscle oxidative capacity in obese rats. PLoS ONE 12:e0173028
Ain QU, Iqbal MO, Khan IA, Bano N, Naeem M, Jamaludin MI, Devaraj S (2023) Phytochemical, antioxidant, antipyretic and anti-inflammatory activities of aqueous-methanolic leaf extract of Mangifera indica. Am J Transl Res 15:4533–4543
Allaw M, Pleguezuelos-Villa M, Manca ML, Caddeo C, Aroffu M, Nacher A, Diez-Sales O, Saurí AR, Ferrer EE, Fadda AM (2020) Innovative strategies to treat skin wounds with mangiferin: fabrication of transfersomes modified with glycols and mucin. Nanomedicine 15:1671–1685
Amidon GL, Lennernäs H, Shah VP, Crison JR (1995) A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res 12:413–420
Barakat S, Nasr M, Ahmed RF, Badawy S, Mortada N (2022) Recent formulation advances of mangiferin. Rev Bras 32:871–882
Barreto JC, Trevisan MT, Hull WE, Erben G, de Brito ES, Pfundstein B, Würtele G, Spiegelhalder B, Owen RW (2008) Characterization and quantitation of polyphenolic compounds in bark, kernel, leaves, and peel of mango (Mangifera indica L.). J Agric Food Chem 56:5599–5610
Bhargava S, Shah MB (2020) Evaluation of efficacy of Bombax ceiba extract and its major constituent, mangiferin in streptozotocin (STZ)-induced diabetic rats. J Complement Integr Med 18:311–318
Bhatia HS, Candelario-Jalil E, de Oliveira AC, Olajide OA, Martínez-Sánchez G, Fiebich BL (2008) Mangiferin inhibits cyclooxygenase-2 expression and prostaglandin E2 production in activated rat microglial cells. Arch Biochem Biophys 477:253–258
Biradar SM, Joshi H, Chheda TK (2012) Neuropharmacological effect of Mangiferin on brain cholinesterase and brain biogenic amines in the management of Alzheimer’s disease. Eur J Pharmacol 683:140–147
Bulugonda RK, Kumar KA, Gangappa D, Beeda H, Philip GH, Muralidhara Rao D, Faisal SM (2017) Mangiferin from Pueraria tuberosa reduces inflammation via inactivation of NLRP3 inflammasome. Sci Rep 7:42683
Cao C, Su M, Zhou F (2017) Mangiferin inhibits hippocampal NLRP3 inflammasome and exerts antidepressant effects in a chronic mild stress mice model. Behav Pharmacol 28:356–364
Carvalho AC, Guedes MM, de Souza AL, Trevisan MT, Lima AF, Santos FA, Rao VS (2007) Gastroprotective effect of mangiferin, a xanthonoid from Mangifera indica, against gastric injury induced by ethanol and indomethacin in rodents. Planta Med 73:1372–1376
Cavalcante Morais T, Cavalcante Lopes S, Bezerra Carvalho KM, Rodrigues Arruda B, Correia de Souza FT, Salles Trevisan MT, Rao VS, Almeida Santos F (2012) Mangiferin, a natural xanthone, accelerates gastrointestinal transit in mice involving cholinergic mechanism. World J Gastroenterol 18:3207–14
Chae S, Piao MJ, Kang KA, Zhang R, Kim KC, Youn UJ, Nam KW, Lee JH, Hyun JW (2011) Inhibition of matrix metalloproteinase-1 induced by oxidative stress in human keratinocytes by mangiferin isolated from Anemarrhena asphodeloides. Biosci Biotechnol Biochem 75:2321–2325
Cheng Y, Wan S, Yao L, Lin D, Wu T, Chen Y, Zhang A, Lu C (2023) Bamboo leaf: A review of traditional medicinal property, phytochemistry, pharmacology, and purification technology. J Ethnopharmacol 306:116166
Chu C, Li M, Li J, Zhou C (2018) The protective effects of mangiferin on metabolic and organs functions in the adolescent rat model of alcohol abuse. J Funct Foods 46:90–100
Cvetković S, Nastasijević B, Mitić-Ćulafić D, Đukanović S, Tenji D, Knežević-Vukčević J, Nikolić B (2020) New insight into the antigenotoxic activity of Gentiana lutea extracts - Protective effect against food borne mutagens. Mutat Res Genet Toxicol Environ Mutagen 858–860:503251
Dimitrov M, Nikolova I, Benbasat N, Kitanov G, Danchev N (2011) Acute toxicity, antidepressive and MAO inhibitory activity of mangiferin isolated from Hypericum aucheri. Biotechnol Biotechnol Equip 25:2668–2671
Dou W, Zhang J, Ren G, Ding L, Sun A, Deng C, Wu X, Wei X, Mani S, Wang Z (2014) Mangiferin attenuates the symptoms of dextran sulfate sodium-induced colitis in mice via NF-κB and MAPK signaling inactivation. Int Immunopharmacol 23:170–178
Du Z, Fanshi F, Lai YH, Chen JR, Hao E, Deng J, Hsiao CD (2019) Mechanism of antidementia effects of mangiferin in a senescence accelerated mouse (SAMP8) model. Biosci Rep 39
Dutta T, Das T, Gopalakrishnan AV, Saha SC, Ghorai M, Nandy S, Kumar M, Radha, Ghosh A, Mukerjee N, Dey A (2023) Mangiferin: the miraculous xanthone with diverse pharmacological properties. Naunyn-Schmiedeberg’s Arch Pharmacol 396:851–863
Ediriweera MK, Tennekoon KH, Samarakoon SR (2017a) A Review on Ethnopharmacological Applications, Pharmacological Activities, and Bioactive Compounds of. Evid Based Complement Alternat Med 2017:6949835
Ediriweera MK, Tennekoon KH, Samarakoon SR (2017b) A review on ethnopharmacological applications, pharmacological activities, and bioactive compounds of Mangifera indica (Mango). Evid Based Complement Alternat Med 2017:6949835
Fabián RF, Mayra HM, Manuel ZV, Guadalupe SS, Alejandro PL, Alberto SJ (2023) Characterization of functionalized PLGA nanoparticles loaded with mangiferin and lupeol, and their effect on BEAS-2B and HepG2 cell lines. Anticancer Agents Med Chem 23:1174–1183
Feng X, Xue JH, **e KX, Liu SP, Zhong HP, Wang CC, Feng XQ (2017) Beneficial effect of mangiferin against sleep deprivation-induced neurodegeneration and memory impairment in mice. Biomed Res 0970-938X:28
Fu Y, Liu H, Song C, Zhang F, Liu Y, Wu J, Wen X, Liang C, Ma K, Li L (2015a) Mangiferin regulates cognitive deficits and heme oxygenase-1 induced by lipopolysaccharide in mice. Int Immunopharmacol 29:950–956
Fu Y, Liu H, Song C, Zhang F, Liu Y, Wu J, Wen X, Liang C, Ma K, Li L, Zhang X, Shao X, Sun Y, Du Y, Song Y (2015b) Mangiferin regulates cognitive deficits and heme oxygenase-1 induced by lipopolysaccharide in mice. Int Immunopharmacol 29:950–956
Garrido-Suárez BB, Garrido G, López-Mantecón AM, Piñeros O, Castro-Lopes JM, Delgado-Hernández R (2022) Mangifera indica effects on knee osteoarthritis pain. J Pharm Pharmacogn Res 10:239–252
GBD Colorectal Cancer Collaborators (2022) Global, regional, and national burden of colorectal cancer and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Gastroenterol Hepatol 7(7):627–647. https://doi.org/10.1016/S2468-1253(22)00044-9
GBD collaborators (2023) Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 402:203-234
Ginsburg H, Deharo E (2011) A call for using natural compounds in the development of new antimalarial treatments–an introduction. Malar J 10:51
Grauzdytė D, Pukalskas A, EL Kalamouni C, Venskutonis PR (2020) Mangiferin rich products from. Molecules 25
Guo H-W, Yun C-X, Hou G-H, Du J, Huang X, Lu Y, Keller ET, Zhang J, Deng J-G (2014) Mangiferin attenuates TH1/TH2 cytokine imbalance in an ovalbumin-induced asthmatic mouse model. PLoS ONE 9:e100394
Guo X, Cheng M, Hu P, Shi Z, Chen S, Liu H, Shi H, Xu Z, Tian X, Huang C (2018) Absorption, metabolism, and pharmacokinetics profiles of norathyriol, an Aglycone of Mangiferin, in rats by HPLC-MS/MS. J Agric Food Chem 66:12227–12235
He L, Peng X, Zhu J, Chen X, Liu H, Tang C, Dong Z, Liu F, Peng Y (2014) Mangiferin attenuate sepsis-induced acute kidney injury via antioxidant and anti-inflammatory effects. Am J Nephrol 40:441–450
Hirano R, Oo TH, Watanabe K (2010) Myanmar mango landraces reveal genetic uniqueness over common cultivars from Florida, India, and Southeast Asia. Genome 53:321–330
Hou S, Wang F, Li Y, Wang M, Sun D, Sun C (2012) Pharmacokinetic study of mangiferin in human plasma after oral administration. Food Chem 132:289–294
Hou J, Zheng D, Zhong G, Hu Y (2013) Mangiferin mitigates diabetic cardiomyopathy in streptozotocin-diabetic rats. Can J Physiol Pharmacol 91:759–763
Hou J, Zheng D, Fung G, Deng H, Chen L, Liang J, Jiang Y, Hu Y (2016) Mangiferin suppressed advanced glycation end products (AGEs) through NF-κB deactivation and displayed anti-inflammatory effects in streptozotocin and high fat diet-diabetic cardiomyopathy rats. Can J Physiol Pharmacol 94:332–340
Infante-Garcia C, Ramos-Rodriguez JJ, Delgado-Olmos I, Gamero-Carrasco C, Fernandez-Ponce MT, Casas L, Mantell C, Garcia-Alloza M (2017) Long-term mangiferin extract treatment improves central pathology and cognitive deficits in APP/PS1 mice. Mol Neurobiol 54:4696–4704
Ishimoto Y, Tanaka T, Yoshida Y, Inagi R (2018) Physiological and pathophysiological role of reactive oxygen species and reactive nitrogen species in the kidney. Clin Exp Pharmacol Physiol 45:1097–1105
Jagetia GC, Baliga MS (2005) Radioprotection by mangiferin in DBAxC57BL mice: a preliminary study. Phytomedicine 12:209–215
Jangra A, Lukhi MM, Sulakhiya K, Baruah CC, Lahkar M (2014) Protective effect of mangiferin against lipopolysaccharide-induced depressive and anxiety-like behaviour in mice. Eur J Pharmacol 740:337–345
Jangra A, Arora MK, Kisku A, Sharma S (2020) The multifaceted role of mangiferin in health and diseases: a review. Adv Tradit Med 21:619–643
Jia L, Sun P, Gao H, Shen J, Gao Y, Meng C, Fu S, Yao H, Zhang G (2019) Mangiferin attenuates bleomycin-induced pulmonary fibrosis in mice through inhibiting TLR4/p65 and TGF-β1/Smad2/3 pathway. J Pharm Pharmacol 71:1017–1028
Jung K, Lee B, Han SJ, Ryu JH, Kim DH (2009) Mangiferin ameliorates scopolamine-induced learning deficits in mice. Biol Pharm Bull 32:242–246
Jyotshna Khare P, Shanker K (2016) Mangiferin: A review of sources and interventions for biological activities. BioFactors 42:504–514
Kammalla AK, Ramasamy MK, Inampudi J, Dubey GP, Agrawal A, Kaliappan I (2015) Comparative pharmacokinetic study of mangiferin after oral administration of pure mangiferin and US patented polyherbal formulation to rats. AAPS PharmSciTech 16:250–258
Kasbe P, Jangra A, Lahkar M (2015) Mangiferin ameliorates aluminium chloride-induced cognitive dysfunction via alleviation of hippocampal oxido-nitrosative stress, proinflammatory cytokines and acetylcholinesterase level. J Trace Elem Med Biol 31:107–112
Kulkarni VM, Rathod VK (2018) Exploring the potential of Mangifera indica leaves extract versus mangiferin for therapeutic application. Agric Nat Res 52:155–161
Lasano NF, Hamid AH, Karim R, Dek MSP, Shukri R, Ramli NS (2019) Nutritional composition, anti-diabetic properties and identification of active compounds using UHPLC-ESI-orbitrap-MS/MS in mangifera odorata L. peel and seed kernel. Molecules 24
Lazarus JV, Han H, Mark HE, Alqahtani SA, Schattenberg JMJ, Soriano JB, White TM, Zelber-Sagi S, Aali A, Abbasi-Kangevari M, Abu-Gharbieh E, Abu-Shawer O, Aggarwal M, Ahinkorah BO, Ahmad A, Ahmadi A, Alahdab F, Alkhayyat M, Almustanyir S, Amu H, Anoushiravani A, Ashraf T, Atinafu BTT, Azadnajafabad S, Baghcheghi N, Bagherieh S, Bardhan M, Ramirez DFB, Bhagavathula AS, Bhala N, Bhat AN, Malak Bilalaga M, Biondi A, Bitaraf S, Bolla SR, Boustany A, Calina D, Campos-Nonato IR, Chattu VK, Chung S-C, Contreras D, Cortés S, Da’ar OB, Dadras O, Dai X, Danaei B, Desai R, Djalalinia S, Dongarwar D, Dsouza HL, Ekundayo TC, El Sayed I, Elemam NM, Elmonem MA, Elsharkawy A, Ezzikouri S, Farwati A, Farzadfar F, Fekadu G, Fischer F, GARG T, George ES, Ghamari S-H, Nour MG, Goel A, Golechha M, Goleij P, Gupta VK, Habibzadeh P, Ali AH, Hamid SS, Harapan H, Hariri S, Hashemian M, Hassan AM, Hassen MB, Hay SI, Hiraike Y, Homayounfar R, Hosseini M-S, Shabanan SH, Hoveidamanesh S, Hsieh VC-R, Huang J, Hussain S, Ibitoye SE, Ilesanmi OS, Ismail NE, Iwu CCD, Merin JL, Jamshidi E, Jayarajah U, Jayaram S, Mukesh Jeswani B, Jonas JB, Joseph A, Joshua CE, Jozwiak JJ, Kabir A, Vidya Kadashetti et al (2023) The global Fatty Liver Disease-Sustainable Development Goal country score for 195 countries and territories. Hepatology. https://doi.org/10.1097/HEP.0000000000000361
Li HW, Deng JG, Du ZC, Yan MS, Long ZX, Pham Thi PT, Yang KD (2013) Protective effects of mangiferin in subchronic developmental lead-exposed rats. Biol Trace Elem Res 152:233–42
Li J, Liu M, Yu H, Wang W, Han L, Chen Q, Ruan J, Wen S, Zhang Y, Wang T (2018a) Mangiferin improves hepatic lipid metabolism mainly through its metabolite-norathyriol by modulating SIRT-1/AMPK/SREBP-1c signaling. Front Pharmacol 9:201
Li Q-K, Li J-M, Chen S-Y (2018b) Regulating effects of mangiferin on oxidative stress response and mitochondrial pathway apoptosis in spinal cord injury model. J Hainan Med Univ 24:1–4
Li X, Yan Z, Carlström M, Tian J, Zhang X, Zhang W, Wu S, Ye F (2020a) Mangiferin ameliorates hyperuricemic nephropathy which is associated with downregulation of AQP2 and increased urinary uric acid excretion. Front Pharmacol 11:49
Li N, **ong R, He R, Liu B, Wang B, Geng Q (2021) Mangiferin mitigates Lipopolysaccharide-induced lung injury by inhibiting NLRP3 inflammasome activation. J Inflamm Res 14:2289
Li X, Yan Z, Carlström M, Tian J, Zhang X, Zhang W, Wu S, Ye F (2020b) Mangiferin ameliorates hyperuricemic nephropathy which is associated with downregulation of AQP2 and increased urinary uric acid excretion. Front Pharmacol 11
Liu H, Wang K, Tang Y, Sun Z, Jian L, Li Z, Wu B, Huang C (2011) Structure elucidation of in vivo and in vitro metabolites of mangiferin. J Pharm Biomed Anal 55:1075–1082
Liu YW, Zhu X, Yang QQ, Lu Q, Wang JY, Li HP, Wei YQ, Yin JL, Yin XX (2013) Suppression of methylglyoxal hyperactivity by mangiferin can prevent diabetes-associated cognitive decline in rats. Psychopharmacology 228:585–594
Liu C, Cong Z, Wang S, Zhang X, Song H, Xu T, Kong H, Gao P, Liu X (2023) A review of the botany, ethnopharmacology, phytochemistry, pharmacology, toxicology and quality of Anemarrhena asphodeloides Bunge. J Ethnopharmacol 302:115857
Lopes SC, da Silva AV, Arruda BR, Morais TC, Rios JB, Trevisan MT, Rao VS, Santos FA (2013) Peripheral antinociceptive action of mangiferin in mouse models of experimental pain: role of endogenous opioids, K(ATP)-channels and adenosine. Pharmacol Biochem Behav 110:19–26
Lum PT, Sekar M, Gan SH, Jeyabalan S, Bonam SR, Rani NNIM, Ku-Mahdzir KM, Seow LJ, Wu YS, Subramaniyan V, Fuloria NK, Fuloria S (2022) Therapeutic potential of mangiferin against kidney disorders and its mechanism of action: A review. Saudi J Biol Sci 29(3):1530–1542. https://doi.org/10.1016/j.sjbs.2021.11.016
Ma H, Chen H, Sun L, Tong L, Zhang T (2014) Improving permeability and oral absorption of mangiferin by phospholipid complexation. Fitoterapia 93:54–61. https://doi.org/10.1016/j.fitote.2013.10.016
Mahalanobish S, Saha S, Dutta S, Sil PC (2019) Mangiferin alleviates arsenic induced oxidative lung injury via upregulation of the Nrf2-HO1 axis. Food Chem Toxicol 126:41–55
Mahmoud-Awny M, Attia AS, Abd-Ellah MF, El-Abhar HS (2015) Mangiferin Mitigates Gastric Ulcer in Ischemia/ Reperfused Rats: Involvement of PPAR-γ, NF-κB and Nrf2/HO-1 Signaling Pathways. PLoS ONE 10:e0132497
Mao X, Liu L, Cheng L, Cheng R, Zhang L, Deng L, Sun X, Zhang Y, Sarmento B, Cui W (2019) Adhesive nanoparticles with inflammation regulation for promoting skin flap regeneration. J Control Release 297:91–101
Mei S, Ma H, Chen X (2021) Anticancer and anti-inflammatory properties of mangiferin: a review of its molecular mechanisms. Food Chem Toxicol 149:111997
Mei S, Perumal M, Battino M, Kitts DD, **ao J, Ma H, Chen X (2023) Mangiferin: a review of dietary sources, absorption, metabolism, bioavailability, and safety. Crit Rev Food Sci Nutr 63:3046–3064
Meng F, Zhang F, Meng M, Chen Q, Yang Y, Wang W, **e H, Li X, Gu W, Yu J (2023) Effects of the synbiotic composed of mangiferin and Lactobacillus reuteri 1–12 on type 2 diabetes mellitus rats. Front Microbiol 14:1158652
Mirza B, Croley CR, Ahmad M, Pumarol J, Das N, Sethi G, Bishayee A (2021) Mango (Mangifera indica L.): a magnificent plant with cancer preventive and anticancer therapeutic potential. Crit Rev Food Sci Nutr 61:2125–2151
Morais TC, Arruda BR, De Sousa Magalhães H, Trevisan MT, De Araújo Viana D, Rao VS, Santos FA (2015) Mangiferin ameliorates the intestinal inflammatory response and the impaired gastrointestinal motility in mouse model of postoperative ileus. Naunyn Schmiedebergs Arch Pharmacol 388:531–8
Morais TC, Arruda BR, De Sousamagalhães H, Trevisan MTS, De Araújo Viana D, Rao VS, Santos FA (2015) Mangiferin ameliorates the intestinal inflammatory response and the impaired gastrointestinal motility in mouse model of postoperative ileus. Naunyn-Schmiedeberg’s Arch Pharmacol 388:531–538
Morozkina SN, Nhung Vu TH, Generalova YE, Snetkov PP, Uspenskaya MV (2021) Mangiferin as new potential anti- cancer agent and mangiferin-integrated polymer systems-a novel research direction. Biomolecules 11(1):79. https://doi.org/10.3390/biom11010079
Mujawdiya PK, Kapur S (2015) Mangiferin: a potential natural molecule for management of metabolic syndrome. Int J Pharm Pharm Sci 7:9–13
Muruganandan S, Gupta S, Kataria M, Lal J, Gupta PK (2002) Mangiferin protects the streptozotocin-induced oxidative damage to cardiac and renal tissues in rats. Toxicology 176:165–173
Na L, Zhang Q, Jiang S, Du S, Zhang W, Li Y, Sun C, Niu Y (2015) Mangiferin supplementation improves serum lipid profiles in overweight patients with hyperlipidemia: a double-blind randomized controlled trial. Sci Rep 5:10344
Ngo TV, Scarlett CJ, Bowyer MC, Vuong QV (2019) Isolation and maximisation of extraction of mangiferin from the root of salacia chinensis L. Separations 6
Niu Y, Li S, Na L, Feng R, Liu L, Li Y, Sun C (2012a) Mangiferin decreases plasma free fatty acids through promoting its catabolism in liver by activation of AMPK. PLoS ONE 7:e30782
Niu Y, Lu W, Gao L, Lin H, Liu X, Li L (2012b) Reducing effect of mangiferin on serum uric acid levels in mice. Pharm Biol 50:1177–1182
Nong C, He W, Fleming D, Pan L, Huang H (2005) Capillary electrophoresis analysis of mangiferin extracted from Mangifera indica L. bark and Mangifera persiciformis C.Y. Wu et T.L. Ming leaves. J Chromatogr B Analyt Technol Biomed Life Sci 826:226–231
Ochocka R, Hering A, Stefanowicz-Hajduk J, Cal K, Barańska H (2017) The effect of mangiferin on skin: Penetration, permeation and inhibition of ECM enzymes. PLoS One 12:e0181542
Opović Z, Krstić-Milošević D, Marković M, Vidaković V, Bojović S (2021) L. from two high mountainous habitats: inter- and intrapopulation variability based on species' phytochemistry. Plants (Basel):10
Pardo Andreu GL, Maurmann N, Reolon GK, De Farias CB, Schwartsmann G, Delgado R, Roesler R (2010) Mangiferin, a naturally occurring glucoxilxanthone improves long-term object recognition memory in rats. Eur J Pharmacol 635:124–8
Pleguezuelos-Villa M, Nácher A, Hernández MJ, Ofelia Vila Buso MA, Ruiz Sauri A, Díez-Sales O (2019) Mangiferin nanoemulsions in treatment of inflammatory disorders and skin regeneration. Int J Pharm 564:299–307
Pleguezuelos-Villa M, Diez-Sales O, Manca ML, Manconi M, Sauri AR, Escribano-Ferrer E, Nácher A (2020) Mangiferin glycethosomes as a new potential adjuvant for the treatment of psoriasis. Int J Pharm 573:118844
Prado Y, Merino N, Acosta J, Herrera JA, Luque Y, Hernández I, Prado E, Garrido G, Delgado R, Rodeiro I (2015) Acute and 28-day subchronic toxicity studies of mangiferin, a glucosyl xanthone isolated from Mangifera indica L. stem bark. J Pharm Pharmacogn Res 3:13–23
PUBCHEM (2022) PubChem [Online]. Available: https://pubchem.ncbi.nlm.nih.gov/. Accessed 27 Feb 2023
Rechenchoski DZ, Agostinho KF, Faccin-Galhardi LC, Lonni AASG, da Silva JVH, de Andrade FG, Cunha AP, Ricardo NMPS, Nozawa C, Linhares REC (2020) Mangiferin: a promising natural xanthone from Mangifera indica for the control of acyclovir–resistant herpes simplex virus 1 infection. Bioorg Med Chem 28:115304
Reddeman RA, Glávits R, Endres JR, Clewell AE, Hirka G, Vértesi A, Béres E, Szakonyiné IP (2019) A toxicological evaluation of mango leaf extract (mangifera indica) containing 60% mangiferin. J Toxicol 1(2019):4763015. https://doi.org/10.1155/2019/4763015
Ren K, Li H, Zhou HF, Liang Y, Tong M, Chen L, Zheng XL, Zhao GJ (2019) Mangiferin promotes macrophage cholesterol efflux and protects against atherosclerosis by augmenting the expression of ABCA1 and ABCG1. Aging (albany NY) 11:10992–11009
Sadhukhan P, Saha S, Dutta S, Sil PC (2018) Mangiferin ameliorates cisplatin induced acute kidney injury by upregulating Nrf-2 via the activation of PI3K and exhibits synergistic anticancer activity with cisplatin. Front Pharmacol 9
Saha S, Sadhukhan P, Sinha K, Agarwal N, Sil PC (2016) Mangiferin attenuates oxidative stress induced renal cell damage through activation of PI3K induced Akt and Nrf-2 mediated signaling pathways. Biochem Biophys Rep 5:313–327
Sahu AK, Verma VK, Mutneja E, Malik S, Nag TC, Dinda AK, Arya DS, Bhatia J (2019) Mangiferin attenuates cisplatin-induced acute kidney injury in rats mediating modulation of MAPK pathway. Mol Cell Biochem 452:141–152
Sanugul K, Akao T, Li Y, Kakiuchi N, Nakamura N, Hattori M (2005) Isolation of a human intestinal bacterium that transforms mangiferin to norathyriol and inducibility of the enzyme that cleaves a C-glucosyl bond. Biol Pharm Bull 28:1672–1678
Sekar M (2015) Molecules of interest–mangiferin–a review. Ann Res Rev Biol, 307–320
Sellamuthu PS, Muniappan BP, Perumal SM, Kandasamy M (2009) Antihyperglycemic effect of Mangiferin in streptozotocin induced diabetic rats. J Health Sci 55:206–214
Singh AK, Raj V, Keshari AK, Rai A, Kumar P, Rawat A, Maity B, Kumar D, Prakash A, De A, Samanta A, Bhattacharya B, Saha S (2018) Isolated mangiferin and naringenin exert antidiabetic effect via PPAR(γ)/GLUT4 dual agonistic action with strong metabolic regulation. Chem Biol Interact 280:33–44
Somani S, Zambad S, Modi K (2016) Mangiferin attenuates DSS colitis in mice: Molecular docking and in vivo approach. Chem Biol Interact 253:18–26
Song Y, Liu W, Tang K, Zang J, Li D, Gao H (2020) Mangiferin alleviates renal interstitial fibrosis in streptozotocin-induced diabetic mice through regulating the PTEN/PI3K/Akt signaling pathway. J Diabetes Res
Sulaiman SF, Ooi KL (2012) Polyphenolic and vitamin C contents and antioxidant activities of aqueous extracts from mature-green and ripe fruit fleshes of Mangifera sp. J Agric Food Chem 60:11832–11838
Tan OJ, Loo HL, Thiagarajah G, Palanisamy UD, Sundralingam U (2021) Improving oral bioavailability of medicinal herbal compounds through lipid-based formulations - a sco** review. Phytomedicine 90:153651
Telange DR, Sohail NK, Hemke AT, Kharkar PS, Pethe AM (2021) Phospholipid complex-loaded self-assembled phytosomal soft nanoparticles: evidence of enhanced solubility, dissolution rate, ex vivo permeability, oral bioavailability, and antioxidant potential of mangiferin. Drug Deliv Transl Res 11:1056–1083
Tikhomirova LI, Gulikova AA (2021) Stock plant material of St John’s Wort (hypericum perforatum l.) based on biotechnology, pp 624
Tirichen H, Yaigoub H, Xu W, Wu C, Li R, Li Y (2021) Mitochondrial reactive oxygen species and their contribution in chronic kidney disease progression through oxidative stress. Front Physiol 12
WFO (2021) WFO The World Flora Online [Online]. Available: http://www.worldfloraonline.org/. Accessed 27 Feb 2023
Wilkinson AS, Monteith GR, Shaw PN, Lin CN, Gidley MJ, Roberts-Thomson SJ (2008) Effects of the mango components mangiferin and quercetin and the putative mangiferin metabolite norathyriol on the transactivation of peroxisome proliferator-activated receptor isoforms. J Agric Food Chem 56:3037–3042
Xu M, Zhang M, Wang D, Yang CR, Zhang YJ (2011) Phenolic compounds from the whole plants of Gentiana rhodantha (Gentianaceae). Chem Biodivers 8:1891–1900
Xu X, Chen Y, Song J, Hou F, Ma X, Liu B, Huang F (2018) Mangiferin suppresses endoplasmic reticulum stress in perivascular adipose tissue and prevents insulin resistance in the endothelium. Eur J Nutr 57:1563–1575
Xuan XY, Wang YJ, Tian H, Pi JX, Sun SZ, Zhang WL (2012) Study on prescription of self-microemulsifying drug delivery system of mangiferin phospholipid complex. Zhong Yao Cai 35:1508–1511
Yang Z, Weian C, Susu H, Hanmin W (2016) Protective effects of mangiferin on cerebral ischemia-reperfusion injury and its mechanisms. Eur J Pharmacol 771:145–151
Yue Y, Chen X, Qin J, Yao X (2009) Characterization of the mangiferin-human serum albumin complex by spectroscopic and molecular modeling approaches. J Pharm Biomed Anal 49:753–759
Yun C, Chang M, Hou G, Lan T, Yuan H, Su Z, Zhu D, Liang W, Li Q, Zhu H (2019) Mangiferin suppresses allergic asthma symptoms by decreased Th9 and Th17 responses and increased Treg response. Mol Immunol 114:233–242
Zhang H, Hou Y, Liu Y, Yu X, Li B, Cui H (2010) Determination of mangiferin in rat eyes and pharmacokinetic study in plasma after oral administration of mangiferin-hydroxypropyl-beta-cyclodextrin inclusion. J Ocul Pharmacol Ther 26:319–324
Zhang D, Han S, Zhou Y, Qi B, Wang X (2020) Therapeutic effects of mangiferin on sepsis-associated acute lung and kidney injuries via the downregulation of vascular permeability and protection of inflammatory and oxidative damages. Eur J Pharm Sci 152:105400
Zhao Y, Wang W, Wu X, Ma X, Qu R, Chen X, Liu C, Liu Y, Wang X, Yan P (2017) Mangiferin antagonizes TNF-α-mediated inflammatory reaction and protects against dermatitis in a mice model. Int Immunopharmacol 45:174–179
Zheng D, Hou J, **ao Y, Zhao Z, Chen L (2012) Cardioprotective effect of mangiferin on left ventricular remodeling in rats. Pharmacology 90:78–87
Zhou Y, Li X, Luo W, Zhu J, Zhao J, Wang M, Sang L, Chang B, Wang B (2022) Allicin in digestive system cancer: from biological effects to clinical treatment. Front Pharmacol 13:903259
Zhou H, Mao Z, Zhang X, Li R, Yin J, Xu Y (2023) Neuroprotective effect of mangiferin against Parkinson's disease through G-protein-coupled receptor-interacting protein 1 (GIT1)-mediated antioxidant defense. ACS Chem Neurosci
Zhu X, Cheng YQ, Du L, Li Y, Zhang F, Guo H, Liu YW, Yin XX (2015) Mangiferin attenuates renal fibrosis through down-regulation of osteopontin in diabetic rats. Phytother Res 29:295–302
Author information
Authors and Affiliations
Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas that is, revising or critically reviewing the article, giving final approval of the version to be published; agreeing on the journal to which the article has been submitted; and confirming to be accountable for all aspects of the work. All authors have read and agreed to the published version of the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not Applicable.
Consent for publication
Not Applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zivković, J., Kumar, K.A., Rushendran, R. et al. Pharmacological properties of mangiferin: bioavailability, mechanisms of action and clinical perspectives. Naunyn-Schmiedeberg's Arch Pharmacol 397, 763–781 (2024). https://doi.org/10.1007/s00210-023-02682-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02682-4