Abstract
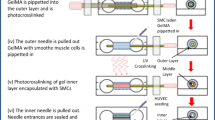
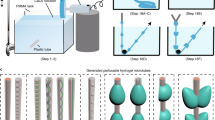
Three-dimensional bioprinting represents promising approach for fabricating standalone and perfusable vascular conduits using biocompatible materials. Here we describe a step-by-step method by using a multichannel coaxial extrusion system (MCCES) and a blend bioink constituting gelatin methacryloyl, sodium alginate, and eight-arm poly(ethylene glycol)-acrylate with a tripentaerythritol core for the fabrication of standalone circumferentially multilayered hollow tubes. This microfluidic bioprinting method allows the fabrication of perfusable vascular conduits with a core lumen, an inner endothelial layer resembling the tunica intima, and an outer smooth muscle cell layer resembling the tunica media of the blood vessel. Biocompatible and perfusable blood vessels with a widely tunable size range in terms of luminal diameters and wall thicknesses can be successfully fabricated using the MCCES.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Benjamin EJ, Muntner P, Bittencourt MSJC (2019) Heart disease and stroke statistics-2019 update: a report from the American Heart Association. Circulation 139(10):e56–e528
Pashneh-Tala S, MacNeil S, Claeyssens F (2015) The tissue-engineered vascular graft—past, present, and future. Tissue Eng Part B Rev 22:68–100
Seifu DG, Purnama A, Mequanint K, Mantovani DJNRC (2013) Small-diameter vascular tissue engineering. Nat Rev Cardiol 10(7):410
Ma Z, Kotaki M, Yong T, He W, Ramakrishna S (2005) Surface engineering of electrospun polyethylene terephthalate (PET) nanofibers towards development of a new material for blood vessel engineering. Biomaterials 26:2527–2536
Lord MS, Yu W, Cheng B, Simmons A, Poole-Warren L, Whitelock JM (2009) The modulation of platelet and endothelial cell adhesion to vascular graft materials by perlecan. Biomaterials 30:4898–4906
Taylor LM, Edwards JM, Brant B, Phinney ES, Porter JM (1987) Autogenous reversed vein bypass for lower extremity ischemia in patients with absent or inadequate greater saphenous vein. Am J Surg 153:505–510
Kannan RY, Salacinski HJ, Butler PE, Hamilton G, Seifalian AM (2005) Current status of prosthetic bypass grafts: a review. J Biomed Mater Res B Appl Biomater 74(1):570–581
Catto V, Farè S, Freddi G, Tanzi MC (2014) Vascular tissue engineering: recent advances in small diameter blood vessel regeneration. Vasc Med 2014:923030
Bordenave L, Menu P, Baquey C (2008) Developments towards tissue-engineered, small-diameter arterial substitutes. Expert Rev Med Devices 5:337–347
Dean EW, Udelsman B, Breuer CK, medicine (2012) Current advances in the translation of vascular tissue engineering to the treatment of pediatric congenital heart disease. Yale J Biol Med 85:229
Tara S, Rocco KA, Hibino N, Sugiura T, Kurobe H, Breuer CK, Shinoka T (2014) Vessel bioengineering– development of small-diameter arterial grafts. Circ J 78(1):17–19
Schöneberg J, De Lorenzi F, Theek B, Blaeser A, Rommel D, Kuehne AJC, Kießling F, Fischer H (2018) Engineering biofunctional in vitro vessel models using a multilayer bioprinting technique. Sci Rep 8(1):10430
Datta P, Ayan B, Ozbolat IT (2017) Bioprinting for vascular and vascularized tissue biofabrication. Acta Biomater 51:1–20
Christensen K, Xu C, Chai W, Zhang Z, Fu J, Huang Y (2015) Freeform inkjet printing of cellular structures with bifurcations. Biotechnol Bioeng 112:1047–1055
Bishop ES, Mostafa S, Pakvasa M, Luu HH, Lee MJ, Wolf JM, Ameer GA, He T-C, Reid RR (2017) 3-D bioprinting technologies in tissue engineering and regenerative medicine: current and future trends. Genes Dis 4:185–195
Murphy SV, Atala A (2014) 3D bioprinting of tissues and organs. Nat Biotechnol 32:773
Yu Y, Zhang Y, Martin JA, Ozbolat IT (2013) Evaluation of cell viability and functionality in vessel-like bioprintable cell-laden tubular channels. J Biomech Eng 135:091011
Zhang Y, Yu Y, Ozbolat IT (2013) Direct bioprinting of vessel-like tubular microfluidic channels. J Nanotechnol Eng Med 4:020902
Seol Y-J, Kang H-W, Lee SJ, Atala A, Yoo JJ (2014) Bioprinting technology and its applications. Eur J Cardiothorac Surg 46:342–348
**ong R, Zhang Z, Chai W, Huang Y, Chrisey DB (2015) Freeform drop-on-demand laser printing of 3D alginate and cellular constructs. Biofabrication 7:045011
Guillotin B, Ali M, Ducom A, Catros S, Keriquel V, Souquet A, Remy M, Fricain J-C, Guillemot F (2013) Laser-assisted bioprinting for tissue engineering. In: Biofabrication. Elsevier, pp 95–118
Heinrich MA, Liu W, Jimenez A, Yang J, Akpek A, Liu X, Pi Q, Mu X, Hu N, Schiffelers RM (2019) 3D bioprinting: from benches to translational applications. Small 15(23):e1805510
Maina RM, Barahona MJ, Finotti M, Lysyy T, Geibel P, D’Amico F, Mulligan D, Geibel JP (2018) Generating vascular conduits: from tissue engineering to three-dimensional bioprinting. Innov Surg Sci 3:203–213
Wang Z, Abdulla R, Parker B, Samanipour R, Ghosh S, Kim K (2015) A simple and high-resolution stereolithography-based 3D bioprinting system using visible light crosslinkable bioinks. Biofabrication 7:045009
Ma X, Qu X, Zhu W, Li Y-S, Yuan S, Zhang H, Liu J, Wang P, Lai CSE, Zanella F, Feng G-S, Sheikh F, Chien S, Chen S (2016) Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting. Proc Natl Acad Sci U S A 113:2206–2211
Pi Q, Maharjan S, Yan X, Liu X, Singh B, van Genderen AM, Robledo-Padilla F, Parra-Saldivar R, Hu N, Jia WJAM (2018) Digitally tunable microfluidic bioprinting of multilayered cannular tissues. Adv Mater 30(43):1706913
Jia W, Gungor-Ozkerim PS, Zhang YS, Yue K, Zhu K, Liu W, Pi Q, Byambaa B, Dokmeci MR, Shin SRJB (2016) Direct 3D bioprinting of perfusable vascular constructs using a blend bioink. Biomaterials 106:58–68
Kolesky DB, Homan KA, Skylar-Scott MA, Lewis JA (2016) Three-dimensional bioprinting of thick vascularized tissues. Proc Natl Acad Sci U S A 113(12):3179–3184
Jafarkhani M, Salehi Z, Aidun A, Shokrgozar MA (2019) Bioprinting in Vascularization Strategies. Iran Biomed J 23(1):9–20
Norotte C, Marga FS, Niklason LE, Forgacs G (2009) Scaffold-free vascular tissue engineering using bioprinting. Biomaterials 30(30):5910–5917
Freeman FE, Kelly DJ (2017) Tuning alginate bioink stiffness and composition for controlled growth factor delivery and to spatially direct MSC fate within bioprinted tissues. Sci Rep 7(1):17042–17042
Ooi HW, Mota C, Ten Cate AT, Calore A, Moroni L, Baker MBJB (2018) Thiol–ene alginate hydrogels as versatile bioinks for bioprinting. Biomacromolecules 19(8):3390–3400
Loessner D, Meinert C, Kaemmerer E, Martine LC, Yue K, Levett PA, Klein TJ, Melchels FPW, Khademhosseini A, Hutmacher DW (2016) Functionalization, preparation and use of cell-laden gelatin methacryloyl–based hydrogels as modular tissue culture platforms. Nat Protoc 11(4):727–746
Klotz BJ, Gawlitta D, Rosenberg AJ, Malda J, Melchels FPW (2016) Gelatin-methacryloyl hydrogels: towards biofabrication-based tissue repair. Trends Biotechnol 34(5):394–407
Yue K, Li X, Schrobback K, Sheikhi A, Annabi N, Leijten J, Zhang W, Zhang YS, Hutmacher DW, Klein TJ, Khademhosseini A (2017) Structural analysis of photocrosslinkable methacryloyl-modified protein derivatives. Biomaterials 139:163–171
Cao X, Ashfaq R, Cheng F, Maharjan S, Li J, Ying G, Hassan S, **ao H, Yue K, Zhang YS (2019) A tumor-on-a-Chip system with bioprinted blood and lymphatic vessel pair. Adv Funct Mater 29(31):1807173
Acknowledgments
This work was supported by funding from the National Institutes of Health (R00CA201603, R21EB025270, R21EB026175, R01EB028143, R01HL153857, R21EB030257), National Science Foundation (CBET-EBMS-1936105), the Brigham Research Institute, and the American Fund for Alternatives to Animal Research (AFAAR). Y.S.Z. sits on the Scientific Advisory Board of Allevi, Inc., which however, did not support or bias this work. The other authors declare no competing financial/commercial interest.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Maharjan, S., He, J.J., Lv, L., Wang, D., Zhang, Y.S. (2022). Microfluidic Coaxial Bioprinting of Hollow, Standalone, and Perfusable Vascular Conduits. In: Zhao, F., Leong, K.W. (eds) Vascular Tissue Engineering. Methods in Molecular Biology, vol 2375. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1708-3_6
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1708-3_6
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1707-6
Online ISBN: 978-1-0716-1708-3
eBook Packages: Springer Protocols