Search
Search Results
-
Practical Management of the JAK1 Inhibitor Abrocitinib for Atopic Dermatitis in Clinical Practice: Special Safety Considerations
Abrocitinib, an oral, once-daily, Janus kinase (JAK) 1-selective inhibitor, is approved for the treatment of adults and adolescents with...

-
Cost-Effectiveness Analysis of Abrocitinib Compared with Other Systemic Treatments for Severe Atopic Dermatitis in Spain
IntroductionAtopic dermatitis (AD) is a chronic, inflammatory skin disease characterized by itchy, painful, and dry skin. Despite the great number of...

-
Efficacy and safety of abrocitinib for the treatment of adolescents and adults with moderate-to-severe atopic dermatitis: update of a living systematic review and meta-analysis
BackgroundDespite extensive research on biological therapies for atopic dermatitis (AD), recent clinical trials of the Janus kinase inhibitor 1,...
-
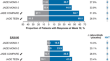
Abrocitinib 100 mg Once Daily for Moderate-to-Severe Atopic Dermatitis: A Review of Efficacy and Safety, and Expert Opinion on Use in Clinical Practice
Abrocitinib is a Janus kinase (JAK) 1-selective inhibitor approved for the treatment of moderate-to-severe atopic dermatitis (AD). Although specific...
-
Predicting Abrocitinib Efficacy at Week 12 Based on Clinical Response at Week 4: A Post Hoc Analysis of Four Randomized Studies in Moderate-to-Severe Atopic Dermatitis
IntroductionEarly prediction of abrocitinib efficacy in atopic dermatitis (AD) could help identify candidates for an early dose increase. A...

-
Integrated Safety Update of Abrocitinib in 3802 Patients with Moderate-to-Severe Atopic Dermatitis: Data from More than 5200 Patient-Years with Up to 4 Years of Exposure
BackgroundAbrocitinib, an oral, once-daily, Janus kinase 1-selective inhibitor, is efficacious in moderate-to-severe atopic dermatitis with a...

-
Abrocitinib: First Approval
Abrocitinib (Cibinqo ® ) is an oral small-molecule inhibitor of Janus kinase 1 (JAK1) being developed by Pfizer for the treatment of moderate-to-severe...

-
Efficacy and Safety of Abrocitinib in Patients with Severe and/or Difficult-to-Treat Atopic Dermatitis: A Post Hoc Analysis of the Randomized Phase 3 JADE COMPARE Trial
BackgroundTraditional systemic immunosuppressants and advanced therapies improve signs and symptoms of moderate-to-severe atopic dermatitis (AD)....

-
Durability of Response to Abrocitinib in Patients with Moderate-to-Severe Atopic Dermatitis After Treatment Discontinuation in a Phase 2b Trial
IntroductionMultiple clinical trials showed that 12 weeks of abrocitinib monotherapy was safe and effective for the treatment of moderate-to-severe...

-
Early Itch Response with Abrocitinib Is Associated with Later Efficacy Outcomes in Patients with Moderate-to-Severe Atopic Dermatitis: Subgroup Analysis of the Randomized Phase III JADE COMPARE Trial
BackgroundAbrocitinib, an oral Janus kinase 1 inhibitor, provided significant itch relief by week 2 in patients with moderate-to-severe atopic...

-
Population Pharmacokinetics of Abrocitinib in Healthy Individuals and Patients with Psoriasis or Atopic Dermatitis
Background and ObjectiveAbrocitinib is a Janus kinase 1 inhibitor in development for the treatment of atopic dermatitis (AD). This work characterized...

-
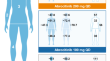
Rapidity of Improvement in Signs/Symptoms of Moderate-to-Severe Atopic Dermatitis by Body Region with Abrocitinib in the Phase 3 JADE COMPARE Study
IntroductionAtopic dermatitis (AD) can affect multiple body regions and is especially burdensome when involving exposed skin areas. Rapid, effective...
-
Integrated Safety Analysis of Abrocitinib for the Treatment of Moderate-to-Severe Atopic Dermatitis From the Phase II and Phase III Clinical Trial Program
BackgroundPivotal phase III studies demonstrated that abrocitinib, an oral, once-daily, JAK1-selective inhibitor, is effective treatment for...

-
Impact of Oral Abrocitinib Monotherapy on Patient-Reported Symptoms and Quality of Life in Adolescents and Adults with Moderate-to-Severe Atopic Dermatitis: A Pooled Analysis of Patient-Reported Outcomes
BackgroundAtopic dermatitis imparts a substantial patient burden, including itch, sleep disturbance, and decreased health-related quality of life.
...
