Search
Search Results
-
An update and overview of the literature on late inflammatory reactions (LIRs) in soft tissue fillers after SARS-CoV-2 infection and vaccination
IntroductionSoft tissue fillers are widely used and are commonly considered to be safe. Nonetheless, adverse events such as late inflammatory...

-
A pilot study of multi-antigen stimulated cell therapy-I plus camrelizumab and apatinib in patients with advanced bone and soft-tissue sarcomas
BackgroundCell-based immunotherapy shows the therapeutic potential in sarcomas, in addition to angiogenesis-targeted tyrosine kinase inhibitor (TKI)...

-
Evaluation of Analgesic Efficacy of Dexmedetomidine as an Adjuvant to Local Anaesthesia in Maxillofacial Soft Tissue Injuries: A Prospective Randomised Clinical Trial
Aims and ObjectivesDexmedetomidine is a relatively new, potent, and highly selective α2-adrenergic receptor agonist used for perioperative...

-
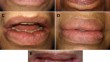
Adverse Reactions Associated with Dermal Fillers in the Oral and Maxillofacial Region: A Venezuelan Experience
BackgroundDermal facial fillers are increasingly popular. Published reports on the clinical and histopathologic characteristics related to adverse...
-
Data mining for adverse drug reaction signals of daptomycin based on real-world data: a disproportionality analysis of the US Food and Drug Administration adverse event reporting system
BackgroundPrevious reports on daptomycin’s adverse drug reactions (ADRs) have been insufficient, often because of limited data. Pharmacovigilance...

-
Data mining and analysis for emicizumab adverse event signals based on the Food and Drug Administration Adverse Event Reporting System database
BackgroundEmicizumab is the latest treatment for patients with hemophilia A. Its safety in real-world data is limited, and regulatory agencies and...
-
CACA guidelines for holistic integrative management of anticancer treatment - induced cutaneous adverse events
PurposeThe skin and mucous membrane of cancer patients can be directly or indirectly impaired during the treatment of cancers, bringing about not...
-
Adverse Events and Device Failures Associated with Pancreatic Stents: A Comprehensive Analysis Using the FDA’s MAUDE Database
IntroductionPancreatic duct stents (PDS) are widely used for the prevention of post-endoscopic retrograde cholangiopancreatography (ERCP)...

-
Uncovering the Burden of Immune-Related Adverse Events in Immunotherapy: Insights from a Nationally Representative Sample
BackgroundImmune checkpoint inhibitors have shown promising efficacy in multiple malignancies and, therefore, have been increasingly used over the...

-
Laser treatment for adverse reactions to injectable facial filling: a systematic review
Facial filling is widespread in society, albeit associated with inherent risks. This review analyzes clinical studies using laser therapy for filler...

-
Absorbable Barbed Threads for Lower Facial Soft-Tissue Repositioning in Asians
IntroductionAsian patients often seek to address lower facial aging through clinical interventions that increase anterior projection and jawline...

-
Frequency and Risk Factor Analysis for Metronidazole-Associated Neurologic Adverse Events
BackgroundLittle is known about the risk factors and frequency of metronidazole-associated neurological adverse events.
ObjectiveTo investigate the...

-
Managing Adverse Effects of Novel Therapeutic Agents in Gynecologic Malignancies
Gynecologic cancer treatments have evolved significantly. Several targeted therapies are approved for use in combination with cytotoxic therapy, as...

-
Imaging features of gluteal in vitro fertilization injection granulomas, with delayed clinical presentation simulating soft tissue sarcoma
ObjectivesTo review the clinical and imaging findings of patients with remote history of intramuscular (IM) in vitro fertilization (IVF) gluteal...

-
Outcome of revision surgery for adverse local tissue reactions in patients with recalled total hip arthroplasty
IntroductionRecalls of total hip arthroplasty (THA) implants, including metal-on-metal (MoM) THA and dual taper stems, due to increased risk of...

-
The SToICAL trial: study protocol for the soft tissue injection of corticosteroid and local anaesthetic trial—a single site, non-inferiority randomised control trial evaluating pain after soft tissue corticosteroid injections with and without local anaesthetic
BackgroundCorticosteroid injections are used in the treatment of hand and wrist conditions. The co-administration of a local anaesthetic and...

-
Head-to-head evaluation of [18F]FDG and [68 Ga]Ga-DOTA-FAPI-04 PET/CT in recurrent soft tissue sarcoma
PurposeWe aimed to evaluate the value of [ 68 Ga]Ga-DOTA-FAPI-04 PET/CT for the diagnosis of recurrent soft tissue sarcoma (STS), compared with [ 18 F]F...

-
A real-world pharmacovigilance study of abaloparatide based on the FDA Adverse Event Reporting System (FAERS)
SummaryAbaloparatide (ABL) is a US Food and Drug Administration-approved parathyroid hormone-related peptide analog for treatment of osteoporosis in...

-
Using New Autologous Tissue Filler Improved Nasolabial Folds−Single-Armed Pilot Study
ObjectiveTo evaluate the clinical efficacy and safety of the new autologous tissue filler derived from autologous skin and hairs for correction of...

