Abstract
B-cell lymphoma is a group of hematological malignancies with high clinical and biological heterogeneity. The pathogenesis of B-cell lymphoma involves a complex interaction between tumor cells and the tumor microenvironment (TME), which is composed of stromal cells and extracellular matrix. Although the roles of the TME have not been fully elucidated, accumulating evidence implies that TME is closely relevant to the origination, invasion and metastasis of B-cell lymphoma. Explorations of the TME provide distinctive insights for cancer therapy. Here, we epitomize the recent advances of TME in B-cell lymphoma and discuss its function in tumor progression and immune escape. In addition, the potential clinical value of targeting TME in B-cell lymphoma is highlighted, which is expected to pave the way for novel therapeutic strategies.
Similar content being viewed by others
Introduction
Lymphomas mainly comprise Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL), representing a heterogeneous group of lymphoproliferative diseases. B-cell lymphomas account for almost 95% of all lymphoma cases [1], among which diffuse large B-cell lymphoma (DLBCL) is the most common subtype, accounting for approximately 30% of all NHL cases [2]. Patients with B-cell lymphomas are usually characterized by lymphadenopathy, extranodal disease or both and present the potential for multiple organ involvement. Therefore, early diagnosis and therapy are essential. With the development of molecular diagnosis techniques, efforts have been made to better classify B-cell lymphoma. However, due to the heterogeneity of this disease, only a few strategies are applied to routine diagnosis and prognosis prediction.
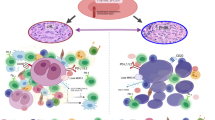
The tumor microenvironment (TME) is a complex network that comprises cellular and noncellular components, forming a physical barrier around tumor cells [3]. Accumulating studies have suggested that the TME components play important roles in the initiation and maintenance of carcinogenesis instead of being bystanders [ECM ECM represents a protein network surrounding cells, including collagens, proteoglycans, laminin and fibronectin [41]. CAFs are the main source of ECM synthesis and modification [42]. ECM is crucial for tissue homeostasis and normal organ development. Aberrant remodeling of the ECM mediated by collagen deposition or degradation could promote tumor progression. Mechanistically, the remodeled ECM performs various biological functions, including enhancing cell proliferation, increasing cell death resistance and inducing angiogenesis [43, 44]. Despite the importance of the interactions between the above cells and tumor progression, it is notable that other components of the TME could also influence the fate of tumors. Reprogrammed monocytes could accelerate tumor growth by promoting angiogenesis and remodeling the ECM. Different monocyte subsets can also differentiate into TAMs or DCs, which indirectly participate in tumor progression [45]. Immunosuppressive tumor-infiltrating DCs suppress the antitumor immunity of T cells [46]. These results indicate that the TME is an essential intrinsic portion for the regulation of tumor occurrence, development, invasion and metastasis. Thus, understanding the components of the TME involved in tumorigenesis will contribute to develo** novel therapeutic strategies. As mentioned above, the cellular and noncellular components of the TME are involved in tumor progression and the immune response, which provides novel insights for targeted therapies (Fig. 2). Therapeutic strategies are mainly divided into three categories, including depleting existing cells, preventing them from being recruited to tumor sites and reprogramming them into antitumor subtypes [47]. Several promising agents targeting the TME in B-cell lymphoma are summarized in Table 2. Several therapeutic strategies targeting TAMs in B-cell lymphoma are currently being investigated. The colony-stimulating factor-1 (CSF-1)/CSF-1 receptor (CSF-1R) signaling pathway is essential for the recruitment, polarization and functional regulation of TAMs [48]. In mantle cell lymphoma (MCL), the secretion of CSF-1 polarizes monocytes into specific CD163+ M2-like TAMs (MϕMCLs), which promotes the proliferation of lymphoma cells. It has been demonstrated that targeting CSF-1R could abrogate MϕMCL-dependent MCL survival [49]. TAMs, also known as nurse-like cells (NLCs), are correlated with the tumorigenesis of chronic lymphocytic leukemia (CLL). Pacritinib, a JAK2/FLT3 inhibitor, was proved to prevent CLL progression by depleting NLCs [50]. Recent studies have verified that CSF-1/CSF-1R blockade improves the efficacy of diverse immunotherapy modalities, such as programmed cell death 1 (PD-1) or cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) antagonists [51]. TAMs are highly dependent on the CCL2-CCR2 signaling to mobilize from the bone marrow to the site of inflammation in the TME. CCR2 inhibitors induce the accumulation of monocytes in bone marrow, resulting in reduced numbers of TAMs [51]. Yao et al. reported that CREBBP/EP300 mutations could regulate the FBXW7-NOTCH-CCL2/CSF1, polarizing TAMs to the M2 phenotype and promoting cell proliferation in DLBCL [52]. In addition, the combination of CD47 on the surface of tumor cells and SIRPα on TAMs could induce immune escape. Targeting the CD47-SIRPα axis has shown promising results in hematological malignancies [53]. It was recently demonstrated that the therapeutic effect of CD47 blockade (Hu5F9-G4) combined with rituximab has synergistic activity in an early phase clinical trial of DLBCL and follicular lymphoma (FL) [54]. MiRNAs are endogenous noncoding small RNAs participate in the occurrence and development of human malignancies. Recent studies have clarified that the specific miRNAs are involved in regulating the polarization direction and functional phenotype of TAMs. For example, miR-130, miR-33 and miR-155 can transform TAMs from the M2-like to M1-like phenotype [55, 56]. Considering the roles of MDSCs in hematological malignancies, it is reasonable to serve MDSCs as a promising target. Signal transducer and activator of transcription 3 (STAT3) and cyclooxygenase 2 (COX2)/PGE2 play a carcinogenic role in a variety of malignant tumors, which participate in the generation, maturation and accumulation of MDSCs [57]. The application of COX2 inhibitors could significantly reduce the abundance of MDSCs and block the function of MDSCs [58]. The results of a large population-based study demonstrated the survival advantages for newly diagnosed DLBCL patients who received COX2 inhibitor [59]. Emerging studies indicate that the phosphatidylinositol 3-kinase (PI3K)/AKT pathway participates in tumorigenesis by facilitating the immunosuppressive state of TME [60]. In HL, previous investigations have revealed that RP6530, a PI3Kδ/γ inhibitor, decreases the percentage of MDSCs, repolarizes TAMs to the M1-like phenotype and downregulates the expression of iNOS, thereby leading to tumor regression [61]. In addition, miRNAs could also affect the function of MDSCs. MiR-30a increases the immunosuppressive function of MDSCs by decreasing SOCS3 mRNA in B-cell lymphoma. Targeting miR-30a could reduce MDSC-mediated immunosuppressive and the number of MDSCs [62]. Li et al. reported that c-Rel, a novel immune checkpoint in MDSCs, participated in various processes, containing development, function and metabolism of MDSCs. Chen and colleagues developed R96A, a c-Rel inhibitor, which can significantly reduce the progression of lymphoma and synergistically enhance the response to anti-PD-1 antibodies [63]. Substantial studies have examined various compounds capable of modulating neutrophils [64]. Similar to the cases for TAMs, the combination of SIRPα and CD47 on TANs also mediates the immune escape. In Burkitt lymphoma (BL), KWAR23 (an anti-SIRPα antibody) was found to combine with SIRPα at high affinity and consequently increased the TANs-mediated phagocytosis of BL cells [65]. Inhibition of TANs during tumor progression serves as another effective strategy. In TME, it is implied that the CXCL12/CXCR4 axis plays a complex role in regulating the retention of TANs at inflammatory sites [66]. Emerging studies have revealed that AMD3100, an effective CXCR4 antagonist, could reverse migration and maintain the balance between bone marrow and peripheral blood, thereby inhibiting tumor growth and metastasis [67]. In addition, the CXCR2 axis is proved to be involved in the recruitment of N2 phenotype cells. DLBCL-derived IL-8 interacts with CXCR2 on TANs, forming neutrophil extracellular traps, and further activates the downstream pathway of Toll-like receptor 9 (TLR9) to boost the proliferation and migration of DLBCL cells. Nevertheless, preliminary evidence showed that deoxyribonuclease I, neutrophil elastase inhibitor and blocking CXCR2 or TLR9 could restrain the progression of DLBCL [74]. Although many strategies have been explored, clinical success is still lacking in B-cell lymphoma, which may be related to the heterogeneity and lack specific biomarkers of CAFs [75]. MMPs, enzymes targeting the ECM that cause collagen degradation, could delay the process of tissue regeneration and influence the survival, expansion and progression of tumors [76]. MMP-9, one type of MMPs, has been proved to be involved in the angiogenesis of NHL [77]. In DLBCL, the M2 TAMs could promote tumor progression by inducing cleavage of ECM via legumain [167]. Notably, before nano-immunotherapy becomes a large-scale clinical strategy, researchers need to be cautious about kee** the balance between therapeutic benefit and toxicity risks. Owing to their superparamagnetic properties and high surface-to-volume ratios, the novel nanomaterials are engineered in clinical applications by enhancing the specificity of chemotherapy and controlling the release speed of drugs [168]. The NPs are considered to be an emerging dimension of immunotherapy research. Given that there are numerous tumor-associated mutations and phenotypic variations in tumors, explorations of targeting the TME with genetic therapies and armed oncolytic viruses could promote the tumor response or restrain tumor tolerance [169]. For instance, the hyaluronidase-armed oncolytic virus could degrade the hyaluronan-rich matrix in an attempt to improve virus penetration and inhibit tumor growth in xenograft models. A phase I clinical study also supports that hyaluronidase-armed oncolytic viruses could modulate the TME more pro-inflammatory and alleviate potential toxicity and unwanted cytokine release [170]. TME–targeted therapies in combination with immunotherapies have emerged as a promising approach for cancer treatment. Modified second-generation CAR-T cells could remodel the immunosuppressive TME and revive exhausted T cells, which may further improve clinical efficacy [7].Other components of TME
Targeting the TME in B-cell lymphoma
Targeting components of the TME
Targeting TAMs
Targeting MDSCs
Targeting TANs
Targeting the ECM
Other novel technologies
Conclusions
In this review, we systematically summarize that the composition of the TME plays a vital role in various processes, including the progression, treatment, drug resistance and prognosis of B-cell lymphoma. Targeting TME components is expected to provide novel insights for the precise treatment of B-cell lymphoma. Nevertheless, there are still many unresolved issues, such as drug resistance and the feasibility of drug combination. Further studies are warranted to verify and promote the clinical applications of TME-based targeted therapy. A deep understanding of the contribution of the TME to B-cell lymphomas will help us provide patients with more feasible and effective treatment strategies.
Availability of data and materials
Not applicable.
Abbreviations
- Arg1:
-
Arginase1
- BL:
-
Burkitt lymphoma
- CAFs:
-
Cancer-associated fibroblasts
- CAR:
-
Chimeric antigen receptor
- cHL:
-
Classical Hodgkin lymphoma
- CLL:
-
Chronic lymphocytic leukemia
- COX2:
-
Cyclooxygenase 2
- CSF-1:
-
Colony-stimulating factor-1
- CSF-1R:
-
CSF-1 receptor
- CTLs:
-
Cytotoxic T lymphocytes
- CTLA-4:
-
Cytotoxic T-lymphocyte-associated antigen 4
- DCs:
-
Dendritic cells
- DLBCL:
-
Diffuse large B-cell lymphoma
- ECM:
-
Extracellular matrix
- FAP:
-
Fibroblast activation protein
- FL:
-
Follicular lymphoma
- FRβ:
-
Folate receptor β
- HIF:
-
Hypoxia inducible factor
- HL:
-
Hodgkin lymphoma
- HPSE:
-
Heparanase
- HSPG:
-
Heparan sulfate proteoglycan
- IFN:
-
Interferon
- IL:
-
Interleukin
- iNOS:
-
Inducible nitric oxide synthase
- KIRs:
-
Killer cell immunoglobulin-like receptors
- M:
-
Monocytic
- mAbs:
-
Monoclonal antibodies
- MAPK:
-
Mitogen-activated protein kinase
- MCL:
-
Mantle cell lymphoma
- MDSCs:
-
Myeloid-derived suppressor cells
- MHC-I:
-
Major histocompatibility complex class I
- MiRNAs:
-
MicroRNAs
- MMPs:
-
Matrix metalloproteinases
- NHL:
-
Non-Hodgkin lymphoma
- NK:
-
Natural killer
- NKG2A:
-
Natural killer cell group 2A
- NKG2D:
-
Natural killer cell group 2D
- NLCs:
-
Nurse-like cells
- NP:
-
Nanoparticle
- OS:
-
Overall survival
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death ligand 1
- PFS:
-
Progression-free survival
- PGE2:
-
Prostaglandin E2
- PI3K:
-
Phosphatidylinositol 3-kinase
- PMN:
-
Polymorphonuclear
- ROS:
-
Reactive oxygen species
- R/R:
-
Relapsed/refractory
- scRNA-seq:
-
Single-cell RNA sequencing
- STAT3:
-
Signal transducer and activator of transcription 3
- TAMs:
-
Tumor-associated macrophages
- TANs:
-
Tumor-associated neutrophils
- TILs:
-
Tumor infiltration lymphocytes
- TLR9:
-
Toll-like receptor 9
- TME:
-
Tumor microenvironment
- TGF-β:
-
Transforming growth factor-β
- Tregs:
-
Regulatory T cells
- VEGFA:
-
Vascular endothelial growth factor A
- VEGFR1:
-
Vascular endothelial growth factor receptor 1
References
Ennishi D, Hsi ED, Steidl C, Scott DW. Toward a new molecular taxonomy of diffuse large B-cell lymphoma. Cancer Discov. 2020;10(9):1267–81.
Sehn LH, Salles G. Diffuse large B-cell lymphoma. N Engl J Med. 2021;384(9):842–58.
Casey S, Amedei A, Aquilano K, Azmi A, Benencia F, Bhakta D, Bilsland A, Boosani C, Chen S, Ciriolo M, Crawford S, Fujii H, Georgakilas A, Guha G, Halicka D, Helferich W, Heneberg P, Honoki K, Keith W, Kerkar S, Mohammed S, Niccolai E, Nowsheen S, Vasantha Rupasinghe H, Samadi A, Singh N, Talib W, Venkateswaran V, Whelan R, Yang X, et al. Cancer prevention and therapy through the modulation of the tumor microenvironment. Semin Cancer Biol. 2015;8:S199–223.
Wang L, Ding K, Zheng C, **ao H, Liu X, Sun L, Omer R, Feng Q, Zhang Z. Detachable nanoparticle-enhanced chemoimmunotherapy based on precise killing of tumor seeds and normalizing the growing soil strategy. Nano Lett. 2020;20(9):6272–80.
Hui L, Chen Y. Tumor microenvironment: sanctuary of the devil. Cancer Lett. 2015;368(1):7–13.
Junttila M, de Sauvage F. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature. 2013;501(7467):346–54.
Bejarano L, Jordāo M, Joyce J. Therapeutic targeting of the tumor microenvironment. Cancer Discov. 2021;11(4):933–59.
Steidl C, Lee T, Shah S, Farinha P, Han G, Nayar T, Delaney A, Jones S, Iqbal J, Weisenburger D, Bast M, Rosenwald A, Muller-Hermelink H, Rimsza L, Campo E, Delabie J, Braziel R, Cook J, Tubbs R, Jaffe E, Lenz G, Connors J, Staudt L, Chan W, Gascoyne R. Tumor-associated macrophages and survival in classic Hodgkin’s lymphoma. N Engl J Med. 2010;362(10):875–85.
Taskinen M, Karjalainen-Lindsberg M, Nyman H, Eerola L, Leppä S. A high tumor-associated macrophage content predicts favorable outcome in follicular lymphoma patients treated with rituximab and cyclophosphamide-doxorubicin-vincristine-prednisone. Clin Cancer Res. 2007;13(19):5784–9.
Wu K, Lin K, Li X, Yuan X, Xu P, Ni P, Xu D. Redefining tumor-associated macrophage subpopulations and functions in the tumor microenvironment. Front Immunol. 2020;11:1731.
Marini O, Spina C, Mimiola E, Cassaro A, Malerba G, Todeschini G, Perbellini O, Scupoli M, Carli G, Facchinelli D, Cassatella M, Scapini P, Tecchio C. Identification of granulocytic myeloid-derived suppressor cells (G-MDSCs) in the peripheral blood of Hodgkin and non-Hodgkin lymphoma patients. Oncotarget. 2016;7(19):27676–88.
Azzaoui I, Uhel F, Rossille D, Pangault C, Dulong J, Le Priol J, Lamy T, Houot R, Le Gouill S, Cartron G, Godmer P, Bouabdallah K, Milpied N, Damaj G, Tarte K, Fest T, Roussel M. T-cell defect in diffuse large B-cell lymphomas involves expansion of myeloid-derived suppressor cells. Blood. 2016;128(8):1081–92.
Gabrilovich D. Myeloid-derived suppressor cells. Cancer Immunol Res. 2017;5(1):3–8.
Jaillon S, Ponzetta A, Di Mitri D, Santoni A, Bonecchi R, Mantovani A. Neutrophil diversity and plasticity in tumour progression and therapy. Nat Rev Cancer. 2020;20(9):485–503.
Manfroi B, Moreaux J, Righini C, Ghiringhelli F, Sturm N, Huard B. Tumor-associated neutrophils correlate with poor prognosis in diffuse large B-cell lymphoma patients. Blood Cancer J. 2018;8(7):66.
Marcheselli R, Bari A, Tadmor T, Marcheselli L, Cox M, Pozzi S, Ferrari A, Baldini L, Gobbi P, Aviv A, Pugliese G, Federico M, Polliack A, Sacchi S. Neutrophil-lymphocyte ratio at diagnosis is an independent prognostic factor in patients with nodular sclerosis Hodgkin lymphoma: results of a large multicenter study involving 990 patients. Hematol Oncol. 2017;35(4):561–6.
Cozar B, Greppi M, Carpentier S, Narni-Mancinelli E, Chiossone L, Vivier E. Tumor-infiltrating natural killer cells. Cancer Discov. 2020;11(1):34–44.
Klanova M, Oestergaard M, Trněný M, Hiddemann W, Marcus R, Sehn L, Vitolo U, Bazeos A, Goede V, Zeuner H, Knapp A, Sahin D, Spielewoy N, Bolen C, Cardona A, Klein C, Venstrom J, Nielsen T, Fingerle-Rowson G. Prognostic impact of natural killer cell count in follicular lymphoma and diffuse large B-cell lymphoma patients treated with immunochemotherapy. Clin Cancer Res. 2019;25(15):4634–43.
Han C, Liu T, Yin R. Biomarkers for cancer-associated fibroblasts. Biomark Res. 2020;8(1):64.
Bankov K, Doring C, Ustaszewski A, Giefing M, Herling M, Cencioni C, Spallotta F, Gaetano C, Kuppers R, Hansmann ML, Hartmann S. Fibroblasts in nodular sclerosing classical hodgkin lymphoma are defined by a specific phenotype and protect tumor cells from brentuximab-vedotin induced injury. Cancers (Basel). 2019;11(11):1687.
Haro M, Orsulic S. A paradoxical correlation of cancer-associated fibroblasts with survival outcomes in B-cell lymphomas and carcinomas. Front Cell Dev Biol. 2018;6:98.
Staiger A, Duppel J, Dengler M, van der Kuip H, Vöhringer M, Aulitzky W, Rosenwald A, Ott G, Horn H. An analysis of the role of follicular lymphoma-associated fibroblasts to promote tumor cell viability following drug-induced apoptosis. Leuk Lymphoma. 2017;58(8):1922–30.
Ciavarella S, Vegliante M, Fabbri M, De Summa S, Melle F, Motta G, De Iuliis V, Opinto G, Enjuanes A, Rega S, Gulino A, Agostinelli C, Scattone A, Tommasi S, Mangia A, Mele F, Simone G, Zito A, Ingravallo G, Vitolo U, Chiappella A, Tarella C, Gianni A, Rambaldi A, Zinzani P, Casadei B, Derenzini E, Loseto G, Pileri A, Tabanelli V, et al. Dissection of DLBCL microenvironment provides a gene expression-based predictor of survival applicable to formalin-fixed paraffin-embedded tissue. Ann Oncol. 2019;30(12):2015.
Sugimoto T, Watanabe T. Follicular lymphoma: the role of the tumor microenvironment in prognosis. J Clin Exp Hematopathol. 2016;56(1):1–19.
Janco JMT, Lamichhane P, Karyampudi L, Knutson KL. Tumor-infiltrating dendritic cells in cancer pathogenesis. J Immunol. 2015;194(7):2985–91.
Pathria P, Louis TL, Varner JA. Targeting tumor-associated macrophages in cancer. Trends Immunol. 2019;40(4):310–27.
Lin Y, Xu J, Lan H. Tumor-associated macrophages in tumor metastasis: biological roles and clinical therapeutic applications. J Hematol Oncol. 2019;12(1):76.
Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P. Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol. 2017;14(7):399–416.
Wang J, Li D, Cang H, Guo B. Crosstalk between cancer and immune cells: role of tumor-associated macrophages in the tumor microenvironment. Cancer Med. 2019;8(10):4709–21.
Zhou J, Nefedova Y, Lei A, Gabrilovich D. Neutrophils and PMN-MDSC: their biological role and interaction with stromal cells. Semin Immunol. 2018;35:19–28.
Tian X, Shen H, Li Z, Wang T, Wang S. Tumor-derived exosomes, myeloid-derived suppressor cells, and tumor microenvironment. J Hematol Oncol. 2019;12(1):84.
Ren W, Zhang X, Li W, Feng Q, Feng H, Tong Y, Rong H, Wang W, Zhang D, Zhang Z, Tu S, Zhu X, Zhang Q. Exosomal miRNA-107 induces myeloid-derived suppressor cell expansion in gastric cancer. Cancer Manag Res. 2019;11:4023–40.
Masucci M, Minopoli M, Carriero M. Tumor associated neutrophils. Their role in tumorigenesis, metastasis, prognosis and therapy. Front Oncol. 2019;9:1146.
Giese M, Hind L, Huttenlocher A. Neutrophil plasticity in the tumor microenvironment. Blood. 2019;133(20):2159–67.
Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021;18(2):85–100.
Crinier A, Narni-Mancinelli E, Ugolini S, Vivier E. SnapShot: natural killer cells. Cell. 2020;180(6):1280–1280.
** Q, Yan R, Cheng X, Wang W, Zhong Y, Hou Z, Shi Y, Wang C, Li R. Cancer-associated fibroblasts: overview, progress, challenges, and directions. Cancer Gene Ther. 2021. https://doi.org/10.1038/s41417-021-00343-3.
Sahai E, Astsaturov I, Cukierman E, DeNardo D, Egeblad M, Evans R, Fearon D, Greten F, Hingorani S, Hunter T, Hynes R, Jain R, Janowitz T, Jorgensen C, Kimmelman A, Kolonin M, Maki R, Powers R, Puré E, Ramirez D, Scherz-Shouval R, Sherman M, Stewart S, Tlsty T, Tuveson D, Watt F, Weaver V, Weeraratna A, Werb Z. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020;20(3):174–86.
De Jaeghere EA, Denys HG, De Wever O. Fibroblasts fuel immune escape in the tumor microenvironment. Trends Cancer. 2019;5(11):704–23.
Mhaidly R, Mechta-Grigoriou F. Role of cancer-associated fibroblast subpopulations in immune infiltration, as a new means of treatment in cancer. Immunol Rev. 2021;302(1):259–72.
Walker C, Mojares E, Del Río HA. Role of extracellular matrix in development and cancer progression. Int J Mol Sci. 2018;19(10):3028.
Baghban R, Roshangar L, Jahanban-Esfahlan R, Seidi K, Ebrahimi-Kalan A, Jaymand M, Kolahian S, Javaheri T, Zare P. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun Signal. 2020;18(1):59.
Bonnans C, Chou J, Werb Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786–801.
Pickup M, Mouw J, Weaver V. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15(12):1243–53.
Olingy C, Dinh H, Hedrick C. Monocyte heterogeneity and functions in cancer. J Leukoc Biol. 2019;106(2):309–22.
Tran Janco J, Lamichhane P, Karyampudi L, Knutson K. Tumor-infiltrating dendritic cells in cancer pathogenesis. J Immunol (Baltimore, Md: 1950). 2015;194(7):2985–91.
Wu T, Dai Y. Tumor microenvironment and therapeutic response. Cancer Lett. 2017;387:61–8.
Petty AJ, Yang Y. Tumor-associated macrophages in hematologic malignancies: new insights and targeted therapies. Cells. 2019;8(12):1526.
Papin A, Tessoulin B, Bellanger C, Moreau A, Le Bris Y, Maisonneuve H, Moreau P, Touzeau C, Amiot M, Pellat-Deceunynck C, Le Gouill S, Chiron D. CSF1R and BTK inhibitions as novel strategies to disrupt the dialog between mantle cell lymphoma and macrophages. Leukemia. 2019;33(10):2442–53.
Polk A, Lu Y, Wang T, Seymour E, Bailey N, Singer J, Boonstra P, Lim M, Malek S, Wilcox R. Colony-stimulating factor-1 receptor is required for nurse-like cell survival in chronic lymphocytic leukemia. Clin Cancer Res. 2016;22(24):6118–28.
DeNardo D, Ruffell B. Macrophages as regulators of tumour immunity and immunotherapy. Nat Rev Immunol. 2019;19(6):369–82.
Huang Y, Cai K, Xu P, Wang L, Huang C, Fang Y, Cheng S, Sun X, Liu F, Huang J, Ji M, Zhao W. CREBBP/EP300 mutations promoted tumor progression in diffuse large B-cell lymphoma through altering tumor-associated macrophage polarization via FBXW7-NOTCH-CCL2/CSF1 axis. Signal Transduct Target Ther. 2021;6(1):10.
Eladl E, Tremblay-LeMay R, Rastgoo N, Musani R, Chen W, Liu A, Chang H. Role of CD47 in hematological malignancies. J Hematol Oncol. 2020;13(1):96.
Advani R, Flinn I, Popplewell L, Forero A, Bartlett N, Ghosh N, Kline J, Roschewski M, LaCasce A, Collins G, Tran T, Lynn J, Chen J, Volkmer J, Agoram B, Huang J, Majeti R, Weissman I, Takimoto C, Chao M, Smith S. CD47 blockade by Hu5F9-G4 and rituximab in non-Hodgkin’s lymphoma. N Engl J Med. 2018;379(18):1711–21.
Poles W, Nishi E, de Oliveira M, Eugênio A, de Andrade T, Campos A, de Campos R, Vassallo J, Alves A, Scapulatempo Neto C, Paes R, Landman G, Zerbini M, Colleoni G. Targeting the polarization of tumor-associated macrophages and modulating mir-155 expression might be a new approach to treat diffuse large B-cell lymphoma of the elderly. Cancer Immunol Immunother CII. 2019;68(2):269–82.
Moradi-Chaleshtori M, Bandehpour M, Soudi S, Mohammadi-Yeganeh S, Hashemi SM. In vitro and in vivo evaluation of anti-tumoral effect of M1 phenotype induction in macrophages by miR-130 and miR-33 containing exosomes. Cancer Immunol Immunother. 2021;70(5):1323–39.
Liu Y, Wei G, Cheng W, Dong Z, Sun H, Lee V, Cha S, Smith D, Kwak L, Qin H. Targeting myeloid-derived suppressor cells for cancer immunotherapy. Cancer Immunol Immunother CII. 2018;67(8):1181–95.
Lv M, Wang K, Huang X. Myeloid-derived suppressor cells in hematological malignancies: friends or foes. J Hematol Oncol. 2019;12(1):105.
Smyth L, Blunt D, Gatov E, Nagamuthu C, Croxford R, Mozessohn L, Cheung M. Statin and cyclooxygenase-2 inhibitors improve survival in newly diagnosed diffuse large B-cell lymphoma: a large population-based study of 4913 subjects. Br J Haematol. 2020;191(3):396–404.
De Henau O, Rausch M, Winkler D, Campesato L, Liu C, Cymerman D, Budhu S, Ghosh A, Pink M, Tchaicha J, Douglas M, Tibbitts T, Sharma S, Proctor J, Kosmider N, White K, Stern H, Soglia J, Adams J, Palombella V, McGovern K, Kutok J, Wolchok J, Merghoub T. Overcoming resistance to checkpoint blockade therapy by targeting PI3Kγ in myeloid cells. Nature. 2016;539(7629):443–7.
Locatelli SL, Careddu G, Serio S, Consonni FM, Maeda A, Viswanadha S, Vakkalanka S, Castagna L, Santoro A, Allavena P, Sica A, Carlo-Stella C. Targeting cancer cells and tumor microenvironment in preclinical and clinical models of hodgkin lymphoma using the dual PI3Kdelta/gamma inhibitor RP6530. Clin Cancer Res. 2019;25(3):1098–112.
Xu Z, Ji J, Xu J, Li D, Shi G, Liu F, Ding L, Ren J, Dou H, Wang T, Hou Y. MiR-30a increases MDSC differentiation and immunosuppressive function by targeting SOCS3 in mice with B-cell lymphoma. FEBS J. 2017;284(15):2410–24.
Li T, Li X, Zamani A, Wang W, Lee C, Li M, Luo G, Eiler E, Sun H, Ghosh S, ** J, Murali R, Ruan Q, Shi W, Chen Y. c-Rel Is a myeloid checkpoint for cancer immunotherapy. Nat cancer. 2020;1(5):507–17.
Shaul M, Fridlender Z. Tumour-associated neutrophils in patients with cancer. Nat Rev Clin Oncol. 2019;16(10):601–20.
Ring NG, Herndler-Brandstetter D, Weiskopf K, Shan L, Volkmer JP, George BM, Lietzenmayer M, McKenna KM, Naik TJ, McCarty A, Zheng Y, Ring AM, Flavell RA, Weissman IL. Anti-SIRPalpha antibody immunotherapy enhances neutrophil and macrophage antitumor activity. Proc Natl Acad Sci U S A. 2017;114(49):E10578–85.
Isles H, Herman K, Robertson A, Loynes C, Prince L, Elks P, Renshaw S. The CXCL12/CXCR4 signaling axis retains neutrophils at inflammatory sites in Zebrafish. Front Immunol. 2019;10:1784.
Wang J, Tannous B, Poznansky M, Chen H. CXCR4 antagonist AMD3100 (plerixafor): from an impurity to a therapeutic agent. Pharmacol Res. 2020;159:105010.
Nie M, Yang L, Bi X, Wang Y, Sun P, Yang H, Liu P, Li Z, **a Y, Jiang W. Neutrophil extracellular traps induced by IL8 promote diffuse large B-cell lymphoma progression via the TLR9 signaling. Clin Cancer Res. 2019;25(6):1867–79.
McWilliams E, Mele J, Cheney C, Timmerman E, Fiazuddin F, Strattan E, Mo X, Byrd J, Muthusamy N, Awan F. Therapeutic CD94/NKG2A blockade improves natural killer cell dysfunction in chronic lymphocytic leukemia. Oncoimmunology. 2016;5(10):e1226720.
Kohrt H, Thielens A, Marabelle A, Sagiv-Barfi I, Sola C, Chanuc F, Fuseri N, Bonnafous C, Czerwinski D, Rajapaksa A, Waller E, Ugolini S, Vivier E, Romagné F, Levy R, Bléry M, André P. Anti-KIR antibody enhancement of anti-lymphoma activity of natural killer cells as monotherapy and in combination with anti-CD20 antibodies. Blood. 2014;123(5):678–86.
Lamb M, Rangarajan H, Tullius B, Lee D. Natural killer cell therapy for hematologic malignancies: successes, challenges, and the future. Stem Cell Res Ther. 2021;12(1):211.
Zhang C, Hu Y, Shi C. Targeting natural killer cells for tumor immunotherapy. Front Immunol. 2020;11:60.
Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019;18(2):99–115.
Desbois M, Wang Y. Cancer-associated fibroblasts: key players in sha** the tumor immune microenvironment. Immunol Rev. 2021;302(1):241–58.
Biffi G, Tuveson D. Diversity and biology of cancer-associated fibroblasts. Physiol Rev. 2021;101(1):147–76.
Jabłońska-Trypuć A, Matejczyk M, Rosochacki S. Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J Enzyme Inhib Med Chem. 2016;31:177–83.
Negaard H, Svennevig K, Kolset S, Iversen N, Lothe I, Østenstad B, Sandset P, Iversen P. Alterations in regulators of the extracellular matrix in non-Hodgkin lymphomas. Leuk Lymphoma. 2009;50(6):998–1004.
Shen L, Li H, Shi Y, Wang D, Gong J, Xun J, Zhou S, **ang R, Tan X. M2 tumour-associated macrophages contribute to tumour progression via legumain remodelling the extracellular matrix in diffuse large B cell lymphoma. Sci Rep. 2016;6:30347.
Kesh K, Gupta V, Durden B, Garrido V, Mateo-Victoriano B, Lavania S, Banerjee S. Therapy resistance, cancer stem cells and ECM in cancer: the matrix reloaded. Cancers. 2020;12(10):3067.
Wang Y, **ang Y, **n V, Wang X, Peng X, Liu X, Wang D, Li N, Cheng J, Lyv Y, Cui S, Ma Z, Zhang Q, **n H. Dendritic cell biology and its role in tumor immunotherapy. J Hematol Oncol. 2020;13(1):107.
Grisaru-Tal S, Itan M, Klion A, Munitz A. A new dawn for eosinophils in the tumour microenvironment. Nat Rev Cancer. 2020;20(10):594–607.
Ugel S, Cane S, De Sanctis F, Bronte V. Monocytes in the tumor microenvironment. Annu Rev Pathol. 2021;16:93–122.
Vitale I, Manic G, Coussens LM, Kroemer G, Galluzzi L. Macrophages and metabolism in the tumor microenvironment. Cell Metab. 2019;30(1):36–50.
Chiu DK, Tse AP, Xu IM, Di Cui J, Lai RK, Li LL, Koh HY, Tsang FH, Wei LL, Wong CM, Ng IO, Wong CC. Hypoxia inducible factor HIF-1 promotes myeloid-derived suppressor cells accumulation through ENTPD2/CD39L1 in hepatocellular carcinoma. Nat Commun. 2017;8(1):517.
Terrén I, Orrantia A, Vitallé J, Zenarruzabeitia O, Borrego F. NK cell metabolism and tumor microenvironment. Front Immunol. 2019;10:2278.
Semenza G. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3(10):721–32.
Yu B, Miao ZH, Jiang Y, Li MH, Yang N, Li T, Ding J. c-Jun protects hypoxia-inducible factor-1alpha from degradation via its oxygen-dependent degradation domain in a nontranscriptional manner. Cancer Res. 2009;69(19):7704–12.
Zhang H, Chen Z, Miranda RN, Medeiros LJ, McCarty N. Bifurcated BACH2 control coordinates mantle cell lymphoma survival and dispersal during hypoxia. Blood. 2017;130(6):763–76.
Bhalla K, Jaber S, Nahid MN, Underwood K, Beheshti A, Landon A, Bhandary B, Bastian P, Evens AM, Haley J, Polster B, Gartenhaus RB. Role of hypoxia in diffuse large b-cell lymphoma: metabolic repression and selective translation of HK2 facilitates development of DLBCL. Sci Rep. 2018;8(1):744.
Minoia C, Quero C, Asselti M, Galise I, Marzano AL, Iacobazzi A, Rana A, Merchionne F, Serrati S, De Tullio G, Quintana G, Casiello M, Maiorano E, Simone G, Zito FA, Iacopino P, Guarini A. Changes in angiogenesis and hypoxia-inducible factor-1alpha protein expression in relapsed/refractory indolent non-Hodgkin lymphomas. Br J Haematol. 2013;163(5):640–5.
Chen W, Hill H, Christie A, Kim M, Holloman E, Pavia-Jimenez A, Homayoun F, Ma Y, Patel N, Yell P, Hao G, Yousuf Q, Joyce A, Pedrosa I, Geiger H, Zhang H, Chang J, Gardner K, Bruick R, Reeves C, Hwang T, Courtney K, Frenkel E, Sun X, Zojwalla N, Wong T, Rizzi J, Wallace E, Josey J, **e Y, et al. Targeting renal cell carcinoma with a HIF-2 antagonist. Nature. 2016;539(7627):112–7.
Choueiri T, Kaelin W. Targeting the HIF2-VEGF axis in renal cell carcinoma. Nat Med. 2020;26(10):1519–30.
Courtney K, Ma Y, Diaz de Leon A, Christie A, **e Z, Woolford L, Singla N, Joyce A, Hill H, Madhuranthakam A, Yuan Q, ** Y, Zhang Y, Chang J, Fatunde O, Arriaga Y, Frankel A, Kalva S, Zhang S, McKenzie T, Reig Torras O, Figlin R, Rini B, McKay R, Kapur P, Wang T, Pedrosa I, Brugarolas J. HIF-2 complex dissociation, target inhibition, and acquired resistance with PT2385: a first-in-class HIF-2 inhibitor, in patients with clear cell renal cell carcinoma. Clin Cancer Res. 2020;26(4):793–803.
Vinay DS, Ryan EP, Pawelec G, Talib WH, Stagg J, Elkord E, Lichtor T, Decker WK, Whelan RL, Kumara H, Signori E, Honoki K, Georgakilas AG, Amin A, Helferich WG, Boosani CS, Guha G, Ciriolo MR, Chen S, Mohammed SI, Azmi AS, Keith WN, Bilsland A, Bhakta D, Halicka D, Fujii H, Aquilano K, Ashraf SS, Nowsheen S, Yang X, et al. Immune evasion in cancer: mechanistic basis and therapeutic strategies. Semin Cancer Biol. 2015;35:S185–98.
Zhao R, Song Y, Wang Y, Huang Y, Li Z, Cui Y, Yi M, **a L, Zhuang W, Wu X, Zhou Y. PD-1/PD-L1 blockade rescue exhausted CD8+ T cells in gastrointestinal stromal tumours via the PI3K/Akt/mTOR signalling pathway. Cell Prolif. 2019;52(3):e12571.
Mimura K, Teh J, Okayama H, Shiraishi K, Kua L, Koh V, Smoot D, Ashktorab H, Oike T, Suzuki Y, Fazreen Z, Asuncion B, Shabbir A, Yong W, So J, Soong R, Kono K. PD-L1 expression is mainly regulated by interferon gamma associated with JAK-STAT pathway in gastric cancer. Cancer Sci. 2018;109(1):43–53.
Yi M, Zhang J, Li A, Niu M, Yan Y, Jiao Y, Luo S, Zhou P, Wu K. The construction, expression, and enhanced anti-tumor activity of YM101: a bispecific antibody simultaneously targeting TGF-β and PD-L1. J Hematol Oncol. 2021;14(1):27.
Chu Y, Zhou X, Wang X. Antibody-drug conjugates for the treatment of lymphoma: clinical advances and latest progress. J Hematol Oncol. 2021;14(1):88.
Geoerger B, Kang HJ, Yalon-Oren M, Marshall LV, Vezina C, Pappo A, Laetsch TW, Petrilli AS, Ebinger M, Toporski J, Glade-Bender J, Nicholls W, Fox E, DuBois SG, Macy ME, Cohn SL, Pathiraja K, Diede SJ, Ebbinghaus S, Pinto N. Pembrolizumab in paediatric patients with advanced melanoma or a PD-L1-positive, advanced, relapsed, or refractory solid tumour or lymphoma (KEYNOTE-051): interim analysis of an open-label, single-arm, phase 1–2 trial. Lancet Oncol. 2020;21(1):121–33.
Song Y, Wu J, Chen X, Lin T, Cao J, Liu Y, Zhao Y, ** J, Huang H, Hu J, Luo J, Zhang L, Xue H, Zhang Q, Wang W, Chen C, Feng J, Zhu J. A single-arm, multicenter, phase ii study of camrelizumab in relapsed or refractory classical Hodgkin lymphoma. Clin Cancer Res. 2019;25(24):7363–9.
Nie J, Wang C, Liu Y, Yang Q, Mei Q, Dong L, Li X, Liu J, Ku W, Zhang Y, Chen M, An X, Shi L, Brock MV, Bai J, Han W. Addition of low-dose decitabine to anti-PD-1 antibody camrelizumab in relapsed/refractory classical hodgkin lymphoma. J Clin Oncol. 2019;37(17):1479–89.
Shi Y, Su H, Song Y, Jiang W, Sun X, Qian W, Zhang W, Gao Y, ** Z, Zhou J, ** C, Zou L, Qiu L, Li W, Yang J, Hou M, Zeng S, Zhang Q, Hu J, Zhou H, **ong Y, Liu P. Safety and activity of sintilimab in patients with relapsed or refractory classical Hodgkin lymphoma (ORIENT-1): a multicentre, single-arm, phase 2 trial. Lancet Haematol. 2019;6(1):e12–9.
Song Y, Gao Q, Zhang H, Fan L, Zhou J, Zou D, Li W, Yang H, Liu T, Wang Q, Lv F, Guo H, Yang L, Elstrom R, Huang J, Novotny W, Wei V, Zhu J. Treatment of relapsed or refractory classical Hodgkin lymphoma with the anti-PD-1, tislelizumab: results of a phase 2, single-arm, multicenter study. Leukemia. 2020;34(2):533–42.
Armand P, Rodig S, Melnichenko V, Thieblemont C, Bouabdallah K, Tumyan G, Ozcan M, Portino S, Fogliatto L, Caballero MD, Walewski J, Gulbas Z, Ribrag V, Christian B, Perini GF, Salles G, Svoboda J, Zain J, Patel S, Chen PH, Ligon AH, Ouyang J, Neuberg D, Redd R, Chatterjee A, Balakumaran A, Orlowski R, Shipp M, Zinzani PL. Pembrolizumab in relapsed or refractory primary mediastinal large B-cell lymphoma. J Clin Oncol. 2019;37(34):3291–9.
Ding W, LaPlant BR, Call TG, Parikh SA, Leis JF, He R, Shanafelt TD, Sinha S, Le-Rademacher J, Feldman AL, Habermann TM, Witzig TE, Wiseman GA, Lin Y, Asmus E, Nowakowski GS, Conte MJ, Bowen DA, Aitken CN, Van Dyke DL, Greipp PT, Liu X, Wu X, Zhang H, Secreto CR, Tian S, Braggio E, Wellik LE, Micallef I, Viswanatha DS, et al. Pembrolizumab in patients with CLL and Richter transformation or with relapsed CLL. Blood. 2017;129(26):3419–27.
Herrera AF, Goy A, Mehta A, Ramchandren R, Pagel JM, Svoboda J, Guan S, Hill JS, Kwei K, Liu EA, Phillips T. Safety and activity of ibrutinib in combination with durvalumab in patients with relapsed or refractory follicular lymphoma or diffuse large B-cell lymphoma. Am J Hematol. 2020;95(1):18–27.
Geoerger B, Zwaan C, Marshall L, Michon J, Bourdeaut F, Casanova M, Corradini N, Rossato G, Farid-Kapadia M, Shemesh C, Hutchinson K, Donaldson F, Liao M, Caron H, Trippett T. Atezolizumab for children and young adults with previously treated solid tumours, non-Hodgkin lymphoma, and Hodgkin lymphoma (iMATRIX): a multicentre phase 1–2 study. Lancet Oncol. 2020;21(1):134–44.
Pascual M, Mena-Varas M, Robles EF, Garcia-Barchino MJ, Panizo C, Hervas-Stubbs S, Alignani D, Sagardoy A, Martinez-Ferrandis JI, Bunting KL, Meier S, Sagaert X, Bagnara D, Guruceaga E, Blanco O, Celay J, Martinez-Baztan A, Casares N, Lasarte JJ, MacCarthy T, Melnick A, Martinez-Climent JA, Roa S. PD-1/PD-L1 immune checkpoint and p53 loss facilitate tumor progression in activated B-cell diffuse large B-cell lymphomas. Blood. 2019;133(22):2401–12.
Wu X, Gu Z, Chen Y, Chen B, Chen W, Weng L, Liu X. Application of PD-1 blockade in cancer immunotherapy. Comput Struct Biotechnol J. 2019;17:661–74.
Havel J, Chowell D, Chan T. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer. 2019;19(3):133–50.
Mitsuiki N, Schwab C, Grimbacher B. What did we learn from CTLA-4 insufficiency on the human immune system? Immunol Rev. 2019;287(1):33–49.
Rowshanravan B, Halliday N, Sansom DM. CTLA-4: a moving target in immunotherapy. Blood. 2018;131(1):58–67.
Tang F, Du X, Liu M, Zheng P, Liu Y. Anti-CTLA-4 antibodies in cancer immunotherapy: selective depletion of intratumoral regulatory T cells or checkpoint blockade? Cell Biosci. 2018;8:30.
Sharma A, Subudhi SK, Blando J, Scutti J, Vence L, Wargo J, Allison JP, Ribas A, Sharma P. Anti-CTLA-4 immunotherapy does not deplete FOXP3(+) regulatory T cells (Tregs) in human cancers. Clin Cancer Res. 2019;25(4):1233–8.
Ingram J, Blomberg O, Rashidian M, Ali L, Garforth S, Fedorov E, Fedorov A, Bonanno J, Le Gall C, Crowley S, Espinosa C, Biary T, Keliher E, Weissleder R, Almo S, Dougan S, Ploegh H, Dougan M. Anti-CTLA-4 therapy requires an Fc domain for efficacy. Proc Natl Acad Sci USA. 2018;115(15):3912–7.
Larkin J, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373(13):1270–1.
Armand P, Lesokhin A, Borrello I, Timmerman J, Gutierrez M, Zhu L, Popa McKiver M, Ansell SM. A phase 1b study of dual PD-1 and CTLA-4 or KIR blockade in patients with relapsed/refractory lymphoid malignancies. Leukemia. 2021;35(3):777–86.
Patel S, Weirather J, Lipschitz M, Lako A, Chen P, Griffin G, Armand P, Shipp M, Rodig S. The microenvironmental niche in classic Hodgkin lymphoma is enriched for CTLA-4-positive T cells that are PD-1-negative. Blood. 2019;134(23):2059–69.
Tuscano JM, Maverakis E, Groshen S, Tsao-Wei D, Luxardi G, Merleev AA, Beaven A, DiPersio JF, Popplewell L, Chen R, Kirschbaum M, Schroeder MA, Newman EM. A phase I study of the combination of rituximab and ipilimumab in patients with relapsed/refractory B-cell lymphoma. Clin Cancer Res. 2019;25(23):7004–13.
Jayaraman J, Mellody M, Hou A, Desai R, Fung A, Pham A, Chen Y, Zhao W. CAR-T design: elements and their synergistic function. EBioMedicine. 2020;58:102931.
Srivastava S, Riddell SR. Engineering CAR-T cells: design concepts. Trends Immunol. 2015;36(8):494–502.
Schuster S, Bishop M, Tam C, Waller E, Borchmann P, McGuirk J, Jäger U, Jaglowski S, Andreadis C, Westin J, Fleury I, Bachanova V, Foley S, Ho P, Mielke S, Magenau J, Holte H, Pantano S, Pacaud L, Awasthi R, Chu J, Anak Ö, Salles G, Maziarz R. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med. 2019;380(1):45–56.
Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, Lin Y, Braunschweig I, Hill BT, Timmerman JM, Deol A, Reagan PM, Stiff P, Flinn IW, Farooq U, Goy A, McSweeney PA, Munoz J, Siddiqi T, Chavez JC, Herrera AF, Bartlett NL, Wiezorek JS, Navale L, Xue A, Jiang Y, Bot A, Rossi JM, Kim JJ, Go WY, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet Oncol. 2019;20(1):31–42.
Jacobson CA, Chavez JC, Sehgal AR, William BM, Munoz J, Salles GA, Casulo C, Munshi PN, Maloney DG, De Vos S, Reshef R, Leslie LA, Yakoub-Agha I, Oluwole OO, Fung HC, Plaks V, Yang Y, Lee J, Avanzi MP, Neelapu SS. Interim analysis of ZUMA-5: A phase II study of axicabtagene ciloleucel (axi-cel) in patients (pts) with relapsed/refractory indolent non-Hodgkin lymphoma (R/R iNHL). J Clin Oncol. 2020;38(15):8008–8008.
Wang M, Munoz J, Goy A, Locke FL, Jacobson CA, Hill BT, Timmerman JM, Holmes H, Jaglowski S, Flinn IW, McSweeney PA, Miklos DB, Pagel JM, Kersten M-J, Milpied N, Fung H, Topp MS, Houot R, Beitinjaneh A, Peng W, Zheng L, Rossi JM, Jain RK, Rao AV, Reagan PM. KTE-X19 CAR T-cell therapy in relapsed or refractory mantle-cell lymphoma. N Engl J Med. 2020;382(14):1331–42.
Abramson JS, Palomba ML, Gordon LI, Lunning MA, Wang M, Arnason J, Mehta A, Purev E, Maloney DG, Andreadis C, Sehgal A, Solomon SR, Ghosh N, Albertson TM, Garcia J, Kostic A, Mallaney M, Ogasawara K, Newhall K, Kim Y, Li D, Siddiqi T. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396(10254):839–52.
Brudno J, Lam N, Vanasse D, Shen Y, Rose J, Rossi J, Xue A, Bot A, Scholler N, Mikkilineni L, Roschewski M, Dean R, Cachau R, Youkharibache P, Patel R, Hansen B, Stroncek D, Rosenberg S, Gress R, Kochenderfer J. Safety and feasibility of anti-CD19 CAR T cells with fully human binding domains in patients with B-cell lymphoma. Nat Med. 2020;26(2):270–80.
Ramos C, Grover N, Beaven A, Lulla P, Wu M, Ivanova A, Wang T, Shea T, Rooney C, Dittus C, Park S, Gee A, Eldridge P, McKay K, Mehta B, Cheng C, Buchanan F, Grilley B, Morrison K, Brenner M, Serody J, Dotti G, Heslop H, Savoldo B. Anti-CD30 CAR-T cell therapy in relapsed and refractory hodgkin lymphoma. J Clin Oncol. 2020;38(32):3794–804.
Fry T, Shah N, Orentas R, Stetler-Stevenson M, Yuan C, Ramakrishna S, Wolters P, Martin S, Delbrook C, Yates B, Shalabi H, Fountaine T, Shern J, Majzner R, Stroncek D, Sabatino M, Feng Y, Dimitrov D, Zhang L, Nguyen S, Qin H, Dropulic B, Lee D, Mackall C. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. 2018;24(1):20–8.
Lynn RC, Weber EW, Sotillo E, Gennert D, Xu P, Good Z, Anbunathan H, Lattin J, Jones R, Tieu V, Nagaraja S, Granja J, de Bourcy CFA, Majzner R, Satpathy AT, Quake SR, Monje M, Chang HY, Mackall CL. c-Jun overexpression in CAR T cells induces exhaustion resistance. Nature. 2019;576(7786):293–300.
Huang R, Li X, He Y, Zhu W, Gao L, Liu Y, Gao L, Wen Q, Zhong J, Zhang C, Zhang X. Recent advances in CAR-T cell engineering. J Hematol Oncol. 2020;13(1):86.
Amor C, Feucht J, Leibold J, Ho Y, Zhu C, Alonso-Curbelo D, Mansilla-Soto J, Boyer J, Li X, Giavridis T, Kulick A, Houlihan S, Peerschke E, Friedman S, Ponomarev V, Piersigilli A, Sadelain M, Lowe S. Senolytic CAR T cells reverse senescence-associated pathologies. Nature. 2020;583(7814):127–32.
Dana H, Chalbatani G, Jalali S, Mirzaei H, Grupp S, Suarez E, Rapôso C, Webster T. CAR-T cells: early successes in blood cancer and challenges in solid tumors. Acta Pharmaceutica Sinica B. 2021;11(5):1129–47.
Henze J, Tacke F, Hardt O, Alves F, Al Rawashdeh W. Enhancing the efficacy of CAR T cells in the tumor microenvironment of pancreatic cancer. Cancers. 2020;12(6):1389.
Rodriguez-Garcia A, Lynn R, Poussin M, Eiva M, Shaw L, O’Connor R, Minutolo N, Casado-Medrano V, Lopez G, Matsuyama T, Powell D. CAR-T cell-mediated depletion of immunosuppressive tumor-associated macrophages promotes endogenous antitumor immunity and augments adoptive immunotherapy. Nat Commun. 2021;12(1):877.
Liu G, Rui W, Zhao X, Lin X. Enhancing CAR-T cell efficacy in solid tumors by targeting the tumor microenvironment. Cell Mol Immunol. 2021;18(5):1085–95.
Larson R, Maus M. Recent advances and discoveries in the mechanisms and functions of CAR T cells. Nat Rev Cancer. 2021;21(3):145–61.
Klichinsky M, Ruella M, Shestova O, Lu XM, Best A, Zeeman M, Schmierer M, Gabrusiewicz K, Anderson NR, Petty NE, Cummins KD, Shen F, Shan X, Veliz K, Blouch K, Yashiro-Ohtani Y, Kenderian SS, Kim MY, O’Connor RS, Wallace SR, Kozlowski MS, Marchione DM, Shestov M, Garcia BA, June CH, Gill S. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat Biotechnol. 2020;38(8):947–53.
Liu E, Marin D, Banerjee P, Macapinlac HA, Thompson P, Basar R, Nassif Kerbauy L, Overman B, Thall P, Kaplan M, Nandivada V, Kaur I, Nunez Cortes A, Cao K, Daher M, Hosing C, Cohen EN, Kebriaei P, Mehta R, Neelapu S, Nieto Y, Wang M, Wierda W, Keating M, Champlin R, Shpall EJ, Rezvani K. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med. 2020;382(6):545–53.
Zhang L, Tian L, Dai X, Yu H, Wang J, Lei A, Zhu M, Xu J, Zhao W, Zhu Y, Sun Z, Zhang H, Hu Y, Wang Y, Xu Y, Church G, Huang H, Weng Q, Zhang J. Pluripotent stem cell-derived CAR-macrophage cells with antigen-dependent anti-cancer cell functions. J Hematol Oncol. 2020;13(1):153.
Sharma P, Hu-Lieskovan S, Wargo J, Ribas A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707–23.
Chen D, Mellman I. Elements of cancer immunity and the cancer-immune set point. Nature. 2017;541(7637):321–30.
Goliwas KF, Deshane JS, Elmets CA, Athar M. Moving immune therapy forward targeting TME. Physiol Rev. 2021;101(2):417–25.
Rotte A. Combination of CTLA-4 and PD-1 blockers for treatment of cancer. J Exp Clin Cancer Res CR. 2019;38(1):255.
Scott E, Gocher A, Workman C, Vignali D. Regulatory T cells. Barriers of immune infiltration into the tumor microenvironment. Front Immunol. 2021;12:702726.
Galon J, Bruni D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat Rev Drug Discov. 2019;18(3):197–218.
Scott D, Gascoyne R. The tumour microenvironment in B cell lymphomas. Nat Rev Cancer. 2014;14(8):517–34.
Cencini E, Fabbri A, Schiattone L, Sicuranza A, Mecacci B, Granai M, Mancini V, Lazzi S, Bocchia M, Leoncini L. Prognostic impact of tumor-associated macrophages, lymphocyte-to-monocyte and neutrophil-to-lymphocyte ratio in diffuse large B-cell lymphoma. Am J Blood Res. 2020;10(4):97–108.
Keane C, Law SC, Gould C, Birch S, Sabdia MB, Merida de Long L, Thillaiyampalam G, Abro E, Tobin JW, Tan X, Xu-Monette ZY, Young KH, Gifford G, Gabreilli S, Stevenson WS, Gill A, Talaulikar D, Jain S, Hernandez A, Halliday SJ, Bird R, Cross D, Hertzberg M, Gandhi MK. LAG3: a novel immune checkpoint expressed by multiple lymphocyte subsets in diffuse large B-cell lymphoma. Blood Adv. 2020;4(7):1367–77.
Schwaller J, Schneider P, Mhawech-Fauceglia P, McKee T, Myit S, Matthes T, Tschopp J, Donze O, Le Gal FA, Huard B. Neutrophil-derived APRIL concentrated in tumor lesions by proteoglycans correlates with human B-cell lymphoma aggressiveness. Blood. 2007;109(1):331–8.
Jia Q, Qin D, He F, **e Q, Ying Z, Zhang Y, Song Y, Cheng J, Zuo X, Xu L, Fang H, Hu C, Peng L, ** T, Shi Z, Alexander P, Wang Y, Liu Y, Han W, Zhu J, Wang P, Li Q, Zhu B. Peripheral eosinophil counts predict efficacy of anti-CD19 CAR-T cell therapy against B-lineage non-Hodgkin lymphoma. Theranostics. 2021;11(10):4699–709.
Kiyasu J, Miyoshi H, Hirata A, Arakawa F, Ichikawa A, Niino D, Sugita Y, Yufu Y, Choi I, Abe Y, Uike N, Nagafuji K, Okamura T, Akashi K, Takayanagi R, Shiratsuchi M, Ohshima K. Expression of programmed cell death ligand 1 is associated with poor overall survival in patients with diffuse large B-cell lymphoma. Blood. 2015;126(19):2193–201.
Qiu L, Zheng H, Zhao X. The prognostic and clinicopathological significance of PD-L1 expression in patients with diffuse large B-cell lymphoma: a meta-analysis. BMC Cancer. 2019;19(1):273.
Kwon D, Kim S, Kim PJ, Go H, Nam SJ, Paik JH, Kim YA, Kim TM, Heo DS, Kim CW, Jeon YK. Clinicopathological analysis of programmed cell death 1 and programmed cell death ligand 1 expression in the tumour microenvironments of diffuse large B cell lymphomas. Histopathology. 2016;68(7):1079–89.
Ishikawa E, Nakamura M, Shimada K, Tanaka T, Satou A, Kohno K, Sakakibara A, Furukawa K, Yamamura T, Miyahara R, Nakamura S, Kato S, Fujishiro M. Prognostic impact of PD-L1 expression in primary gastric and intestinal diffuse large B-cell lymphoma. J Gastroenterol. 2020;55(1):39–50.
Pollari M, Brück O, Pellinen T, Vähämurto P, Karjalainen-Lindsberg M, Mannisto S, Kallioniemi O, Kellokumpu-Lehtinen P, Mustjoki S, Leivonen S, Leppä S. PD-L1 tumor-associated macrophages and PD-1 tumor-infiltrating lymphocytes predict survival in primary testicular lymphoma. Haematologica. 2018;103(11):1908–14.
Lim B, Lin Y, Navin N. Advancing cancer research and medicine with single-cell genomics. Cancer Cell. 2020;37(4):456–70.
Moncada R, Barkley D, Wagner F, Chiodin M, Devlin JC, Baron M, Hajdu CH, Simeone DM, Yanai I. Integrating microarray-based spatial transcriptomics and single-cell RNA-seq reveals tissue architecture in pancreatic ductal adenocarcinomas. Nat Biotechnol. 2020;38(3):333–42.
Zhao T, Lyu S, Lu G, Juan L, Zeng X, Wei Z, Hao J, Peng J. SC2disease: a manually curated database of single-cell transcriptome for human diseases. Nucleic Acids Res. 2021;49(D1):D1413–9.
Yuan H, Yan M, Zhang G, Liu W, Deng C, Liao G, Xu L, Luo T, Yan H, Long Z, Shi A, Zhao T, **ao Y, Li X. CancerSEA: a cancer single-cell state atlas. Nucleic Acids Res. 2019;47(D1):D900–8.
He L, Vanlandewijck M, Mae MA, Andrae J, Ando K, Del Gaudio F, Nahar K, Lebouvier T, Lavina B, Gouveia L, Sun Y, Raschperger E, Segerstolpe A, Liu J, Gustafsson S, Rasanen M, Zarb Y, Mochizuki N, Keller A, Lendahl U, Betsholtz C. Single-cell RNA sequencing of mouse brain and lung vascular and vessel-associated cell types. Sci Data. 2018;5(1):180160.
Franzen O, Gan LM, Bjorkegren JLM. PanglaoDB: a web server for exploration of mouse and human single-cell RNA sequencing data. Database (Oxford). 2019;2019:baz046.
Zhang X, Lan Y, Xu J, Quan F, Zhao E, Deng C, Luo T, Xu L, Liao G, Yan M, ** Y, Li F, Shi A, Bai J, Zhao T, Li X, **ao Y. Cell Marker: a manually curated resource of cell markers in human and mouse. Nucleic Acids Res. 2019;47(D1):D721–8.
Cao ZJ, Wei L, Lu S, Yang DC, Gao G. Searching large-scale scRNA-seq databases via unbiased cell embedding with Cell BLAST. Nat Commun. 2020;11(1):3458.
Björkegren JLM, Franzén O, Gorodkin J. alona: a web server for single-cell RNA-seq analysis. Bioinformatics. 2020;36(12):3910–2.
Gong B, Wang R, Xu H, Miao M, Yao Z. Nanotherapy targeting the tumor microenvironment. Curr Cancer Drug Targets. 2019;19(7):525–33.
Yang M, Li J, Gu P, Fan X. The application of nanoparticles in cancer immunotherapy: targeting tumor microenvironment. Bioact Mater. 2021;6(7):1973–87.
Cheng Z, Li M, Dey R, Chen Y. Nanomaterials for cancer therapy: current progress and perspectives. J Hematol Oncol. 2021;14(1):85.
Del Piccolo N, Shirure V, Bi Y, Peter Goedegebuure S, Gholami S, Hughes C, Fields R, George S. Tumor-on-chip modeling of organ-specific cancer and metastasis. Adv Drug Deliv Rev. 2021;175:113798.
Garcia-Carbonero R, Martin MG, Gallego RA, Mercade TM, Martinez MCR, Guillen-Ponce C, Vidal N, Real FX, Moreno R, Maliandi V, Mato-Berciano A, Bazan-Peregrino M, Capella G, Alemany R, Blasi E, Blasco C, Cascallo M, Salazar R. Systemic administration of the hyaluronidase-expressing oncolytic adenovirus VCN-01 in patients with advanced or metastatic pancreatic cancer: first-in-human clinical trial. Ann Oncol. 2019;30:271–2.
Acknowledgements
Not applicable.
Funding
This study was supported by National Natural Science Foundation (No. 81800194, No. 82070203, No. 81770210, No. 81473486 and No. 81270598); Key Research and Development Program of Shandong Province (No. 2018CXGC1213); Development Project of Youth Innovation Teams in Colleges and Universities of Shandong Province (No. 2020KJL006); China Postdoctoral Science Foundation (No. 2021T140422, No. 2020M672103); Technology Development Projects of Shandong Province (No. 2017GSF18189); Translational Research Grant of NCRCH (No. 2021WWB02, No. 2020ZKMB01); Shandong Provincial Natural Science Foundation (No. ZR2018BH011); Technology Development Project of **an City (No. 201805065); Taishan Scholars Program of Shandong Province; Shandong Provincial Engineering Research Center of Lymphoma; and Academic Promotion Programme of Shandong First Medical University (No. 2019QL018, No. 2020RC006).
Author information
Authors and Affiliations
Contributions
Y.L. wrote the manuscript and created figures and tables. X.Z. reviewed and revised the manuscript. X.W. provided direction and guidance throughout the preparation of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no relevant conflicts.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Liu, Y., Zhou, X. & Wang, X. Targeting the tumor microenvironment in B-cell lymphoma: challenges and opportunities. J Hematol Oncol 14, 125 (2021). https://doi.org/10.1186/s13045-021-01134-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13045-021-01134-x