Abstract
Background
Genus Fritillaria is one among the biggest genera of family Liliaceae comprising of around 130–165 species. Fritillaria is viewed as a significant genus and a source of significant pharmaceutically active compounds utilized in conventional drugs by folklore. Fritillaria is utilized worldwide as medication and food. Different chemically dynamic components separated from genus Fritillaria, their phytochemistry with structure and pharmacology of these compounds have been extensively reviewed.
Main body
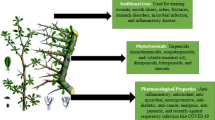
Fritillaria is utilized for treatment of dyspepsia, chest injury, tuberculosis, cough, asthma, gout, bronchitis, dysuria, sinus, boils, stomatitis, malaria, insanity, anaemia, immunity promoter, remedy for child emaciation, fever, burning sensation, phthisis and broncho-asthma, heart diseases, dysfunction of breathing and nervous system, etc. Different chemical components isolated from genus Fritillaria include around 120 alkaloids, 15 terpenoids as well as saponins, glycosides, volatile components, nucleosides, amino acids, nucleobases, flavonoids, fatty acids and so forth.
Conclusions
Many Fritillaria species have been utilized in traditional Chinese medication on account of their effects of clearing heat, moistening the lung, alleviating cough, asthma, tumours, scrofula and so on. Fritillaria is utilized for treatment of dyspepsia, chest injury, tuberculosis, cough, asthma, gout, bronchitis, dysuria, sinus, boils, stomatitis, malaria, insanity, anaemia, immunity promoter, remedy for child emaciation, also for fever, burning sensation, phthisis and broncho-asthma, heart diseases, dysfunction of breathing and nervous system, etc.
Similar content being viewed by others
Background
Genus Fritillaria L. is one among the largest genera belonging to monocot family Liliaceae comprising of around 130–165 species (Rix 2001; **beinine, 57. **beininoside, 58. ussurienine, 59. **beimunone A, 60. ussuriedine, 61. benzofluoreno[2,1-b] quinolizine cevane-3,6,16,20-tetrol, 62. **beimine C, 63. (22R,25S)- solanid-5α-enine-3β,5α,6β-triol, 64. delafrinone, 65. delafrine, 66. ningpeisine, 67. delavine 3-O-β-D-glucopyranoside, 68. persicanidine B 3-O-β-D-glucopyranoside, 69. persicanidine B, 70. (25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)-ene-6,22-dione, 71. (25R)- 22,26-epimino-3β-hydroxy-5α-cholest-22(N)-ene-6,-one 3-O-β-D-glucopyranoside, 72. (25R)- 23,26-epimino-3β-hydroxy-5a-cholest-23(N)- ene-6,22-dione3-O-β-Dglucopyranoside, 73. (20R,25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)- ene-6,22-dione3-O-β-D-glucopyranoside, 74. (20R,25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)- ene-6,22-dione3-O-β-D-glucopyranoside, 75. 15,16-seco-22αH,25βH-solanida-5,14-dien-3β-olO-β-D-glucopyranosyl-(1–4)- β-D-xylopyranoside, 76. 23-isokuroyurinidine 77. hapepunne 3-O-β-cellobioside, 78. siechuansine, 79. taipaienine, 80. puqienine A, 81. puqienine B, 82–84. puqienines C–E, 85. puqienine F, 86. N-dimethylpuqietinone 87. puqietinonoside 88. puqietinone, 89. puqietinedione, 90. 3α—puqiedin-7-ol, 91. lichuanine, 92. lichuanisinine, 93. peimisine-3-O-β-D-glucopyranoside, 94. puqiedinone-3-O-β-D-glucopyranoside, 95–97. frititorines A–C, 98. imperialinol, 99. peimisine-3-O-β-D-glucoside, 100. imperialine-3-O-β-D-glucoside, 101. delavinone, 102. hupehenizioiside
Steroidal alkaloids have been reported in Fritillaria camtschatcensis (Japanese name kuroyuri), such as anrakorinine, veraalkamine, camtschatcanidine (Kaneko et al. 1981a), hapepunine, solasodine, tomatidenol (Kaneko et al. 1981b), solanidine (1) (Mitsuhashi et al. 1969; Kaneko et al. 1981b) and kuroyurinidine (2) (Sashida et al. 1989; Mimaki and Sashida 1990a, b); solanidine (1) has additionally been reported in F. thunbergii (Kitajima et al. 1982a). From Fritillaria imperialis bulbs, ebeinone, a steroidal base, has been isolated (Sener 1994; Farooq et al. 1994), along with the structures of forticine (4) and impericine (3) that have been displayed by spectroscopic studies. Other phytochemicals extracted from F. imperialis include cevanine-type alkaloids-impericine (3) and forticine (4), three steroidal alkaloids-delavine (5), imperialine (7) and persicanidine A (6) (Akhtar et al. 2002), three steroidal bases, dihydroimpranine (8), impranine (9), fetisinine (10) and an alkaloid, korsevine (11) (Akhtar et al. 2003). In F. cirrhosa, the significant alkaloids in particular imperialine (7), verticinone (12), verticine (13), ebeiedine (14), ebeiedinone (15) and chuanbeinone (16) (Li et al. 1992; Wang et al. 2011) were identified; however, their amounts were low (Li et al. 1999). Furthermore, alkaloids like sinpeinine A (17), imperialine-3-β-glucoside (18), imperialine (7) and 3β-acetylimperialine have been reported from Bulbus Fritillaria Cirrhosae (BFC) (Zhang et al. 2003; Zhou et al. 2003; Lin et al. 2006a, b). In F. thunbergii, alkaloids like verticinone (12) and verticine (13) were found as the significant components, while low quantity of ebeiedine (14) and ebeiedinone (15) was known in this herb (Li et al. 1999). Steroidal alkaloids-dongbeinine (19), dongbeirine (20), zhebeinine (21), peimine (22), peiminine, verticine (13) and isoverticine (23) (Wu et al. 2018) have been identified from F. thunbergii Miq. var. chekiangensis (Zhang et al. 1993b). In the same context, isosteroidal alkaloids are major phytochemicals detailed of which peiminine and peimine (22) are two principle alkaloid constituents (Morimoto and Kimata 1960; Li et al. 1992; Cheng et al. 1990c). Additionally, cevanine-type alkaloid ussurienine (58), solanidine (1), verticinone (12) (Wang et al. 2015), imperialine (7), isoverticine (23) (Pae et al. 2002; Oh et al. 2003; Wang et al. 2015), **beimunone A (59) (Yang and Duan 2012), ussuriedine (60) (Kitamura et al. 1989a), benzofluoreno[2,1-b]quinolizine cevane-3,6,16,20-tetrol (61) (Kitamura et al. 1989b), ebeiedinone (15) (Lee et al. 1988), **beimine C (62) (Xu et al. 1990c, d), verticine (13) (Kaneko et al. 1980; Oh et al. 2003; Wang et al. 2015) and peimisine (43) (Oh et al. 2003; Wang et al. 2015) have been identified from Fritillaria ussuriensis (Ito et al. 1976; Kaneko et al. 1981b, Lee et al. 1988; Kitamura et al. 1988). Phytochemical examination on Fritillaria delavayi has detailed three alkaloids specifically a natural solanidanine (22R,25S)- solanid-5α-enine-3β,5α,6β-triol (63) and two cevanine-type delafrinone (64) and delafrine (65) (Kaneko et al. 1988). From Fritillaria ningguoensis, alkaloids ningpeisine (66) (N-methy1-3/3-hydroxy-saveratranine-6-one), peimine (22), peiminine, isoverticine (23) and peimisine (43) were identified (Li et al. 1988). On the other hand, from F. persica bulbs, pyrrolidine and piperidine side-chain alkaloids have been isolated (Ori et al. 1992b), alongside five cerveratrum alkaloids, namely delavine (5), persicanidine A (6) (Kaneko et al.1985; Ori et al. 1992a), persicanidine B 3-O-β-D-glucopyranoside (68), delavine 3-O-β-D-glucopyranoside (67) and persicanidine B (69) (Ori et al. 1992b) as well as five steroidal alkaloids, with structures revealed as (25R)- 22,26-epimino-3β-hydroxy-5α-cholest-22(N)-ene-6,-one 3-O-β-D-glucopyranoside (71), (25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)-ene-6,22-dione (70), (25R)- 23,26-epimino-3β-hydroxy-5a-cholest-23(N)- ene-6,22-dione3-O-β-Dglucopyranoside (72), (20R,25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)- ene-6,22-dione3-O-β-D-glucopyranoside (74) and (20R,25R)- 23,26-epimino-3β-hydroxy-5α-cholest-23(N)- ene-6,22-dione3-O-β-D-glucopyranoside (73), were reported (Ori et al. 1992c). Phytochemical examination on F. maximowiczii bulbs has reported alkaloids like 15,16-seco-22αH,25βH-solanida-5,14-dien-3β-olO-β-D-glucopyranosyl-(1–4)- β-D-xylopyranoside (75), 23-isokuroyurinidine (76), hapepunne 3-O-β-cellobioside (77) and including a jerveratrin alkaloid, kuroyurinidine (2) (Qian and Nohara 1995). In F. siechuanica found in Sichuan region of China, cevanine-type steroidal alkaloid, siechuansine (78), peimisine (43) and imperialine (7) have been identified (Wang et al. 1992a). During phytochemical analysis on Fritillaria taipaiensis L. var. ningxiaensis, Hu et al. 1993 identified C-nor-D-homo-steroidal alkaloid specifically taipaienine (79), chuanbeinone (16), imperialine (7), verticinone (12), perimissine and isovertisine. Steroidal alkaloids puqiedinone (Lin et al. 1995), puqienine A (80), puqienine B (81) (Li et al. 2006b), puqienines C–E (82–84) (Jiang et al. 2006), puqienine F (85), N-dimethylpuqietinone (86), puqietinonoside (87), puqietinone (88), (Jiang et al. 2005) puqietinedione (89), 3α—puqiedin-7-ol (90), puqiedine (8) (Jiang et al. 2006) and peimisine (43) (Wang et al. 1992) were identified from F. puqiensis (Li et al. 1990a; Lin et al. 1995). From Fritillaria roylei alkaloids peiminine, peimine (22), peimisine (43), peimiphine, peimitidine and peimidine were isolated (Singh 2006). Fritillaria lichuanensis P. Li et C.P. Yang is another species from genus Fritillaria endemic to north-west area of Hubei region, China; its phytochemical examination has revealed two new C-nor-D-homosteroidal alkaloids lichuanisinine (92) and lichuanine (91) (Pi et al. 2006a, b). Phytochemical investigation has reported steroidal alkaloids from F. unibracteata bulbs specifically peimisine (43), peimisine-3-O-β-D-glucopyranoside (93), puqiedinone-3-O-β-D-glucopyranoside (94) (Zhang et al. 2011), puqiedine (8) (Jiang et al. 2006) and puqiedinone (Lin et al. 1995). From Bulbs of Fritillaria wabuensis, imperialine (7), isoverticine (23), imperialine-β-N-oxide (44), isoverticine-β-N-oxide (45) (Wang et al. 1992, 2012) were isolated. On Fritillaria tortifolia X. Z. Duan et X. J. Zheng, phytochemical examination has reported isosteroidal alkaloids, frititorines A–C (95–97) (Hu et al.2018), imperialinol (98) (Choudhary et al. 1998), peimisine (43) (Wang et al. 1992), peimisine-3-O-β-D-glucoside (99) (Zhang et al. 2011), ebeinine (48) (Wu et al. 1989), imperialine (7) (Kaneko et al. 1985), yubeinine (32) (Zhang et al. 1993d), imperialine-3-O-β-D-glucoside (100) (Huang et al. 1990), ebeiedinone (15), delavinone (101) (Lin et al.1995) and hupehenizioiside (102) (Pi et al. 2006a, b). Compound frititorines C (97) is a jervine-type alkaloid, and imperialinol (98) is another natural cevanine-type alkaloid. Table 2 shows different alkaloids from Fritillaria along with their uses.
Terpenoids
Terpenoids have been reported as second significant and important chemical constituents in Genus Fritillaria. The structures of these terpenoids are given in Fig. 2. Ten novel diterpenoids, namely fritillebinides A, B and C, fritillebin A, fritillebin B, fritillcbin C, fritillebin D, fritillebin R, fritillebinol and fritillebic acid, have been accounted as non-basic constituents of Fritillaria ebeiensis (Wu et al. 1995), along with a kaurane diterpenes ent-3β-butanoyloxykaur-15-en-17-ol; two labdane diterpenes with structure 6-oxo-2α-hydroxy-labda-7,12(E), 14-triene (104) and 6α,7β-dihydroxy-labda-8 (17),12(E),14-triene (103) were identified from F. ebeiensis. Structure of five more kaurane diterpenes (105–109) isolated from F. ebeiensis was reported as (Xu et al. 2011a):
Structures of various terpenoids of genus Fritillaria: 103. 6α,7β-dihydroxy-labda-8 (17),12(E),14-triene, 104. 6-oxo-2α-hydroxy-labda-7,12(E), 14-triene, 105. ent-3β-butanoyloxykaur-15-en-17-ol, 106. ent-kaur-15-en-17-ol, 107. ent-kaur-15-en-3β,17-diol, 108. ent-3β-acetoxykaur-15-en-17-ol, 109. ent-kauran-16β,17-diol, 110. triterpenoid (23z)- 9,19-cycloart-23-ene-3a,25-di-ol, 111. triterpenoid 9,19-cycloart-25-ene-3b,24j-diol, 112. triterpenoids 25-hydroxyl-9,19-cycloart-22-ene-3-one, 113. cycloeucalenol, 114. bicyclic diterpenoid labdane, 115. isopimaran-19-oic acid, methyl ester, 116. ent-16β, 17-epoxy-kaurane; ent-kauran-16α, 17-diol, 117. trans-comminic acid, methyl ester, 118. ent-15β,16-epoxy-kauran-17-ol and ent-16β-hydroxy-kauran-17-yl ent-kaur-15-en-17-oate
Compound 105: ent-3β-butanoyloxykaur-15-en-17-ol;
106: ent-kaur-15-en-17-ol (Liu et al. 2007);
107: ent-kaur-15-en-3β,17-diol (Bandara and Wimalasiri 1988; Liu et al. 2007)
108: ent-3β-acetoxykaur-15-en-17-ol;
109: ent-kauran-16β,17-diol.
In Fritillaria imperialis bulbs, a tricyclic diterpene isopimara-7,15-dien-19-oic acid (Atta-ur-Rahman, 2005) has been reported. From stems and leaves of F. hupehensis, chemical constituents have been analysed as cycloartane triterpenoids; cycloeucalenol (113) (Pi et al. 2009), triterpenoid (23z)- 9,19-cycloart-23-ene-3a,25-di-ol (110) (Pi et al. 2007), triterpenoids 25-hydroxyl-9,19-cycloart-22-ene-3-one (112) and triterpenoid 9,19-cycloart-25-ene-3b,24j-diol (111) (Yu et al. 1985) have been reported. Fritillaria anhuiensis has been examined to contain a bicyclic diterpenoid labdane (114) which contains a typical sulfonyl group (Shou et al. 2009). Thirteen diterpenoids including isopimaran-19-oic acid, methyl ester (115); isopimaran-19-ol; ent-kauran-16β,17-diol (109); ent-16β, 17-epoxy-kaurane; ent-kauran-16α, 17-diol (116); trans-communol; ent-kaur-15-en-17-ol (106); trans-comminic acid, methyl ester (117); ent-15β,16-epoxy-kauran-17-ol and ent-16β-hydroxy-kauran-17-yl ent-kaur-15-en-17-oate (118) were isolated in two investigations conducted on F. thunbergii (Kitajima et al. 1982a, b, c). Table 3 shows different terpenoids in genus Fritillaria along with their uses.
Other compounds
The structures of these chemical constituents are given in Fig. 3. From the bulbs of Fritillaria imperialis, cevarin and cevacin (Chopra et al. 1956) were isolated; the volatile component of the floral bulbs of Fritillaria imperialis included 3-methyl-2-butene-1-thiol, 2-nitroethanol, 3-methylpentanol, 2,3-butanediol, acidic acid, 3-pentene-2-ol, 1-hexanol,3-hydroxy-2-butanone,cyclohexanone, 1,2-dimethyl benzene, dihydro-3-methyl 2(3H)- furanone, benzaldehyde, hexadecane, octanoic acid, acetophenone, 4,6-trichlorophenol, decanal, tetradecane, nonanoic acid, pentadecane, 2-nonene-1-ol, 3,4-dimethyl-1,5-heptadiene, (Helsper et al. 2006). From Fritillaria verticillata bulbs, β-sitosterol-3-O-glucopyranoside a glucosylsterol has been isolated as main principal component (Kim et al. 2003). From Fritillaria pallidiflora, Schrenk bulbs steroidal saponins named pallidiflosides A (119), pallidiflosides C (121) and pallidiflosides B (120) (Shen et al. 2011), pallidiflosides D (122), pallidiflosides E (123), pallidiflosides G (124), pallidiflosides H (125) and pallidiflosides I (126) (Shen et al. 2012a), spongipregnoloside A (127) (Yin et al. 2003), smilaxchinoside C (128) (Shao et al. 2007), timosaponin H1(129) (Meng et al. 1999), protobioside (130) (Geng et al. 2004), polygonatoside B3(131) (Ono et al. 2007), polyphyllin V (132) (Hou et al. 2006), deltonin (133) (Hayes et al. 2007), parispseudoside B (134) (**ao et al. 2009); (22S,25S)- 26-O-β-D-glucopyranosyl-22, 25-epoxyfurost-5-en-3β, 26-diol-3-O-[α-L-rhamnopyranosyl (1/2)]-β-D-glucopyranoside (136) (Mimaki and Sashida 1990a, b), (25R)- 26-[β-D-glucopyranosyl]oxy]-3β-[(O-α-L-rhamnopyranosyl-(1/2)- β-D-glucopyranosyl)oxy]-cholesta-5,17-diene-16,22-dione (135) (Yokosuka and Mimaki 2008); aspidistrin (138) (Koketsu et al. 1996), and 26-O-β-D-glucopyranosyl-3,26-dihydroxy-(25R)-5β-furost-12-on-20 (22)- ene-3-O-α-L rhamnopyranosyl-(1/2)- β-D-glucopyranoside (137) (Dong et al. 2001) were isolated. Different compounds were identified including three phenolic glucosides; gastrodin (139) (Yang et al. 2007); icariside D2 (141) (Miyase 1989), and 4-(β-D-glucopyranosyloxy) benzoic acid (140) (Chen et al. 2008a, b), three nucleoside compounds adenosine (143) (Stueber and Grant 2002), uracil (144) (Ellis et al. 1973) and uridine (142) (Zhou et al. 2008), twenty one amino acids (S)- β-L-phenylalanine (145) (Tian et al. 2002), arginine (146), lysine (147), tryptophan (148), tyrosine (149), histidine e(150), isoleucine (151), glycine (152), leucine (153), valine (154), oxyproline (155), alanine (156), glutamate (157), threonine (158), proline (159), methionine (160), serine (161), aspartate (162), cysteine (163), phenylalanine (164), ornithine (165), nine fatty acid: pentadecanoic acid C15:0(167), meristic acid C14:0 (166), palmitic acid C16:0(168), linoleic acid C18:2(172), oleic acid C18:1(171), palmitoleic acid C16:1(169), linolenic acid C18:3(173), stearic acid C18:0(170) (Utegenova et al. 2019), α -monopalmitin(174). One coumarin: murrayone (176), one flavonoid: diosmetin (175), one sphingolipid: 1-O-β-D-glucopyranosyl-(2S,3R,4E,8Z)-2-[(2-hydroxyoctadecanoyl) amido]-4, 8-octadecadiene-1,3-diol (177) (Jung et al. 1996) were likewise recognized in Fritillaria pallidiflora. Pharmacological and antimicrobial investigation on F. delavayi has detailed the presence of sterol, glycosides, volatile oil, saponins, reducing compounds, triterpenes, coumarins, quinones and flavonic glycosides (Maharjan et al. 2012). From Fritillaria unibracteata bulbs, five amino γ-butenolide compounds in particular fritenolide A (178), B (179), C (180), D (181), and E (182) (Liu et al. 2014), and β-sitosterol (Wei et al. 2013), 7-ketositosterol (Zhang et al. 2005a, b), 3-methoxy-4-(palmitoyloxy) benzaldehyde (Fiorentino et al. 2008) and methyl octadecanoate (Minakawa et al. 2013) were isolated. It was interesting to realize that the uncommon amide butenolide compounds were reported from thalassic lifeforms as six organic α-amino butenolides acquired from the aquatic ascomycete Leptosphaeria orae–maris (White et al. 1989), bacterium Pseudoalteromonas sp. F-420 (Yoshikawa et al. 1997), sponge Anthosigmella aft. Raromicrosclera (Tsukamoto et al. 1995), and fungus Penicillium sp. No. 13 (Kimura et al. 2000) before their identification from terrestrial plants. In Fritillaria hupehensis Hsiao and K. C. Hsia non-basic components for example, fritillebin D, fritillebin C (Wu et al. 1999), thymidine (Goebel and Schrempf 1972), adenosine (143) and uridine (142) were reported. Phytochemical examination of F. thunbergii has reported volatile components: like some aromatic ketones and aldehydes, for example, benzeneacetaldehyde (Liang et al. 2011), octadecatrienoic acid methyl esters and twenty-one elements to be specific boron, sodium, magnesium, aluminium, phosphorus, sulfur, potassium, calcium, chromium, manganese, iron, cobalt, nickel, copper, zinc, molybdenum, lead, cadmium (Yao et al. 2008) arsenic, selenium and mercury (Wu and Zheng 1997; Wang et al. 2007; Liu et al. 2008; Cai et al. 2013; 2014; Zhou et al. 2014; Lou et al..2014) were identified. Alongside some different compounds including 29 components from essential oils as δ-selinene; methyl ester; δ-elemene; pentadecanoic acid (167); tetradecanoic acid; n-hexadecanoic acid; kaurene;heptadecanoic acid; 9,12-octadecadienoic acid (Z,Z)-, methyl ester; 9-tetradecenal, (Z)-; 9-hexadecenoic acid; linoleic acid, ethyl ester; 9,12-octadecadienoic acid, oleic acid (169); methyl ester, (E, E)-; (Cao et al.2012) L-( +)- ascorbic acid 2,6-dihexadecanoate; butylated hydroxytoluene; ethyl 9-hexadecenoate; hexadecanoic acid, ethyl ester; linoleic acid, ethyl ester; 2(1H)- phenanthrenone, 3,4,4a,4b,5,6,7, kaur-16-ene; octadecanoic acid, ethyl ester; rost-4-En-3-one,17-hydroxy-,(17.β.); 3-methyleneandrostan-17-ol; and podocarp-7-en-3.β.- ol, 13.β.- methyl-13-vinyl-(Du et al. 2018), six nucleosides including guanosine, uridine (142), adenosine (143), cytidine, thymidine, and inosine (Zhang et al. 2008a, 2011, 2016). Two sterols daucosterol and β-sitosterol (Zhang et al. 1993c), two carbohydrates sucrose and β-D-glucose4-1β-D-galactose (Chen and Wang 2012), eighteen amino acids: leucine (153), glycine (152), methionine (160), histidine (150), tyrosine (149), threonine (158), isoleucine (151), alanine (156), tryptophan (148), lysine (147), cystine, aspartic acid, phenylalanine (164), valine (154), proline (159), glutamic acid, serine (161), and arginine (146) (Zhang et al. 2012), ussuriedine (60) (Kitamura et al. 1989a), benzofluoreno[2,1-b] quinolizine cevane-3,6,16,20-tetrol (61) (Kitamura et al. 1989b), ebeiedinone (15) (Lee et al. 1988), **beimine C (62) (Xu et al. 1990d, c), and verticine (13) (Kaneko et al. 1980) were identified and reported to show low AChE inhibitory actions in vitro in same species of genus Fritillaria (Yang and Duan 2012). Antitussive, expectorant and antiasthmatic effects of BFU (bulbus F. ussuriensis) are reported to be present because of its alkaloids content (Qu et al. 1990; Du 1996). Inhibition of angiotensin -I-changing enzyme action in a dose-dependent way is accounted for by in vitro investigation of verticinone (12), imperialine (7) and peimisine (43) alkaloids from BFU (Oh et al. 2003). Verticinone (12) is also studied in vitro to incite differentiation of Leukaemia HL-60 cells to granulocytes (Pae et al. 2002). In vivo investigations of BFU have detailed that its butanol and ethyl acetate extracts have action of bringing down the mean arterial pressure, rising cGMP and nitric oxide (NO) production in intact vascular tissues, and they decline angiotensin-changing enzyme and angiotensin I-influenced vasoconstriction (Kang et al. 2002). Other than, this ethanol extract is additionally known to repress the formation of MAPKs and inflammatory cytokine in mast cells (Cho et al. 2011). From Fritillaria ussuriensis bulbs, four steroidal alkaloids imperialine (7), peimisine (43), verticinone (12) and verticine (13) were extracted and identified, indicated surprising cytotoxic impacts and critical inhibitory impact is shown by peimisine (43) and verticinone (12) than the others. And all the four alkaloids show restriction of cell expansion in a time- and concentration-dependent manner (Lu et al.2004).
From F. cirrhosa and F. ussuriensis—peimisine (43), imperialine (7), verticine (13) and vericinone indicated critical cytotoxic impacts on A2780, HepG2, A549 and LLC cells (Wang et al. 2015). From Fritillaria ussuriensis and Fritillaria thunbergii known as "**-beimu" (Yang and Duan 2012) and "Zhe-beimu", respectively, isosteroidal alkaloids peimine (22) and peiminine were isolated as significant constituents of these species (Xu et al. 2016) demonstrating different pharmacological impacts (Li et al. 2006a) like antitumour (Lyu et al. 2015; Zheng et al. 2017), anti-inflammatory (Lee et al. 2015; Xu et al. 2016) antioxidant (Ruan et al. 2016) depressant and antitussive (Qian and Xu 1985) impacts. Peimine (22) are reported to repress the formation of pro-inflammatory cytokines, similar to IL-6, TNF-α and IL-8, and furthermore, in PMACI-induced HMC-1 cells, phosphorylation of MAPKs and NF-KB expression is also reduced (Park et al. 2017), ideally repressed the Kv1.3 ion channel and block the Nav1.7 ion channel (Xu et al. 2016). Peiminine is reported to protect dopaminergic neurons, inhibiting neuroinflammation and to treat Parkinson's infection (PD) and atopic dermatitis (AD) (Chen et al. 2018). From Fritillaria verticillata bulbs, β-sitosterol-3-O-glucopyranoside; a glucosylsterol has been extracted as a major functioning component, which shows sortase inhibitory effect. Sortase is a transpeptidase attaching surface protein in bacteria. The antibacterial action has been experimently demonstrated against Micrococcus luteus, Bacillus subtillis and Staphylococcus aureus bacteria (Kim et al. 2003). In gram-positive bacteria, sortase is associated with anchoring and secretion of cell wall protein (Pallen et al. 2001). From F. puqiensis steroidal alkaloids, puqiedinone and puqietinone (88) (Li et al. 1990a, ; Lin et al. 1995) were known for their antitussive and antitumour effects (Ji et al. 1993; Li et al. 1995a, b) while steroidal alkaloids of this species specifically puqienine B (81), puqietinonoside (87), N-demethylpuqietinone (126) and puqienine A (80) were assessed to show various actions as follows:
-
1)
antitussive action on cough induced by ammonia liquor in mice
-
2)
activity on human lung carcinoma A549 cell line
-
3)
activity on human stomach adenocarcinoma BGC-823 cell line
-
4)
activity on human hepatocarcinoma SMMC-7721 cell line
-
5)
against human promyelocytic leukaemia HL-60 cell line (Jiang et al. 2005).
It was additionally revealed that Fritillaria puqiensis G. D. Yu et. G. Y. Chen, a local species of china Hubie Provience, acts as substitute for Beimu with its antitussive action (Ji et al. 1993; Li et al. 1995a, b). In addition, Fritillaria puqiensis crude alkaloids content and puqietinone (88) are accounted to block the development of three varieties of tumour (hepatoma, cervical carcinoma, Ehrlich ascites carcinoma) (Li et al. 1995a, b). The total alkaloids from Fritillaria hupehensis show antiasthmatic activity and have been reported by Xu et al. 2009. From Fritillaria hupehensis bulbs, cevan-based and six penta-hexacyclic veratraman steroidal alkaloids were separated with the structure of compound 1, which was explained as 3-O-acetylverticine (55), compound 2 as (3β,5α,13α,23β)- 7,8,12,14-tetradehydro-5,6,12,13-tetrahydro-3,23-dihydroxyveratraman-6-one (52), compound 3 as 3-O-acetoxyverticinone (54), and structure of compound 4 was resolved as (3β,5α,13α,23β)- 7,8,12,14-tetradehydro-5,6,12,13-tetrahydro-3,13,23-trihydroxyveratraman-6-one (53) (Zhang et al. 2008b), alongside two common alkaloids zhebeirine (27) and ebeinine (48) which were identified and evaluated to show cytostatic action against the HepG2 (human hepatoma) and HeLa (human cervical squamous carcinoma) cell lines of which compounds 2 and 4 demonstrated huge inhibitory impacts against the both kinds of tumour cells (Zhang et al. 2008b). From F. ebeiensis compounds like ent-kaur-15-en-3B,17-diol; ent-3B-butanoyloxykaur-15-en-17-ol, ent-kaur-15-en-17-ol (106) (Bandara and Wimalasiri 1988; Liu et al. 2007), ent-3B-acetoxykaur-15-en-17-ol and ent-kauran-16B,17-diol were isolated, from which compound 3 was accounted to show a solid neuroprotective impact at low concentration than other compounds, and these compounds did not show cytotoxcity action in the absence of MPP + . These diterpenoids were hence valuable in dealing with other neurological issues like Alzheimer's disease, Huntington's disease and Parkinson's disease (Xu et al. 2011a, b). Two labdane diterpenes with structure 6-oxo-2α-hydroxy-labda-7,12(E), 14-triene (104), and 6α,7β-dihydroxy-labda-8 (17),12(E),14-triene (103) extracted from same species were reported to show neuroprotective impacts in human dopaminergic neuroblastoma SH-SY5Y neuronal cell death induced by MPP + (Xu et al. 2011a, b). From Fritillaria ebeiensis crude alkaloids, verticine (13), verticinone (12), ebeiedine (14) were reported in hepatoma in mice and Ehrlich ascites carcinoma showing strong growth inhibition, while strongest inhibition of tumours was shown by ebeiedine (14) (Li et al. 1995a, 1995b). The pharmacological investigations of the BFC (Bulbus F. cirrhosa) demonstrated alkaloids as principal constituents with expectorant, antiasthmatic and antitussive effects (Yan 2005; Chen 2008a, b). It has additionally been accounted that alkaloids like sinpeinine A (17), imperialine-3-β-glucoside (18), imperialine (7), verticinone (12) and 3β-acetylimperialine show strong antiasthmatic effect in vitro (Zhang et al. 2003; Zhou et al. 2003; Lin et al. 2006a, b). The pharmacological investigations of BFC (Bulbus Fritillaria cirrhosa)/BFW (Bulbus Fritillaria walujewii) showed crude alkaloids extracts with antitussive, antiasthmatic and expectorant actions (Yan 2005; Chen 2008a, b). Similarly, alkaloids like sinpeinine A (17), imperialine-3-β-glucoside (18), imperialine (7), verticinone (12) and 3β-acetylimperialine have been reported to have successful antiasthmatic actions (Zhang et al. 2003; Zhou et al. 2003; Lin et al. 2006a, b). Other alkaloids—imperialine (7), imperialine-β-N-oxide (44), isoverticine (23), and isoverticine-β-N-oxide (45)—were extracted from Fritillaria walujewii with their antitussive, anti-inflammatory and expectorant impacts (Wang et al. 2012) and in an investigation of alkaloids substances of Fritillaria cirrhosae bulbs, demonstrated striking antitumour action (Wang et al. 2014). Phytochemical investigation of alkaloids imperialine (7), verticinone (12) chuanbeinone (16) and verticine (13) isolated from BFC has resulted weaker expectorant impacts; however, more grounded antitussive and anti-inflammatory impacts of chuanbeinone (16), imperialine (7) than that of verticine (13), verticinone (12), which likewise showed that these alkaloids may act in a synergistic manner in the BFC (Wang et al. 2011). In Fritillaria thunbergii, isosteroidal alkaloids are significant phytochemicals revealed, of which peiminine and peimine (22) are two principle alkaloids constituents (Li et al. 1992) and as indicated by the Chinese pharmacopeia these are considered as the marker components for the quality control in China likewise demonstrating anticancer impacts by restraining the development of tumour cells (Yang et al. 2005; Li et al. 2013; Liu et al. 2015; Tong 2016). The compounds show the order peimine (22) > peiminine > ebeiedine (14) > puqietinone (88) in tracheobronchial relaxation in vitro (Chan 2000). From Fritillaria thunbergii (‘Zhe Beimu’ in Chinese), with antitussive and expectorant properties, the alkaloids-peimisine (43), peiminine and peimine (22) were isolated. They show the capacity of soothing cough, reducing sputum and furthermore indicating antioxidant action (Ruan et al. 2016). In recent investigations, it has been accounted that peiminine function as analgesic, antitussive and anti-inflammatory, helps to cure acute lung injury (Chan et al. 2000; Xu et al. 2011a, b; Guo et al. 2013), induces autophagic in cells, suppresses colorectal carcinoma cell expansion and fights cancer (Lyu et al. 2015). FTB additionally had other effects: prevent ulceration, antimuscarinic, neuroprotection, antithyroid, antidiarrheal, rheological properties and regulation of blood (Zhang et al. 1998a, 1998b; Jiang et al. 2005; Zhou et al. 2006; Suh et al. 2018; Zhang et al. 2018; Lin et al. 2010). In Fritillaria anhuiensis, a bicyclic diterpenoid labdane (114) has been reported to inhibit NO (nitric oxide) production notably (Popova et al. 2009). Phytochemical investigation has reported steroidal alkaloids from F. unibracteata bulbs, in particular puqiedinone-3-O-β-D-glucopyranoside (94), peimisine-3-O-β-D-glucopyranoside (93) (Zhang et al. 2011), peimisine (43) (Wang et al. 1992), puqiedinone (Lin et al. 1995) and puqiedine (8) (Jiang et al. 2006) indicating protective effect of PC12 cells (Zhang et al. 2011). Moreover, five amino γ-butenolides compounds specifically fritenolide A (178), C (180), B (179), E (182) and D (181) (Liu et al. 2014), and β-sitosterol (Wei et al. 2013), 7-ketositosterol (Zhang et al. 2005a, b), 3-methoxy-4-(palmitoyloxy)benzaldehyde (Fiorentino et al. 2008) and methyl octadecanoate (Minakawa et al. 2013) were isolated and reported to show cytotoxic action on human cancer cells and defensive action on injured hepatocytes in vitro in same species (Liu et al. 2014). Antimicrobial and pharmacological examination on F. delavayi has reported the presence of triterpenes, volatile oil, sterols, saponins, polyoses, coumarins, glycosides, quinones and flavonic glycosides with antimicrobial activity against bacterial pathogen Klebsiella pneumoniae (22 mm), and most noteworthy inhibitory action was seen against a fungal pathogen, Fusarium moniliforme (19 mm). These antimicrobial actions recommended possible utilization of the plant in treatment of different diseases (Maharjan et al. 2012). Steroidal saponins are known to show anti-inflammatory (Shao et al. 2007), antitumour (Furuya et al. 2001), antifungal (Zhang et al. 1993c), antithrombotic (Li et al. 2010), activities. Steroidal saponins, spongipregnoloside A (127) (Yin et al. 2003), pallidiflosides D (122), E (123), G (124), H (125) and I (126) (Shen et al. 2012a), smilaxchinoside C (128) (Shao et al. 2007), timosaponin H1(129) (Meng et al. 1999), protobioside (130) (Geng et al. 2004), polygonatoside B3(131) (Ono et al. 2007), polyphyllin V (132) (Hou et al. 2006) and deltonin (133) (Hayes et al. 2007) were extracted from the dry bulbs of F. pallidiflora some with cytotoxic action against Hela cervix cancer cell lines and human C6 brain gliomas, as steroidal saponin pallidiflosides D (122) and spirostanol saponins polygonatoside B3(131), polyphyllin V (132) and deltonin (133) showed cytotoxicity against Hela and C6 cells with compound deltonin (133) indicating the most strong cytotoxic action than other. It was likewise revealed that cytotoxic action revealed by these saponins is connected with the structures of sugar unit and aglycones (Shen et al. 2012a). Fritillaria pallidiflora found in **njiang territory, China, has been accounted to show antiasthmatic, antitussive and expectorant effects (Shen et al. 2011); the alkaloid substance of the plant was reported to show growth inhibition action on bacteria and strong antioxidant activity (Dang and Liu, 2013; Guo et al. 2013). Alkaloids were discovered to be primary constituents, isosteroidal alkaloids like imperialine-β-N-oxide (44), chuanbeinone (16), isoverticine-β-N-oxide (45) and isoverticine (23) were reported to show significant cytotoxic action against four types of tumour cell lines (A2780, LLC, A549, and HepG2) in concentration and time-dependent manner and chuanbeinone (16) was discovered more effective than other three compounds. Chuanbeinone (16) was additionally reported to alter the equilibrium of Bax/Bcl-2, induce apoptosis, reduce the development of transplantable LLC, S180 tumours in mice, seize the cell cycle in the S phase and activates caspase-3 protein (Wang et al. 2015). From Fritillaria pallidiflora and bulbs of Fritillaria ebeiensis, steroidal saponins counting pallidifloside D (122), E (123), G (124), H (125), I (126)) and kaurane diterpenes were isolated, respectively, and the previous demonstrated cytotoxic action against Hela and C6 cervix cancer cell lines (Shen et al. 2012a) and the later show neuroprotective impacts in human dopaminergic neuroblastoma SH-SY5Y neuronal cell death induced by MPP (Xu et al.2011a, b). Fritillaria pallidiflora (Yi Bei-Mu) utilized in conventional Chinese medication as antitussive, expectorant and antiasthmatic agents (Li et al. 1993; Zhou et al. 2003). Phytochemical investigation revealed fifteen isosteroidal alkaloids including ten cevane-type ones (Liu et al. 1984; Xu et al. 1990a; 1993; Xu et al. 2014), four jervine-type ones (Xu et al. 1990a, 1992) and a veratramine-type alkaloid (Shen et al. 2012b; Hao et al. 2013); cevane-type isosteroidal alkaloids in particular imperialine (7), verticine (13), ebeiedine (14) and verticinone (12) were isolated to act as the muscarinic M2-specific antagonist showing relaxant impact on smooth muscle (Eglen et al. 1992; Lin et al. 2006a, b; Kitazawa et al. 2007; Wang et al. 2012). Isosteroidal alkaloids from F. pallidiflora, yibeinones A-D (39–42) (Li et al. 2016), imperialine (7), imperialine-3β-D-glucoside (35), imperialine β-N-oxide (44) (Chen et al. 2004) and dongbeinine (19) (Zhang et al. 1993b) were isolated from which a few compounds indicated relaxation impact on tracheal preparation—compound 7 shows relaxant impact against the KCl-induced compression of isolated tracheas, while as compounds 40, 41, 42, and 7 demonstrated relaxation power on the Ach-induced tracheal preparation (Li et al. 2016). From Fritillaria tortifolia X. Z. Duan et X. J. Zheng, compound frititorines C (97) is a jervine-type alkaloid and imperialinol (98) is another natural cevanine-type alkaloid. Imperialine (7) is known to possess remarkable effect in relaxing the isolated tracheas (Li et al. 2016) with imperialinol (98).
A brief account of morphology of some of the species is given below:
-
a.
Fritillaria imperialis, commonly called crown imperial, is an impressive plant, grows about 1 m (3 ft) in height. Each bulb produces a thick, stout, upright, ramrod-straight flowering stem which bears lance-shaped, glossy leaves with wavy margins appear in whorls around the lower 1/2 of the stem. It bears a prominent whorl of 3–5 droo** or downward facing, bell-shaped orange or red flowers at the top of the stem, topped by a 'crown' of small pineapple-like tuft of leaf-like bracts. While the wild form is usually orange-red, various colours are found in cultivation, ranging from nearly a true scarlet through oranges to yellow. All parts of the plant have a skunky order (Fig. 4a).
-
b.
Fritillaria cirrhosa is a perennial herb producing bulbs up to 20 mm (0.8 in) in diameter. Stem is up to 60 cm (24 in) tall with oppositely arranged leaves 7–11 in number, sometimes 3–4 whorled and alternate. Leaves are linear to linear lance-shaped with the tip often curved or coiled. The plant bears nodded, bell-shaped, one flower at the top, sometimes flowers are in groups of one or three subtended by three leaflike bracts. Flower stalks (pedicel) shorter than tepals. Tepals are yellowish-green to brownish-purple, usually with a chequered pattern in dull purple. It is in flower from April to May. The species is hermaphrodite (has both male and female organs). Stamens are 2–3 cm long, style is three lobed, capsule is broadly oblong, 25–35 mm long, winged. Seeds are many and are pollinated by insects (Fig. 4b).
-
c.
F. pallidiflora is a perennial reaching up to 38 cm (15 in) in height, stems are thick having broadly lance-shaped bluish leaves scattered up and down the slender stem. Fritillaria pallidiflora bears 1–5 nodding greenish-yellow bell-shaped flowers 3–4 cm in length flowers are usually faintly checkered brownish red inside, pale yellow, nodding bell-shaped flowers. It is in flower from May to June. The species is hermaphrodite (has both male and female organs) and is pollinated by insect (Fig. 4c).
-
d.
Fritillaria roylei is a herbaceous plant, 0.5–2 ft tall, flowers are yellowish-green to brownish-purple and usually with a chequered pattern in dull purple. Flowers are broadly bell-shaped, hanging looking down, borne singly on the stems, but sometimes in groups of 2–4. Petals are narrow-ovate, 4–5 cm long. Leaves are linear-lancelike, often long-pointed, 5–10 cm, arranged oppositely or in whorls of 2–6 on the stem. It is in flower from April to May. The flowers are hermaphrodite (have both male and female organs) and are pollinated by insects (Fig. 4d).