Abstract
Chimeric antigen receptor (CAR) T cell therapy has been successful in treating B cell malignancies in clinical trials; however, fewer studies have evaluated CAR T cell therapy for the treatment of T cell malignancies. There are many challenges in translating this therapy for T cell disease, including fratricide, T cell aplasia, and product contamination. To the best of our knowledge, no tumor-specific antigen has been identified with universal expression on cancerous T cells, hindering CAR T cell therapy for these malignancies. Numerous approaches have been assessed to address each of these challenges, such as (i) disrupting target antigen expression on CAR-modified T cells, (ii) targeting antigens with limited expression on T cells, and (iii) using third party donor cells that are either non-alloreactive or have been genome edited at the T cell receptor α constant (TRAC) locus. In this review, we discuss CAR approaches that have been explored both in preclinical and clinical studies targeting T cell antigens, as well as examine other potential strategies that can be used to successfully translate this therapy for T cell disease.
Similar content being viewed by others
Introduction
T cell malignancies encompass a heterogeneous group of diseases, each reflecting a clonal evolution of dysfunctional T cells at various stages of development. T cell acute lymphoblastic leukemia (T-ALL) accounts for 15% and 25% of childhood and adult ALL cases respectively, and is the most common form of T cell cancer seen in children [1, 2]. T-lymphoblastic lymphoma (T-LLy) is a non-Hodgkin lymphoma with similar biology to T-ALL. Adult T cell leukemia/lymphoma (ATLL) is an extremely aggressive form of blood cancer driven by the human T cell lymphocytic virus type 1 (HTLV1) [3,4,5]. Other rare forms of T cell leukemia include T cell large granular lymphocytic leukemia (T-LGL) and T-prolymphocytic leukemia (T-PLL) [6]. T cell lymphomas are broadly divided into two categories, cutaneous T cell lymphoma (CTCL) and peripheral T cell lymphoma (PTCL) [7]. Mycosis fungoides (MF) and Sezary syndrome (SS) represent the two most common subtypes of CTCL, accounting for the majority of cases [8]. PTCL can be classified into several different subtypes, among which include anaplastic large cell lymphoma (ALCL), angioimmunoblastic T cell lymphoma (AITL), extranodal natural killer (NK)-T cell lymphoma (ENKTL), enteropathy-associated T cell lymphoma (EATL), hepatosplenic T cell lymphoma (HSTCL), and PTCL-not otherwise specified (PTCL-NOS) which is the most common of the group [9, 10].
The overall prognosis for T cell malignancies varies depending on the type of disease, but in general is much poorer when compared to B cell malignancies. While the survival in T-ALL/LLy has significantly improved with the intensification of chemotherapy, there still remain very limited options for patients with relapsed/refractory disease [11,12,13]. ATLL remains a very challenging disease to treat, with a median survival of less than 12 months for the acute form of this disease [3,4,5]. Advanced stage CTCL has a median overall survival of 5 years [14, 15], whereas outcomes of PTCL vary depending upon the subtype, with ENKTL, EATL, and HSTCL having the poorest prognosis [9, 10]. While immunotherapy has revolutionized the treatment landscape of various cancers with the use of monoclonal antibodies, checkpoint inhibitors, bispecific T cell engagers, and chimeric antigen receptor (CAR) T cell therapy, only limited responses have been seen in T cell disease [15]. Some promising results have been seen with use of brentuximab vedotin, a CD30-directed immunotoxin, in CD30-positive PTCL and CTCL [16, 17] and the use of pembroluzimab, a programmed cell death receptor 1 (PD-1) inhibitor, in the treatment of ENKTL [18]; however, these positive results have been limited to very specific subsets of T cell disease. One form of immunotherapy that has not yet been successfully translated to T cell malignancies is that of chimeric antigen receptor (CAR)-based immunotherapy. CAR T cell therapy has been extremely successful in relapsed/refractory B cell malignancies as evidenced by the recent Food and Drug Administration (FDA) approval of two CAR T cell therapeutics for this disease [19,20,21,22,23]. However, implementing this technology to treat T cell malignancies has been difficult, primarily due to the lack of a tumor-specific surface antigen in cancerous T cells. In this review, we will discuss the challenges involved in translating this novel technology to T cell disease, review all the preclinical and clinical progress made in adapting this therapy for this challenging disease, and examine potential solutions for the future development of this innovative therapy.
CAR T cell therapy
Genetic engineering of primary T cells was first presented in the late 1980s [24]. Since then, chimeric antigen receptor T cells have emerged as a promising technique for the treatment of relapsed/refractory malignancies. CAR therapy brings together numerous fields including immunology, tumor biology, genetic engineering, synthetic biology, and pharmacology. CARs are comprised of the intracellular signaling domain from the natural T cell receptor (TCR), CD3ζ, linked to a single-chain variable fragment (scFv) which serves as the antigen recognition domain. The scFv sequence is derived from a monoclonal antibody by combining the variable heavy (VH) and light (VL) domains using a small peptide linker. Commonly used CARs also include one or two costimulatory domains, such as CD28, 4-1BB, ICOS, and/or OX40. Although the kinetics have yet to be fully elucidated, it is essential that CAR T cells have mechanisms of trafficking to the tumor site where they can recognize their cognate antigen. This results in CAR T cell activation and expansion, and ultimately cytolytic activity against cells expressing the target antigen. CAR-based ligand recognition is advantageous compared to TCR-based ligand recognition because CAR-targeting is not restricted by major histocompatibility complex (MHC) interactions. Therefore, CARs can recognize cell surface proteins that have not been processed and presented by antigen presenting cells (APCs). Importantly, the interactions between scFvs and ligands have much higher affinity and avidity compared to that of TCR-ligand interactions [25]. Furthermore, the immune synapse formed from the interaction between a CAR and its ligand likely results in a much greater functional avidity than is observed using a targeted antibody approach with the same antibody (25).
CARs targeting the B cell antigen CD19 have been studied extensively for the treatment of B cell malignancies. In 2017, the FDA approved the first CAR T cell therapy, Kymriah, a CD19-directed CAR therapy for the treatment of relapsed/refractory B cell acute lymphoblastic leukemia (B-ALL) and in 2018, Yescarta was approved to treat relapsed diffuse large B cell lymphoma (DLBCL). These therapies, including others in clinical trials, have been widely successful in eliminating malignant cells and re-inducing remission in patients who were otherwise treatment-refractory [19,20,21, 26, 27]. Patients receiving CAR therapy undergo leukapheresis resulting in the collection of T cells, which are subsequently modified using a lentiviral or retroviral vector to express the CAR. These cells are expanded ex vivo while the patient undergoes lymphodepletion, a process involving chemotherapeutic agents. Finally, the CAR T cells are re-infused into the patient [28]. Lymphodepletion prior to re-infusion of the autologous T cells has been shown to augment both CAR T cell proliferation as well as persistence [29,30,31]. The administered dose of CAR T cells and the pre-existing tumor burden do not appear to be the sole determinants of the degree of T cell expansion, engraftment, and overall response. Other factors may be involved, such as the density of cognate antigen expression on the cancer cells [32]. However, the optimal degree of persistence of CAR T cells required to prevent leukemic relapse has not been determined [25, 33].
One of the mechanisms of relapse post-CD19 CAR T cell therapy is due to surface antigen escape with relapsed leukemia cells being CD19-negative. The mechanism may be due to the expansion of a small subset of CD19-negative cancer cells or alternatively, the cells may downregulate CD19 from the cell surface in order to evade detection by CAR T cells, rendering them resistant [19, 21, 34,35,36,37]. Additionally, it was recently shown that a phenomenon referred to as trogocytosis is a mechanism of antigen escape whereby the antigen is transferred to the CAR T cell [38]. It has also been shown that transduction of a single leukemic blast with an anti-CD19 CAR that was re-infused into a B-ALL patient, ultimately resulted in relapse and death of the patient [39]. Transduction of the leukemic cell resulted in masking of the target antigen through interactions between the CAR and the cognate antigen on the same cell. Clonal expansion of this population resulted in resistance to CAR therapy. This report emphasized the importance of strict and perfect isolation of normal, healthy T cells for modification with the CAR construct. As we discuss below, this is particularly challenging in T cell leukemia patients who are more likely to have circulating cancerous T cells, and therefore have a higher probability of these cells being inadvertently isolated, transduced, and re-infused.
Of note, there are severe toxicities that have been associated with CAR therapy. Cytokine release syndrome (CRS) is a systemic inflammatory response directly resulting from robust T cell activation following infusion. IL-6 is one pro-inflammatory cytokine that is secreted at high levels during CRS. During a particularly severe CRS condition, tocilizumab, an IL-6R antagonist monoclonal antibody, was used to rapidly and effectively reverse the symptoms of a pediatric patient [27]. Tocilizumab has since been FDA approved for treatment of CAR T cell-induced life-threatening CRS [40]. Neurological toxicities have been reported following CAR T cell infusion as well; however, preventative approaches remain elusive [36, 41,42,43,44]. Compared to CRS and neurotoxicity, a much more manageable consequence of CAR T cell therapy targeting B cell malignancies is the resulting B cell aplasia. This is a potentially lifelong outcome due to memory cell formation against a B cell antigen; but currently is managed by periodic infusions of intravenous immunoglobulins. Unfortunately, this is an extremely problematic outcome for T cell malignancies, as persistent T cell aplasia would be life threatening. There are currently > 200 clinical trials using CAR T cells registered at clinicaltrials.gov being carried out in the USA. However, the majority of these trials are enrolling patients with B cell malignancies. Advances are being made to expand CAR T cell therapy to the treatment of other cancers, and to minimize toxicities associated with treatment while reducing difficulty and cost of production.
Translating CAR T cell therapy for treatment of T cell malignancies
Harnessing and redirecting the cytotoxicity of T cells to malignant B cells has been established, but reprogramming T cells to kill malignant T cells, while sparing normal T cells, is much more complex and challenging. This requires aberrant expression of an antigen on malignant T cells that is absent or expressed at very low levels on normal T cells. CAR therapy requires isolation of healthy T cells from malignant T cells, a complicated procedure that can result in product contamination and subsequent CAR-modification of tumor cells. Additionally, expression of the targeted antigen on CAR T cells results in fratricide and limited expansion of the CAR T cells. Furthermore, targeting of an antigen regularly expressed on normal T cells would result in T cell aplasia, leading to profound immunosuppression, likely to be associated with high rates of morbidity and mortality (Fig. 1).
Potential outcomes of CAR T cell therapy in a patient with T cell disease. Upon re-infusion into a patient, CAR T cells recognize their cognate antigen, expanding upon this recognition, and initiating an attack. However, due to shared antigen expression on CAR T cells, normal T cells, and tumor cells, numerous outcomes can be observed. CAR T cells target tumor cells as intended, reducing tumor burden. However, without further engineering, the CAR-modified T cells are likely to express the targeted antigen as well, resulting in fratricide. CAR T cells would also target healthy T cells, resulting in unintended T cell aplasia. Lastly, CAR T cell therapy involves isolating normal T cells from malignant T cells for CAR-modification. A single malignant cell contaminating this population can result in masking of the antigen, leading to antigen-positive relapse. *Figure was created using BioRender
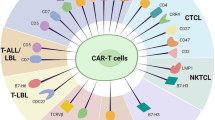
Various approaches have been used to overcome these challenges, including CRISPR-Cas9 genome editing to remove the antigen from the CAR T cells [45,46,47], Tet-OFF expression system to limit fratricide during ex vivo expansion [48], protein expression blocker (PEBL) to retain the antigen in the ER/Golgi to prevent cell surface expression [49, 50], or using CAR-modified natural killer cells instead of T cells [47, 51,52,53,54]. Additionally, to date, four targets have been investigated as targets for CAR T cell therapy for the treatment of T cell malignancies with limited to no expression in the normal population of T cells, CD30, CD37, TRBC1, and CD1a [55,56,57,58]. Table 1 provides a summary of potential solutions to the three main challenges seen in adapting CAR technology for T cell malignancies—fratricide, T cell aplasia, and product contamination. A list of all current CAR-based clinical trials targeting T cell disease is presented in Table 2. Below, we review all preclinical and clinical CAR studies targeting T cell malignancies categorized according to the target antigen of interest.
CD5
CD5 expression is limited to normal T cells and a small subpopulation of B cells, called B-1a cells [65,66,67,68,69]. CD5 acts as a negative regulator of TCR signaling and has a role in protecting against autoimmunity [70, 71]. CD5 is highly expressed on many T cell malignancies, particularly T-ALL and PTCLs, rendering it a good target for CAR T cell therapy [72,73,74]. Since CD5 expression on T cells is approximately ten times that on B cells [75], a low-affinity, high-avidity CAR targeting CD5 may steer clear of CD5-positive B cells while selectively killing T cells [76, 77]. Furthermore, CD8+ tumor-infiltrating lymphocytes (TILs) express lower levels of CD5 compared to that of peripheral blood T cells, and one study showed downregulation of CD5 improves the ability of T cells to lyse malignant cells [78]. CD5 was previously targeted as a tumor antigen in clinical trials using immunotoxin-conjugated CD5 monoclonal antibodies, with responses seen in patients with cutaneous T cell lymphoma and T-ALL [79, 80].
A preclinical study showed that expression of a CD5-CAR with a CD28 costimulatory domain resulted in surface downregulation of CD5 in CAR T cells. As a result, fratricide was observed only transiently, allowing the CD5-CAR T cells to expand. These cells had significant in vitro cytotoxicity against two T-ALL cell lines and primary T-ALL cells and delayed leukemia progression in two different CD5-positive T-ALL models [59]. Based on these results, CD5-CAR T cells with a CD28 costimulatory domain are being tested in patients with relapsed or refractory T cell disease (MAGENTA trial, NCT03081910). Our group used CRISPR-Cas9 to knockout CD5 expression in primary T cells prior to transduction with the CD5-CAR. We showed that gene editing of CD5 in effector CAR T cells increased CAR surface expression and decreased self-activation [47]. The increased CAR surface expression is predicted to enhance CAR T cell anti-tumor efficacy. We also showed antagonism of vasoactive intestinal peptide (VIP) signaling in conjunction with inhibition of the PI3Kδ pathway increased expansion of CD5-CAR-modified T cells as well as their cytotoxicity against CD5-specific tumor cell lines. This combination of compounds was also demonstrated to prolong in vivo persistence of treated T cells in NOD scid IL2Rγ-chain knockout (NSG) mice [81].
Interestingly, use of 4-1BB as the costimulatory domain in a CD5-CAR resulted in a significant fratricidal effect [48]. It was shown that tumor necrosis factor (TNF) receptor-associated factor (TRAF) signaling from the 4-1BB endodomain upregulated the intercellular adhesion molecule 1 (ICAM1), which subsequently stabilized the fratricidal immunological synapse between CD5-CAR T cells containing the 4-1BB costimulatory domain. To limit and control the effects of fratricide, a Tet-OFF expression system was used, which allowed for controlled transgene expression using the small molecule inhibitor, doxycycline. In the presence of doxycycline, CD5-41BB-CAR T cells expanded ex vivo without evidence of fratricide, while maintaining a more naïve genotype. Doxycycline was removed from the culture prior to injecting the CD5-41BB-CAR T cells into mice, resulting in CD5-CAR expression and improved survival outcomes in a T-ALL mouse model. Furthermore, there was a survival advantage in mice treated with Tet-OFF CD5-41BB-CAR T cells compared to survival of mice treated with CD5-CD28-CAR T cells without the Tet-OFF expression system [48].
Alternatively, we expressed the CD5-CAR in NK-92 cells, an interleukin-2 (IL-2) dependent natural killer cell line, which are inherently CD5-negative. Our data demonstrates that CD5-CAR-modified NK-92 cells have increased cytotoxicity against T cell leukemia cell lines compared to the cytotoxicity of naïve NK-92 cells [47, 51], and there is a significant improvement in survival of T-ALL xenograft mouse models compared to survival of mice treated with naïve NK-92 cells [47]. This data confirms previously published data illustrating significantly improved survival and enhanced tumor reduction in irradiated T-ALL mouse models treated with CD5-CAR-modified NK-92 cells compared to that of mice treated with control NK-92 cells [53]. Recently, another group tested CD5-CAR-modified NK-92 cells, using a NK-specific costimulatory domain 2B4 in their CAR constructs [82]. Interestingly, the CD5-2B4-CAR NK-92 cells displayed superiority to CD5-41BB-CAR NK-92 cells, in both in vitro and in vivo experiments [82].
CD7
CD7 is a transmembrane glycoprotein with expression on T cells and NK cells [83]. The majority of T-ALLs are CD7-positive, despite some populations lacking expression of other common markers, such as the TCR [74, 84]. Additionally, early T cell precursor acute lymphoblastic leukemia (ETP-ALL), a high-risk subset of T-ALL, highly express CD7 [84,85,86]. Two clinical trials have been initiated in China studying CD7-CAR-modified T cells for the treatment of CD7-positive malignancies (NCT04033302 and NCT04004637). However, preclinical studies showed significantly reduced expansion of CD7-CAR T cells compared to control T cells, as a result of fratricide [45, 49]. Fratricide appears to be observed to a greater extent in CD7-CAR T cells compared to CD5-CAR T cells [45]. It is hypothesized that this is due to a more incomplete internalization mechanism of CD7 from the cell surface following ligation of the antigen with an anti-CD7 scFv. CRISPR-Cas9 editing of CD7 from the cell surface of T cells prior to CAR expression demonstrated a superior method of develo** CD7-CAR T cells. These cells exhibited limited fratricide, expanded in vitro, and showed no evidence of impaired cytotoxicity in vitro nor in vivo. Investigations in a T-ALL mouse xenograft model revealed a statistically significant prolonged survival of CD7-edited CD7-CAR-treated mice compared to survival of control mice [45]. Based on these results, a phase I clinical trial has been initiated testing CD7-CD28-CAR T cells in T-ALL patients (NCT03690011). Additionally, a UCART7 was generated using CRISPR-Cas9 genome editing to disrupt the CD7 and TCRα constant (TRAC) loci. This study demonstrated that NSG mice engrafted with primary T-ALL blasts and treated with UCART7 donor cells exhibited tumor clearance from the peripheral blood, and, did not develop graft versus host disease (GvHD) or other severe side effects [46].
A new technique using protein expression blockers (PEBLs) has been established as an alternative to genome editing. This strategy couples an scFv with a retention peptide to maintain the protein of interest in the ER/Golgi preventing cell surface expression of the antigen. PEBL-CD7-CAR T cells exhibited superior cytotoxicity against primary T-ALL cells in vitro compared to non-PEBL CD7-CAR T cells. Using a patient-derived xenograft (PDX) model of ETP-ALL, upon detection of leukemic cell expansion in peripheral blood, PEBL-CD7-CAR T cells were injected. PEBL-CD7-CAR T cell-treated mice had a significant survival advantage over control mice. However, CD7-positive relapse did occur in all PEBL-CD7-CAR T cell-treated mice [49].
Despite CD7 expression on NK-92MI cells (IL-2 producing NK-92 cells), they have been used for CD7-CAR therapy demonstrating only a small percentage of cells are CD7-positive, and upon CD7-CAR expression, fewer than 1% CD7-positive NK-92MI cells remain [60]. Two CD7-CAR constructs, a monovalent and bivalent construct, were generated using a humanized CD7 nanobody sequence that had been previously developed in the laboratory. Both CAR constructs demonstrated enhanced CD7-specific cytotoxicity against T-ALL cell lines and primary patient cells ex vivo when expressed in NK-92MI cells. The bivalent CD7-CAR-modified-NK-92MI cells exhibited slightly greater cytotoxicity compared to that of the monovalent CAR-modified cells, and significantly inhibited disease progression in a T-ALL PDX model when compared to naïve unmodified NK-MI cells.
CD4
Most cancers derived from lineage-differentiated T cells are likely to be of CD4-positive origin, making CD4 a potential target for CAR therapy. A preclinical study was performed to consider the cytotoxicity of CD4-CAR-modified T cells against T-ALL tumors in NSG mice. This study also included the use of alemtuzumab to clear the CAR T cells as a safety mechanism. NSG mice were injected with luciferase-expressing Jurkat T cells and subsequently treated with naïve T cells or CD4-CAR-modified T cells. CAR-treated mice displayed a survival advantage and an ~ 80% reduction in tumor burden compared to mice treated with naïve T cells. CD4-CAR-modified T cells were also injected into mice to evaluate the ability of alemtuzumab to effectively eliminate CAR-modified T cells. Alemtuzumab was administered 24 h post-CAR T cell injection. A > 95% depletion of CD4-CAR-modified T cells was observed within 6 h following injection signifying the use of alemtuzumab as a safety mechanism for CAR T cell therapy [87]. Additionally, a phase I clinical trial to assess the safety and feasibility of CD4-CAR T cell infusions in patients with relapsed/refractory T cell lymphoma and T cell leukemia has been initiated (NCT03829540).
However, expression of CD4 on T cells can complicate CD4-CAR T cell therapy as previously described. NK-92 cells are inherently CD4-negative, and therefore the use of NK-92 cells as opposed to T cells reduces risk of fratricide and avoids the need for further modifications. Additionally, it abrogates the risk of aplasia of CD4-positive cells that can occur with long-term engraftment of CAR T cells. Anti-CD4-CAR NK-92 cells have shown in vitro success eliminating PTCL cell lines and both adult and pediatric primary cells. Using a xenograft model in NSG mice, CD4-CAR NK-92 cell-treated mice demonstrate significantly prolonged survival compared to control-modified NK-92 cell-treated mice [54].
CD37
CD37 is a member of the tetraspanin superfamily with expression limited to lymphoid tissues, particularly B cells [88, 89]. CD37 expression in cancer cells is typically characteristic of B cell malignancies; however, its expression can be found in some cases CTCL and PTCL [90, 91]. Since CD37 is not expressed in T cells, there is no evidence of fratricide occurring in anti-CD37 CAR T cells. However, in the presence of CD37-positive PTCL cell lines, CD37-CAR T cells exhibit increased activation and degranulation as well as specific cytolytic activity in vitro [56]. The restricted expression of CD37 makes it a safer target for CAR T cell therapy, given there would be no concern of T cell aplasia. Additionally, CD37 is not expressed in NK cells, providing an opportunity to utilize NK cells as effector cells in place of T cells. The versatility of CD37-CARs to treat B cell and T cell lymphomas suggests that this may be an important target for further investigations. While CD37 is predominantly being examined for dual targeting for B cell malignancies, the target has potential for CAR therapy against T cell malignancies.
CD30
CD30, a member of the tumor necrosis factor receptor (TNFR) superfamily, promotes T cell proliferation and cytokine production following TCR stimulation, while also having an opposing role in promoting apoptosis [92]. Expression is limited to a subset of activated lymphocytes found around the follicular regions of lymphoid tissues [93,94,95]. While CD30 is well known for its strong expression in virtually all classical Hodgkin lymphoma, expression of CD30 can also be found on a subset of PTCLs, including ALCL [92,93,94, 96]. One study demonstrated that CD30 expression is upregulated during chemotherapy regimens in T-ALL patients. Of 34 T-ALL patients, approximately 38% had CD30-positive T-ALL [96]. Therefore, some T-ALL patients who relapse following chemotherapy may still respond to CD30-directed CAR therapy.
Preclinical studies have previously demonstrated CD30-CAR T cell capacity for lysing tumor cells [97, 98] and numerous clinical investigations into CD30-CAR T cell therapy have been launched with encouraging results. Eleven phase I/II trials treating patients with CD30-positive malignancies are currently active (NCT01316146 [55], NCT01192464, NCT03049449, NCT02690545 [62], NCT02958410, NCT02663297, NCT03383965, NCT02917083 [64], NCT04008394, NCT02259556 [63], and NCT03602157). To date, no toxicities related to CAR T cell infusion nor impaired immunity against common viruses has been reported from these trials. However, one trial reported that the in vivo CAR T cell expansion and persistence was reduced following subsequent infusions compared to those following initial doses [55]. The decreased persistence of the CAR T cells may have prevented the development of severe adverse events such as CRS and neurotoxicity that are commonly observed following CAR T cell infusion. Of the two ALCL patients in this trial, one patient was non-responsive to the therapy, while the other entered complete remission lasting 9 months [55]. Results from another phase I trial in China for patients with relapsed/refractory CD30-positive lymphomas (NCT02259556) corroborate the limited toxicity and anti-tumor activity of CD30-CAR T cells [63].
TRBC1
T cells express the αβ TCR; the β-chain can either be encoded by the T cell receptor beta constant 1 (TRBC1) gene or TRBC2 gene [99, 100]. Therefore, expression of TRBC1 and TRBC2 is mutually exclusive. Additionally, CD4- and CD8-positive T cell populations express both subsets and CD8-positive T cell populations specific for common viruses also contain both TRBC1 and TRBC2 cells [58]. However, as malignant T cells develop from a single cell, the entire population of cancerous cells will be either TRBC1- or TRBC2-positive. Numerous T cell malignancy cell lines and primary samples have been analyzed by flow cytometry to validate the homogeneity of β-chain expression in a malignant cell population [58]. Many cancer cells downregulate the αβ TCR; however, it is expressed on > 95% of PTCLs [101] and > 30% of T-ALLs [102].
Anti-TRBC1 CAR T cells exhibited specific and efficient cytotoxicity against the JKO T cell line transduced with TRBC1, but not against non-transduced cells or cells transduced with TRBC2, even in a mixed population. Furthermore, in primary samples from patients with T cell malignancies, the anti-TRBC1 CAR T cells preserved a significant fraction of healthy T cells (TRBC2 cells), thereby circumventing a limitation of CAR T cell therapy for the treatment of T cell malignancies [58]. In an NSG mouse model using TRBC1-positive Jurkat T cells to establish cancer, mice treated with the anti-TRBC1 CAR T cells exhibited reduced tumor burden and elongated survival. In additional preclinical studies, NSG mice were injected with both TRBC1 and TRBC2 cancer cells, and then treated with either naïve T cells or anti-TRBC1 CAR T cells. TRBC1-positive Jurkat T cells could not be detected in mice treated with anti-TRBC1 CAR T cells; however, TRBC2-positive cells were identified. This is in contrast to mice treated with naïve T cells, whose bone marrow confirmed the presence of both TRBC1-positive and TRBC2-positive cells [58]. Thus, targeting TRBC1-positive malignant cells offers a unique approach to avoiding T cell aplasia, a consequence of many proposed CAR T cell therapies for the treatment of T cell malignancies.
CD3
CD3 is a pan T cell marker comprised of four distinct polypeptide chains, epsilon, gamma, delta, and zeta, which form pairs of dimers, transmitting T cell activation signals. As CD3 is exclusively expressed on T cells, it has been a popular target in preclinical CAR T cell therapies for the treatment of T cell malignancies. As expected, due to fratricidal issues, manufacturing of anti-CD3 CAR T cells does not yield a viable cellular product [61]. Various approaches using an anti-CD3 CAR have been investigated including the use of transcription activator-like effector nuclease (TALEN) mRNA to disrupt the TRAC locus and using NK-92 cells in place of T cells as the effector cell type. Disruption of the TRAC locus prevents assembly of the TCRαβ/CD3 complex, allowing for anti-CD3-CAR expression without compromising cellular proliferation and viability. Enrichment of the CAR-positive, CD3-negative population was observed. In patient T-ALL samples, anti-CD3 CAR T cells demonstrated specific cytotoxicity against CD3-positive cells. In a T-ALL NSG model, anti-CD3 CAR T cells were shown to clear luciferase-expressing CD3-positive Jurkat cells, but showed no effect in NSG mice engrafted with CD3-negative Jurkat cells [61]. To circumvent the need for additional modifications, NK-92 cells can also be used to express the anti-CD3-CAR, since they are CD3-negative cells. CD3-CAR NK-92 cells demonstrated efficient ex vivo lysis of PTCL primary samples, resulting in less than 0.5% lymphoma cells remaining at 5:1 effector to target ratios. Furthermore, CD3-CAR NK-92-treated T-ALL NSG mice exhibited prolonged survival with ~ 87% reduced tumor burden through day 23 [52].
CD1a
CD1a is a lipid-presenting molecule whose expression is restricted to develo** cortical thymocytes, skin Langerhans cells, and some circulating myeloid dendritic cells [145]. However, recently developed methods are being used to enhance NK-cell expansion, such as through K562-feeder cell expression of OX40 ligand [146]. As mentioned above, a limitation to CAR-NK therapy is the extreme sensitivity of NK cells to cryopreservation. They have demonstrated poor viability and diminished cytotoxicity after cryopreservation. While cytotoxicity can be restored to normal levels after a few days in culture with exogenous IL-2, the low viability post-cryopreservation remains a concern [141].
Gamma delta T cells
While αβ T cells function as a part of the adaptive immune system, γδ T cells play roles in both the innate and the adaptive immune systems. γδ T cells and αβ T cells originate from two distinct T cell lineages [224]. γδ T cells are the only innate immune cells expressing a TCR [147]; however, their target recognition is independent of MHC recognition [148, 149]. Lack of MHCI- and MHCII-restriction make γδ T cells optimal candidates for allogeneic cell therapy. The peripheral blood subset of γδ T cells known as Vγ9Vδ2 T cells represents the most commonly studied subset in this context. Studies by our group have demonstrated that similar transduction efficiencies can be achieved in Vγ9Vδ2 T cells grown under cGMP serum-free conditions as are achieved in αβ T cells using lentiviral vectors. Additional studies were performed revealing peak low-density lipoprotein receptor (LDL-R) expression on days 6–8 of γδ T cell expansion [150]. As LDL-R is the major receptor for VSV-G-pseudotyped lentiviral vectors, this data suggests that greater transduction efficiency can be achieved on these days using lentiviral vectors compared to earlier or later in the expansion [151].
To date, numerous preclinical studies have evaluated CAR-modified γδ T cells targeting neuroblastoma [152, 153], melanoma [154], B cell malignancies [153, 155], and epithelial cell adhesion molecule (epCAM)-positive adenocarcinomas [156]. GD2-CAR-modified γδ T cells expressing the RQR8 suicide gene were shown to expand 2.5-fold upon antigen exposure [152]. Furthermore, both GD2-CAR- and CD19-CAR-modified γδ T cells were demonstrated to secrete pro-inflammatory cytokines in the presence of GD2- or CD19-expressing tumor cells, respectively [153]. While these studies utilized viral vectors to express the CAR, electroporation of a Slee** Beauty transposon has also been shown to result in CD19-CAR expression in γδ T cells, resulting in anti-tumor cytotoxicity in both the in vitro and in vivo settings [155]. Additionally, expression of a CAR targeting melanoma-associated chondroitin sulfate proteoglycan (MCSP) was established in γδ T cells using mRNA transfection. Despite comparable anti-tumor cytotoxicity, lower cytokine secretion was observed in MCSP-CAR-modified γδ T cells compared to that from conventional CAR-modified αβ T cells [154]. Reduced pro-inflammatory cytokine secretion is favorable due to anticipated reduced severity of CRS. Lastly, epCAM CAR-modified γδ T cells demonstrated high levels of in vitro cytotoxicity of tumor cell lines when γδ T cells were both fresh and cryopreserved [156]. These studies pave the way for additional trials using CAR-modified γδ T cells targeting T cell malignancies. They demonstrate that engineering of γδ T cells is feasible and results in enhanced in vitro and in vivo cytotoxicity upon CAR expression.
CAR-modified γδ T cells may be able to overcome the obstacle of antigen escape seen in some treatment-resistant cases by relying on their innate ability to recognize tumor cells through other means. Naïve γδ T cells have been shown to have anti-tumorigenic activity against leukemia, neuroblastoma, and colon cancer cell lines as well as primary cancer cells in vitro [2). NK cells (i) are non-alloreactive and can be obtained from healthy donors, eliminating risk of product contamination; (ii) do not form memory responses, preventing T cell aplasia; and (iii) do not express the same antigen repertoire as T cells, avoiding fratricidal concerns. CD7 is an exception as it is expressed on NK cells and therefore fratricide could occur. While several groups have published studies with CAR NK-92 cells targeting T cell malignancies, more effort needs to be put into using primary NK cells for targeting this disease, especially given the limitations of NK-92 cells. Other, equally promising approaches, such as utilizing γδ T cells as the cellular vehicle for CAR therapy represents an alternative, less studied approach. Similar to NK cells, γδ T cells are non-alloreactive and are unlikely to form a memory response against a T cell antigen. γδ T cells are likely to succumb to fratricide in certain circumstances; however, targeting an antigen such as CD5 that results in only transient and limited fratricide may be especially advantageous. Furthermore, γδ T cells exhibit innate MHC-independent mechanisms of cytotoxicity by which they can recognize tumor cells. Thus, CAR therapy using γδ T cells represents an understudied avenue with the potential of develo** into a superior cellular product.
Venn diagram representing challenges and solutions in targeting T cell antigens with CAR therapy. Each circle represents a hurdle associated with translation of CAR therapy to T cell disease—fratricide, T cell aplasia, and product contamination. As seen in the figure, only the use of NK cells or NK-92 cells as the CAR-effector cell can potentially address all three issues concurrently. However, using NK cells or NK-92 cells comes with its own limitations as previously described. All other approaches require multiple modifications to generate a translatable CAR product to target T cell disease. Potential alternative solutions such as use of γδ T cells as the CAR-effector cell, transient CAR expression with mRNA electroporation or AAV viral delivery, as well as incorporating suicide genes and safety switches, remain largely unexplored. A greater focus on implementing such strategies is required to enable successful translation of this therapy for T cell malignancies
Many advances have been made toward translating CAR therapy for the treatment of T cell malignancies. Both academia and industry are focused on the identification of tumor-specific antigens to enhance the safety and efficacy of CAR T cell products as well as on the development of superior cellular products. Unfortunately, due to vast variability in the design and execution of preclinical studies, it is often difficult to compare the different strategies. However, the numerous preclinical and clinical studies currently underway provide optimism for successful translation of this therapy to treat this aggressive and challenging group of diseases.
Availability of data and materials
Not applicable.
Abbreviations
- AAV:
-
Adeno-associated virus
- ADCC:
-
Antibody-dependent cellular cytotoxicity
- AITL:
-
Angioimmunoblastic T cell lymphoma
- ALCL:
-
Anaplastic large cell lymphoma
- AML:
-
Acute myeloid leukemia
- APC:
-
Antigen presenting cell
- ATLL:
-
Adult T cell leukemia/lymphoma
- B-ALL:
-
B cell acute lymphoblastic leukemia
- BCMA:
-
B cell maturation antigen
- CAR:
-
Chimeric antigen receptor
- cGMP:
-
Current good manufacturing process
- CRS:
-
Cytokine release syndrome
- CTCL:
-
Cutaneous T cell lymphoma
- DLBCL:
-
Diffuse large B cell lymphoma
- EATL:
-
Enteropathy-associated T cell lymphoma
- ENKTL:
-
Extranodal natural killer T cell lymphoma
- epCAM:
-
Epithelial cell adhesion molecule
- ETP-ALL:
-
Early T cell precursor acute lymphoblastic leukemia
- FDA:
-
Food and Drug Administration
- FPPS:
-
Farnesyl pyrophosphate synthase
- GvHD:
-
Graft versus host disease
- HER2:
-
Human epidermal growth factor receptor
- HLA:
-
Human leukocyte antigen
- HSCT:
-
Hematopoietic stem cell transplantation
- HSTCL:
-
Hepatosplenic T cell lymphoma
- HSV-TK:
-
Herpes simplex virus thymidine kinase
- HTLV1:
-
Human T cell lymphocytic virus type 1
- huEGFRt:
-
Truncated human epidermal growth factor receptor
- ICAM1:
-
Intercellular adhesion molecule 1
- iCas9:
-
Inducible Cas9
- IL-2:
-
Interleukin-2
- IPP:
-
Isopentenyl pyrophosphate
- KIR:
-
Killer-cell immunoglobulin-like receptor
- MCSP:
-
Melanoma-associated chondroitin sulfate proteoglycan
- MF:
-
Mycosis fungoides
- MHC:
-
Major histocompatibility complex
- MICA/B:
-
MHC class I chain-related protein A/B
- NK:
-
Natural killer
- NKG2D:
-
NK group 2 member D receptor
- NOS:
-
Not otherwise specified
- NSG:
-
NOD scid IL2Rγ-chain
- PD-1:
-
Programmed cell death receptor 1
- PDX:
-
Patient-derived xenograft
- PEBL:
-
Protein expression blocker
- PTCL:
-
Peripheral T cell lymphoma
- scFv:
-
Single-chain variable fragment
- SS:
-
Sezary syndrome
- TALEN:
-
Transcription activator-like effector nuclease
- T-ALL:
-
T cell acute lymphoblastic leukemia
- TCR:
-
T cell receptor
- TIL:
-
Tumor-infiltrating lymphocyte
- T-LGL:
-
T cell large granular lymphocytic leukemia
- T-LLy:
-
T-lymphoblastic lymphoma
- TNF:
-
Tumor necrosis factor
- TNFR:
-
Tumor necrosis factor receptor
- T-PLL:
-
T-prolymphocytic leukemia
- TRAC:
-
T cell receptor α constant
- TRAF:
-
TNF receptor-associated factor
- TRAIL:
-
TNF-related apoptosis-inducing ligand
- TRBC1:
-
T cell receptor beta constant 1
- ULBPs:
-
UL16 binding proteins
- VIP:
-
Vasoactive intestinal peptide
- γδ T cell:
-
Gamma delta T cell
References
Belver L, Ferrando A. The genetics and mechanisms of T cell acute lymphoblastic leukaemia. Nat Rev Cancer. 2016;16(8):494–507.
Hunger SP, Mullighan CG. Acute Lymphoblastic leukemia in children. N Engl J Med. 2015;373(16):1541–52.
Phillips AA, Harewood JCK. Adult T cell leukemia-lymphoma (ATL): State of the Art. Curr Hematol Malig Rep. 2018;13(4):300–7.
Utsunomiya A, Choi I, Chihara D, Seto M. Recent advances in the treatment of adult T-cell leukemia-lymphomas. Cancer Sci. 2015;106(4):344–51.
Matutes E. Adult T-cell leukaemia/lymphoma. J Clin Pathol. 2007;60(12):1373–7.
Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127(20):2375–90.
A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin's lymphoma. The Non-Hodgkin's Lymphoma Classification Project. Blood 1997;89(11):3909-3918.
Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin. 2016;66(6):443–59.
Laribi K, Alani M, Truong C. Baugier de Materre A. Recent advances in the treatment of peripheral T-cell lymphoma. Oncologist. 2018;23(9):1039–53.
Vose J, Armitage J, Weisenburger D, International TCLP. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26(25):4124–30.
Einsiedel HG, von Stackelberg A, Hartmann R, Fengler R, Schrappe M, Janka-Schaub G, et al. Long-term outcome in children with relapsed ALL by risk-stratified salvage therapy: results of trial acute lymphoblastic leukemia-relapse study of the Berlin-Frankfurt-Munster Group 87. J Clin Oncol. 2005;23(31):7942–50.
Reismuller B, Peters C, Dworzak MN, Potschger U, Urban C, Meister B, et al. Outcome of children and adolescents with a second or third relapse of acute lymphoblastic leukemia (ALL): a population-based analysis of the Austrian ALL-BFM (Berlin-Frankfurt-Munster) study group. J Pediatr Hematol Oncol. 2013;35(5):e200–4.
Winter SS, Dunsmore KP, Devidas M, Wood BL, Esiashvili N, Chen Z, et al. Improved survival for children and young adults with T-lineage acute lymphoblastic leukemia: results from the Children's Oncology Group AALL0434 Methotrexate Randomization. J Clin Oncol. 2018;36(29):2926–34.
Whittaker S, Hoppe R, Prince HM. How I treat mycosis fungoides and Sezary syndrome. Blood. 2016;127(25):3142–53.
Arulogun SO, Prince HM, Ng J, Lade S, Ryan GF, Blewitt O, et al. Long-term outcomes of patients with advanced-stage cutaneous T-cell lymphoma and large cell transformation. Blood. 2008;112(8):3082–7.
Horwitz S, O'Connor OA, Pro B, Illidge T, Fanale M, Advani R, et al. Brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma (ECHELON-2): a global, double-blind, randomised, phase 3 trial. Lancet. 2019;393(10168):229–40.
Prince HM, Kim YH, Horwitz SM, Dummer R, Scarisbrick J, Quaglino P, et al. Brentuximab vedotin or physician's choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA): an international, open-label, randomised, phase 3, multicentre trial. Lancet. 2017;390(10094):555–66.
Kwong YL, Chan TSY, Tan D, Kim SJ, Poon LM, Mow B, et al. PD1 blockade with pembrolizumab is highly effective in relapsed or refractory NK/T-cell lymphoma failing l-asparaginase. Blood. 2017;129(17):2437–42.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507–17.
Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K, et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med. 2014;6(224):224ra25.
Lee DW, Kochenderfer JN, Stetler-Stevenson M, Cui YK, Delbrook C, Feldman SA, et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385(9967):517–28.
Schubert ML, Huckelhoven A, Hoffmann JM, Schmitt A, Wuchter P, Sellner L, et al. Chimeric antigen receptor T cell therapy targeting CD19-positive leukemia and lymphoma in the context of stem cell transplantation. Hum Gene Ther. 2016.
Lim WA, June CH. The principles of engineering immune cells to treat cancer. Cell. 2017;168(4):724–40.
Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci U S A. 1989;86(24):10024–8.
Gill S, June CH. Going viral: chimeric antigen receptor T-cell therapy for hematological malignancies. Immunol Rev. 2015;263(1):68–89.
Kochenderfer JN, Dudley ME, Maric I, Feldman SA, Salit R, Hardy NM, et al. Dramatic regression of chronic lymphocytic leukemia in the first patient treated with donor-derived genetically-engineered anti-CD19-chimeric-antigen-receptor-expressing T cells after allogeneic hematopoietic stem cell transplantation. Biology of Blood and Marrow Transplantation. 2011;17(2).
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368(16):1509–18.
Levine BL, Miskin J, Wonnacott K, Keir C. Global Manufacturing of CAR T Cell Therapy. Mol Ther Methods Clin Dev. 2017;4:92–101.
Klebanoff CA, Khong HT, Antony PA, Palmer DC, Restifo NP. Sinks, suppressors and antigen presenters: how lymphodepletion enhances T cell-mediated tumor immunotherapy. Trends Immunol. 2005;26(2):111–7.
Laport GG, Levine BL, Stadtmauer EA, Schuster SJ, Luger SM, Grupp S, et al. Adoptive transfer of costimulated T cells induces lymphocytosis in patients with relapsed/refractory non-Hodgkin lymphoma following CD34+-selected hematopoietic cell transplantation. Blood. 2003;102(6):2004–13.
Dudley ME, Wunderlich JR, Yang JC, Sherry RM, Topalian SL, Restifo NP, et al. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. J Clin Oncol. 2005;23(10):2346–57.
Kalos M, Levine BL, Porter DL, Katz S, Grupp SA, Bagg A, et al. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci Transl Med. 2011;3(95):95ra73.
Park JH, Geyer MB, Brentjens RJ. CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood. 2016;127(26):3312–20.
Grupp SA, Maude SL, Shaw PA, Aplenc R, Barrett DM, Callahan C, et al. Durable remissions in children with relapsed/refractory ALL treated with t cells engineered with a CD19-Targeted chimeric antigen receptor (CTL019). Blood. 2015.
Topp MS, Gokbuget N, Zugmaier G, Klappers P, Stelljes M, Neumann S, et al. Phase II trial of the anti-CD19 bispecific T cell-engager blinatumomab shows hematologic and molecular remissions in patients with relapsed or refractory B-precursor acute lymphoblastic leukemia. J Clin Oncol. 2014;32(36):4134–40.
Maude SL, Teachey DT, Porter DL, Grupp SA. CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood. 2015;125(26):4017–23.
Gardner R, Wu D, Cherian S, Fang M, Hanafi LA, Finney O, et al. Acquisition of a CD19-negative myeloid phenotype allows immune escape of MLL-rearranged B-ALL from CD19 CAR-T-cell therapy. Blood. 2016;127(20):2406–10.
Hamieh M, Dobrin A, Cabriolu A, van der Stegen SJC, Giavridis T, Mansilla-Soto J, et al. CAR T cell trogocytosis and cooperative killing regulate tumour antigen escape. Nature. 2019;568(7750):112–6.
Ruella M, Xu J, Barrett DM, Fraietta JA, Reich TJ, Ambrose DE, et al. Induction of resistance to chimeric antigen receptor T cell therapy by transduction of a single leukemic B cell. Nat Med. 2018;24(10):1499–503.
Le RQ, Li L, Yuan W, Shord SS, Nie L, Habtemariam BA, et al. FDA Approval summary: Tocilizumab for treatment of chimeric antigen receptor T cell-induced severe or life-threatening cytokine release syndrome. Oncologist. 2018;23(8):943–7.
Gust J, Hay KA, Hanafi LA, Li D, Myerson D, Gonzalez-Cuyar LF, et al. Endothelial activation and blood-brain barrier disruption in neurotoxicity after adoptive immunotherapy with CD19 CAR-T Cells. Cancer Discov. 2017;7(12):1404–19.
Gust J, Taraseviciute A, Turtle CJ. Neurotoxicity associated with CD19-targeted CAR-T cell therapies. CNS Drugs. 2018;32(12):1091–101.
Hunter BD, Jacobson CA. CAR T-cell associated neurotoxicity: mechanisms, clinicopathologic correlates, and future directions. J Natl Cancer Inst. 2019.
Karschnia P, Jordan JT, Forst DA, Arrillaga-Romany IC, Batchelor TT, Baehring JM, et al. Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood. 2019;133(20):2212–21.
Gomes-Silva D, Srinivasan M, Sharma S, Lee CM, Wagner DL, Davis TH, et al. CD7-edited T cells expressing a CD7-specific CAR for the therapy of T-cell malignancies. Blood. 2017;130(3):285–96.
Cooper ML, Choi J, Staser K, Ritchey JK, Devenport JM, Eckardt K, et al. An "off-the-shelf" fratricide-resistant CAR-T for the treatment of T cell hematologic malignancies. Leukemia. 2018;32(9):1970–83.
Raikar SS, Fleischer LC, Moot R, Fedanov A, Paik NY, Knight KA, et al. Development of chimeric antigen receptors targeting T-cell malignancies using two structurally different anti-CD5 antigen binding domains in NK and CRISPR-edited T cell lines. Oncoimmunology. 2018;7(3):e1407898.
Mamonkin M, Mukherjee M, Srinivasan M, Sharma S, Gomes-Silva D, Mo F, et al. Reversible transgene expression reduces fratricide and permits 4-1BB costimulation of CAR T cells directed to T-cell malignancies. Cancer Immunol Res. 2018;6(1):47–58.
Png YT, Vinanica N, Kamiya T, Shimasaki N, Coustan-Smith E, Campana D. Blockade of CD7 expression in T cells for effective chimeric antigen receptor targeting of T-cell malignancies. Blood Adv. 2017;1(25):2348–60.
Kamiya T, Wong D, Png YT, Campana D. A novel method to generate T-cell receptor-deficient chimeric antigen receptor T cells. Blood Adv. 2018;2(5):517–28.
Moot R, Raikar SS, Fleischer L, Querrey M, Tylawsky DE, Nakahara H, et al. Genetic engineering of chimeric antigen receptors using lamprey derived variable lymphocyte receptors. Mol Ther Oncolytics. 2016;3:16026.
Chen KH, Wada M, Firor AE, Pinz KG, Jares A, Liu H, et al. Novel anti-CD3 chimeric antigen receptor targeting of aggressive T cell malignancies. Oncotarget. 2016;7(35):56219–32.
Chen KH, Wada M, Pinz KG, Liu H, Lin KW, Jares A, et al. Preclinical targeting of aggressive T-cell malignancies using anti-CD5 chimeric antigen receptor. Leukemia. 2017;31(10):2151–60.
Pinz KG, Yakaboski E, Jares A, Liu H, Firor AE, Chen KH, et al. Targeting T-cell malignancies using anti-CD4 CAR NK-92 cells. Oncotarget. 2017;8(68):112783–96.
Ramos CA, Ballard B, Zhang H, Dakhova O, Gee AP, Mei Z, et al. Clinical and immunological responses after CD30-specific chimeric antigen receptor-redirected lymphocytes. J Clin Invest. 2017;127(9):3462–71.
Scarfo I, Ormhoj M, Frigault MJ, Castano AP, Lorrey S, Bouffard AA, et al. Anti-CD37 chimeric antigen receptor T cells are active against B- and T-cell lymphomas. Blood. 2018;132(14):1495–506.
Sanchez-Martinez D, Baroni ML, Gutierrez-Aguera F, Roca-Ho H, Blanch-Lombarte O, Gonzalez-Garcia S, et al. Fratricide-resistant CD1a-specific CAR T cells for the treatment of cortical T-cell acute lymphoblastic leukemia. Blood. 2019;133(21):2291–304.
Maciocia PM, Wawrzyniecka PA, Philip B, Ricciardelli I, Akarca AU, Onuoha SC, et al. Targeting the T cell receptor beta-chain constant region for immunotherapy of T cell malignancies. Nat Med. 2017;23(12):1416–23.
Mamonkin M, Rouce RH, Tashiro H, Brenner MK. A T-cell-directed chimeric antigen receptor for the selective treatment of T-cell malignancies. Blood. 2015;126(8):983–92.
You F, Wang Y, Jiang L, Zhu X, Chen D, Yuan L, et al. A novel CD7 chimeric antigen receptor-modified NK-92MI cell line targeting T-cell acute lymphoblastic leukemia. Am J Cancer Res. 2019;9(1):64–78.
Rasaiyaah J, Georgiadis C, Preece R, Mock U, Qasim W. TCRalphabeta/CD3 disruption enables CD3-specific antileukemic T cell immunotherapy. JCI Insight. 2018;3(13).
Park SI, Serody JS, Shea TC, Grover NS, Ivanova A, Morrison K, et al. A phase 1b/2 study of CD30-specific chimeric antigen receptor T-cell (CAR-T) therapy in combination with bendamustine in patients with CD30+ Hodgkin and non-Hodgkin lymphoma. J Clin Oncol. 2017;35(15_suppl):TPS3095-TPS.
Wang CM, Wu ZQ, Wang Y, Guo YL, Dai HR, Wang XH, et al. Autologous T cells expressing CD30 Chimeric antigen receptors for relapsed or refractory hodgkin lymphoma: an open-label phase I trial. Clin Cancer Res. 2017;23(5):1156–66.
Ramos CA, Bilgi M, Gerken C, Dakhova O, Mei Z, Wu M-F, et al. CD30-Chimeric Antigen receptor (CAR) T cells for therapy of hodgkin lymphoma (HL). Biol Blood Marrow Transplant. 2019;25(3):S63.
Antin JH, Emerson SG, Martin P, Gadol N, Ault KA. Leu-1+ (CD5+) B cells. A major lymphoid subpopulation in human fetal spleen: phenotypic and functional studies. J Immunol. 1986;136(2):505–10.
Thomas Y, Glickman E, DeMartino J, Wang J, Goldstein G, Chess L. Biologic functions of the OKT1 T cell surface antigen. I. The T1 molecule is involved in helper function. J Immunol. 1984;133(2):724–8.
Engleman EG, Warnke R, Fox RI, Dilley J, Benike CJ, Levy R. Studies of a human T lymphocyte antigen recognized by a monoclonal antibody. Proc Natl Acad Sci U S A. 1981;78(3):1791–5.
Martin PJ, Hansen JA, Nowinski RC, Brown MA. A new human T-cell differentiation antigen: unexpected expression on chronic lymphocytic leukemia cells. Immunogenetics. 1980;11(5):429–39.
Reinherz EL, Kung PC, Goldstein G, Schlossman SF. A monoclonal antibody with selective reactivity with functionally mature human thymocytes and all peripheral human T cells. J Immunol. 1979;123(3):1312–7.
Azzam HS, Grinberg A, Lui K, Shen H, Shores EW, Love PE. CD5 expression is developmentally regulated by T cell receptor (TCR) signals and TCR avidity. J Exp Med. 1998;188(12):2301–11.
Dalloul A. CD5: a safeguard against autoimmunity and a shield for cancer cells. Autoimmun Rev. 2009;8(4):349–53.
Pui CH, Behm FG, Crist WM. Clinical and biologic relevance of immunologic marker studies in childhood acute lymphoblastic leukemia. Blood. 1993;82(2):343–62.
Patel JL, Smith LM, Anderson J, Abromowitch M, Campana D, Jacobsen J, et al. The immunophenotype of T-lymphoblastic lymphoma in children and adolescents: a Children's Oncology Group report. Br J Haematol. 2012;159(4):454–61.
Campana D, van Dongen JJ, Mehta A, Coustan-Smith E, Wolvers-Tettero IL, Ganeshaguru K, et al. Stages of T-cell receptor protein expression in T-cell acute lymphoblastic leukemia. Blood. 1991;77(7):1546–54.
Sigal LH. Basic science for the clinician 54: CD5. J Clin Rheumatol. 2012;18(2):83–8.
Hillerdal V, Boura VF, Bjorkelund H, Andersson K, Essand M. Avidity characterization of genetically engineered T-cells with novel and established approaches. BMC Immunol. 2016;17(1):23.
Liu X, Jiang S, Fang C, Yang S, Olalere D, Pequignot EC, et al. Affinity-Tuned ErbB2 or EGFR Chimeric antigen receptor T cells exhibit an increased therapeutic index against tumors in mice. Cancer Res. 2015;75(17):3596–607.
Dorothee G, Vergnon I, El Hage F, Le Maux CB, Ferrand V, Lecluse Y, et al. In situ sensory adaptation of tumor-infiltrating T lymphocytes to peptide-MHC levels elicits strong antitumor reactivity. J Immunol. 2005;174(11):6888–97.
Bertram JH, Gill PS, Levine AM, Boquiren D, Hoffman FM, Meyer P, et al. Monoclonal antibody T101 in T cell malignancies: a clinical, pharmacokinetic, and immunologic correlation. Blood. 1986;68(3):752–61.
LeMaistre CF, Rosen S, Frankel A, Kornfeld S, Saria E, Meneghetti C, et al. Phase I trial of H65-RTA immunoconjugate in patients with cutaneous T-cell lymphoma. Blood. 1991;78(5):1173–82.
Petersen CT, Hassan M, Morris AB, Jeffery J, Lee K, Jagirdar N, et al. Improving T-cell expansion and function for adoptive T-cell therapy using ex vivo treatment with PI3Kdelta inhibitors and VIP antagonists. Blood Adv. 2018;2(3):210–23.
Xu Y, Liu Q, Zhong M, Wang Z, Chen Z, Zhang Y, et al. 2B4 costimulatory domain enhancing cytotoxic ability of anti-CD5 chimeric antigen receptor engineered natural killer cells against T cell malignancies. J Hematol Oncol. 2019;12(1):49.
Rabinowich H, Pricop L, Herberman RB, Whiteside TL. Expression and function of CD7 molecule on human natural killer cells. J Immunol. 1994;152(2):517–26.
Coustan-Smith E, Mullighan CG, Onciu M, Behm FG, Raimondi SC, Pei D, et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol. 2009;10(2):147–56.
Zhang J, Ding L, Holmfeldt L, Wu G, Heatley SL, Payne-Turner D, et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature. 2012;481(7380):157–63.
Inukai T, Kiyokawa N, Campana D, Coustan-Smith E, Kikuchi A, Kobayashi M, et al. Clinical significance of early T-cell precursor acute lymphoblastic leukaemia: results of the Tokyo Children's Cancer Study Group Study L99-15. Br J Haematol. 2012;156(3):358–65.
Ma G, Shen J, Pinz K, Wada M, Park J, Kim S, et al. Targeting T cell malignancies using CD4CAR T-cells and implementing a natural safety switch. Stem Cell Rev. 2019;15(3):443–7.
Lapalombella R, Yeh YY, Wang L, Ramanunni A, Rafiq S, Jha S, et al. Tetraspanin CD37 directly mediates transduction of survival and apoptotic signals. Cancer Cell. 2012;21(5):694–708.
van Spriel AB, Puls KL, Sofi M, Pouniotis D, Hochrein H, Orinska Z, et al. A regulatory role for CD37 in T cell proliferation. J Immunol. 2004;172(5):2953–61.
Barrena S, Almeida J, Yunta M, Lopez A, Fernandez-Mosteirin N, Giralt M, et al. Aberrant expression of tetraspanin molecules in B-cell chronic lymphoproliferative disorders and its correlation with normal B-cell maturation. Leukemia. 2005;19(8):1376–83.
Pereira DS, Guevara CI, ** L, Mbong N, Verlinsky A, Hsu SJ, et al. AGS67E, an Anti-CD37 monomethyl auristatin E antibody-drug conjugate as a potential therapeutic for B/T-cell malignancies and AML: a new role for CD37 in AML. Mol Cancer Ther. 2015;14(7):1650–60.
Dürkop H, Latza U, Hummel M, Eitelbach F, Seed B, Stein H. Molecular cloning and expression of a new member of the nerve growth factor receptor family that is characteristic for Hodgkin's disease. Cell. 1992;68(3):421–7.
Pierce JM, Mehta A. Diagnostic, prognostic and therapeutic role of CD30 in lymphoma. Expert Rev Hematol. 2017;10(1):29–37.
Stein H, Mason DY, Gerdes J, O'Connor N, Wainscoat J, Pallesen G, et al. The expression of the Hodgkin's disease associated antigen Ki-1 in reactive and neoplastic lymphoid tissue: evidence that Reed-Sternberg cells and histiocytic malignancies are derived from activated lymphoid cells. Blood. 1985;66(4):848–58.
Ellis TM, Simms PE, Slivnick DJ, Jack HM, Fisher RI. CD30 is a signal-transducing molecule that defines a subset of human activated CD45RO+ T cells. J Immunol. 1993;151(5):2380–9.
Zheng W, Medeiros LJ, Young KH, Goswami M, Powers L, Kantarjian HH, et al. CD30 expression in acute lymphoblastic leukemia as assessed by flow cytometry analysis. Leuk Lymphoma. 2014;55(3):624–7.
Hombach A, Heuser C, Sircar R, Tillmann T, Diehl V, Pohl C, et al. An anti-CD30 chimeric receptor that mediates CD3-zeta-independent T-cell activation against Hodgkin's lymphoma cells in the presence of soluble CD30. Cancer Res. 1998;58(6):1116–9.
Hombach A, Heuser C, Sircar R, Tillmann T, Diehl V, Pohl C, et al. Characterization of a chimeric T-cell receptor with specificity for the Hodgkin's lymphoma-associated CD30 antigen. J Immunother. 1999;22(6):473–80.
Sims JE, Tunnacliffe A, Smith WJ, Rabbitts TH. Complexity of human T-cell antigen receptor beta-chain constant- and variable-region genes. Nature. 1984;312(5994):541–5.
Tunnacliffe A, Kefford R, Milstein C, Forster A, Rabbitts TH. Sequence and evolution of the human T-cell antigen receptor beta-chain genes. Proc Natl Acad Sci U S A. 1985;82(15):5068–72.
Went P, Agostinelli C, Gallamini A, Piccaluga PP, Ascani S, Sabattini E, et al. Marker expression in peripheral T-cell lymphoma: a proposed clinical-pathologic prognostic score. J Clin Oncol. 2006;24(16):2472–9.
Pui CH, Behm FG, Singh B, Schell MJ, Williams DL, Rivera GK, et al. Heterogeneity of presenting features and their relation to treatment outcome in 120 children with T-cell acute lymphoblastic leukemia. Blood. 1990;75(1):174–9.
Bechan GI, Lee DW, Zajonc DM, Heckel D, **an R, Throsby M, et al. Phage display generation of a novel human anti-CD1A monoclonal antibody with potent cytolytic activity. Br J Haematol. 2012;159(3):299–310.
Carrera Silva EA, Nowak W, Tessone L, Olexen CM, Ortiz Wilczynski JM, Estecho IG, et al. CD207(+)CD1a(+) cells circulate in pediatric patients with active Langerhans cell histiocytosis. Blood. 2017;130(17):1898–902.
Niehues T, Kapaun P, Harms DO, Burdach S, Kramm C, Korholz D, et al. A classification based on T cell selection-related phenotypes identifies a subgroup of childhood T-ALL with favorable outcome in the COALL studies. Leukemia. 1999;13(4):614–7.
van Grotel M, Meijerink JP, van Wering ER, Langerak AW, Beverloo HB, Buijs-Gladdines JG, et al. Prognostic significance of molecular-cytogenetic abnormalities in pediatric T-ALL is not explained by immunophenotypic differences. Leukemia. 2008;22(1):124–31.
Das RK, Vernau L, Grupp SA, Barrett DM. Naive T-cell deficits at diagnosis and after chemotherapy impair cell therapy potential in pediatric cancers. Cancer Discov. 2019;9(4):492–9.
Rudolph ME, McArthur MA, Barnes RS, Magder LS, Chen WH, Sztein MB. Differences between pediatric and adult T cell responses to in vitro Staphylococcal enterotoxin B stimulation. Front Immunol. 2018;9:498.
Thome JJ, Grinshpun B, Kumar BV, Kubota M, Ohmura Y, Lerner H, et al. Longterm maintenance of human naive T cells through in situ homeostasis in lymphoid tissue sites. Sci Immunol. 2016;1(6).
Shearer WT, Rosenblatt HM, Gelman RS, Oyomopito R, Plaeger S, Stiehm ER, et al. Lymphocyte subsets in healthy children from birth through 18 years of age: the Pediatric AIDS Clinical Trials Group P1009 study. J Allergy Clin Immunol. 2003;112(5):973–80.
Qasim W, Zhan H, Samarasinghe S, Adams S, Amrolia P, Stafford S, et al. Molecular remission of infant B-ALL after infusion of universal TALEN gene-edited. CAR T cells. Sci Transl Med. 2017;9(374).
McCreedy BJ, Senyukov VV, Nguyen KT. Off the shelf T cell therapies for hematologic malignancies. Best Pract Res Clin Haematol. 2018;31(2):166–75.
Brehm C, Huenecke S, Quaiser A, Esser R, Bremm M, Kloess S, et al. IL-2 stimulated but not unstimulated NK cells induce selective disappearance of peripheral blood cells: concomitant results to a phase I/II study. PLoS One. 2011;6(11):e27351.
Glienke W, Esser R, Priesner C, Suerth JD, Schambach A, Wels WS, et al. Advantages and applications of CAR-expressing natural killer cells. Front Pharmacol. 2015;6:21.
Gong JH, Maki G, Klingemann HG. Characterization of a human cell line (NK-92) with phenotypical and functional characteristics of activated natural killer cells. Leukemia. 1994;8(4):652–8.
Boissel L, Betancur M, Lu W, Wels WS, Marino T, Van Etten RA, et al. Comparison of mRNA and lentiviral based transfection of natural killer cells with chimeric antigen receptors recognizing lymphoid antigens. Leuk Lymphoma. 2012;53(5):958–65.
Boissel L, Betancur M, Wels WS, Tuncer H, Klingemann H. Transfection with mRNA for CD19 specific chimeric antigen receptor restores NK cell mediated killing of CLL cells. Leuk Res. 2009;33(9):1255–9.
Boissel L, Betancur-Boissel M, Lu W, Krause DS, Van Etten RA, Wels WS, et al. Retargeting NK-92 cells by means of CD19- and CD20-specific chimeric antigen receptors compares favorably with antibody-dependent cellular cytotoxicity. Oncoimmunology. 2013;2(10):e26527.
Muller T, Uherek C, Maki G, Chow KU, Schimpf A, Klingemann HG, et al. Expression of a CD20-specific chimeric antigen receptor enhances cytotoxic activity of NK cells and overcomes NK-resistance of lymphoma and leukemia cells. Cancer Immunol Immunother. 2008;57(3):411–23.
Chu J, Deng Y, Benson DM, He S, Hughes T, Zhang J, et al. CS1-specific chimeric antigen receptor (CAR)-engineered natural killer cells enhance in vitro and in vivo antitumor activity against human multiple myeloma. Leukemia. 2014;28(4):917–27.
Rafiq S, Purdon TJ, Schultz L, Klingemann H, Brentjens RJ. NK-92 cells engineered with anti-CD33 chimeric antigen receptors (CAR) for the treatment of Acute Myeloid Leukemia (AML). Cytotherapy. 2015;17(6):S23.
Sahm C, Schonfeld K, Wels WS. Expression of IL-15 in NK cells results in rapid enrichment and selective cytotoxicity of gene-modified effectors that carry a tumor-specific antigen receptor. Cancer Immunol Immunother. 2012;61(9):1451–61.
Schonfeld K, Sahm C, Zhang C, Naundorf S, Brendel C, Odendahl M, et al. Selective inhibition of tumor growth by clonal NK cells expressing an ErbB2/HER2-specific chimeric antigen receptor. Mol Ther. 2015;23(2):330–8.
Seidel D, Shibina A, Siebert N, Wels WS, Reynolds CP, Huebener N, et al. Disialoganglioside-specific human natural killer cells are effective against drug-resistant neuroblastoma. Cancer Immunol Immunother. 2015;64(5):621–34.
Han J, Chu J, Keung Chan W, Zhang J, Wang Y, Cohen JB, et al. CAR-Engineered NK Cells targeting wild-type EGFR and EGFRvIII enhance killing of glioblastoma and patient-derived glioblastoma stem cells. Sci Rep. 2015;5:11483.
Arai S, Meagher R, Swearingen M, Myint H, Rich E, Martinson J, et al. Infusion of the allogeneic cell line NK-92 in patients with advanced renal cell cancer or melanoma: a phase I trial. Cytotherapy. 2008;10(6):625–32.
Tonn T, Schwabe D, Klingemann HG, Becker S, Esser R, Koehl U, et al. Treatment of patients with advanced cancer with the natural killer cell line NK-92. Cytotherapy. 2013;15(12):1563–70.
Suck G, Odendahl M, Nowakowska P, Seidl C, Wels WS, Klingemann HG, et al. NK-92: an 'off-the-shelf therapeutic' for adoptive natural killer cell-based cancer immunotherapy. Cancer Immunol Immunother. 2016;65(4):485–92.
Tang X, Yang L, Li Z, Nalin AP, Dai H, Xu T, et al. First-in-man clinical trial of CAR NK-92 cells: safety test of CD33-CAR NK-92 cells in patients with relapsed and refractory acute myeloid leukemia. Am J Cancer Res. 2018;8(6):1083–9.
Farag SS, Fehniger TA, Ruggeri L, Velardi A, Caligiuri MA. Natural killer cell receptors: new biology and insights into the graft-versus-leukemia effect. Blood. 2002;100(6):1935–47.
Farag SS, Caligiuri MA. Human natural killer cell development and biology. Blood Rev. 2006;20(3):123–37.
Klingemann H, Boissel L, Toneguzzo F. Natural killer cells for immunotherapy—advantages of the NK-92 Cell Line over Blood NK Cells. Front Immunol. 2016;7:91.
Maki G, Klingemann HG, Martinson JA, Tam YK. Factors regulating the cytotoxic activity of the human natural killer cell line, NK-92. J Hematother Stem Cell Res. 2001;10(3):369–83.
Klingemann H. Are natural killer cells superior CAR drivers? Oncoimmunology. 2014;3:e28147.
Voskoboinik I, Smyth MJ, Trapani JA. Perforin-mediated target-cell death and immune homeostasis. Nat Rev Immunol. 2006;6(12):940–52.
Screpanti V, Wallin RP, Ljunggren HG, Grandien A. A central role for death receptor-mediated apoptosis in the rejection of tumors by NK cells. J Immunol. 2001;167(4):2068–73.
Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol. 2008;9(5):503–10.
Vivier E, Raulet DH, Moretta A, Caligiuri MA, Zitvogel L, Lanier LL, et al. Innate or adaptive immunity? The example of natural killer cells. Science. 2011;331(6013):44–9.
Bhat R, Watzl C. Serial killing of tumor cells by human natural killer cells—enhancement by therapeutic antibodies. PLoS One. 2007;2(3):e326.
Zhang Y, Wallace DL, de Lara CM, Ghattas H, Asquith B, Worth A, et al. In vivo kinetics of human natural killer cells: the effects of ageing and acute and chronic viral infection. Immunology. 2007;121(2):258–65.
Klingemann H. Challenges of cancer therapy with natural killer cells. Cytotherapy. 2015;17(3):245–9.
Iyengar R, Handgretinger R, Babarin-Dorner A, Leimig T, Otto M, Geiger TL, et al. Purification of human natural killer cells using a clinical-scale immunomagnetic method. Cytotherapy. 2003;5(6):479–84.
Koehl U, Brehm C, Huenecke S, Zimmermann SY, Kloess S, Bremm M, et al. Clinical grade purification and expansion of NK cell products for an optimized manufacturing protocol. Front Oncol. 2013;3:118.
Sutlu T, Stellan B, Gilljam M, Quezada HC, Nahi H, Gahrton G, et al. Clinical-grade, large-scale, feeder-free expansion of highly active human natural killer cells for adoptive immunotherapy using an automated bioreactor. Cytotherapy. 2010;12(8):1044–55.
Angelo LS, Banerjee PP, Monaco-Shawver L, Rosen JB, Makedonas G, Forbes LR, et al. Practical NK cell phenoty** and variability in healthy adults. Immunol Res. 2015;62(3):341–56.
Kweon S, Phan MT, Chun S, Yu H, Kim J, Kim S, et al. Expansion of Human NK Cells Using K562 Cells Expressing OX40 Ligand and Short Exposure to IL-21. Front Immunol. 2019;10:879.
Pardoll DM, Fowlkes BJ, Bluestone JA, Kruisbeek A, Maloy WL, Coligan JE, et al. Differential expression of two distinct T-cell receptors during thymocyte development. Nature. 1987;326(6108):79–81.
Born WK, Reardon CL, O'Brien RL. The function of gammadelta T cells in innate immunity. Curr Opin Immunol. 2006;18(1):31–8.
Chien YH, Jores R, Crowley MP. Recognition by gamma/delta T cells. Annu Rev Immunol. 1996;14:511–32.
Schild H, Mavaddat N, Litzenberger C, Ehrich EW, Davis MM, Bluestone JA, et al. The nature of major histocompatibility complex recognition by γδ T cells. Cell. 1994;76(1):29–37.
Sutton KS, Dasgupta A, McCarty D, Doering CB, Spencer HT. Bioengineering and serum free expansion of blood-derived gammadelta T cells. Cytotherapy. 2016;18(7):881–92.
Finkelshtein D, Werman A, Novick D, Barak S, Rubinstein M. LDL receptor and its family members serve as the cellular receptors for vesicular stomatitis virus. Proc Natl Acad Sci U S A. 2013;110(18):7306–11.
Capsomidis A, Benthall G, Van Acker HH, Fisher J, Kramer AM, Abeln Z, et al. Chimeric antigen receptor-engineered human gamma delta T cells: enhanced cytotoxicity with retention of Cross Presentation. Mol Ther. 2018;26(2):354–65.
Rischer M, Pscherer S, Duwe S, Vormoor J, Jurgens H, Rossig C. Human gammadelta T cells as mediators of chimaeric-receptor redirected anti-tumour immunity. Br J Haematol. 2004;126(4):583–92.
Harrer DC, Simon B, Fujii SI, Shimizu K, Uslu U, Schuler G, et al. RNA-transfection of gamma/delta T cells with a chimeric antigen receptor or an alpha/beta T-cell receptor: a safer alternative to genetically engineered alpha/beta T cells for the immunotherapy of melanoma. BMC Cancer. 2017;17(1):551.
Deniger DC, Switzer K, Mi T, Maiti S, Hurton L, Singh H, et al. Bispecific T-cells expressing polyclonal repertoire of endogenous gammadelta T-cell receptors and introduced CD19-specific chimeric antigen receptor. Mol Ther. 2013;21(3):638–47.
**ao L, Chen C, Li Z, Zhu S, Tay JC, Zhang X, et al. Large-scale expansion of Vgamma9Vdelta2 T cells with engineered K562 feeder cells in G-Rex vessels and their use as chimeric antigen receptor-modified effector cells. Cytotherapy. 2018;20(3):420–35.
D'Asaro M, La Mendola C, Di Liberto D, Orlando V, Todaro M, Spina M, et al. V gamma 9V delta 2 T lymphocytes efficiently recognize and kill zoledronate-sensitized, imatinib-sensitive, and imatinib-resistant chronic myelogenous leukemia cells. J Immunol. 2010;184(6):3260–8.
Chargui J, Combaret V, Scaglione V, Iacono I, Peri V, Valteau-Couanet D, et al. Bromohydrin pyrophosphate-stimulated Vgamma9delta2 T cells expanded ex vivo from patients with poor-prognosis neuroblastoma lyse autologous primary tumor cells. J Immunother. 2010;33(6):591–8.
Todaro M, D'Asaro M, Caccamo N, Iovino F, Francipane MG, Meraviglia S, et al. Efficient killing of human colon cancer stem cells by gammadelta T lymphocytes. J Immunol. 2009;182(11):7287–96.
Zoine JT, Knight KA, Fleischer LC, Sutton KS, Goldsmith KC, Doering CB, et al. Ex vivo expanded patient-derived γδ T-cell immunotherapy enhances neuroblastoma tumor regression in a murine model. OncoImmunology. 2019;8(8):1593804.
Prinz I. Dynamics of the interaction of gammadelta T cells with their neighbors in vivo. Cell Mol Life Sci. 2011;68(14):2391–8.
Meissner N, Radke J, Hedges JF, White M, Behnke M, Bertolino S, et al. Serial analysis of gene expression in circulating gamma delta T cell subsets defines distinct immunoregulatory phenotypes and unexpected gene expression profiles. J Immunol. 2003;170(1):356–64.
Urban EM, Chapoval AI, Pauza CD. Repertoire development and the control of cytotoxic/effector function in human gammadelta T cells. Clin Dev Immunol. 2010;2010:732893.
Gallucci S, Matzinger P. Danger signals: SOS to the immune system. Curr Opin Immunol. 2001;13(1):114–9.
Rincon-Orozco B, Kunzmann V, Wrobel P, Kabelitz D, Steinle A, Herrmann T. Activation of V gamma 9V delta 2 T cells by NKG2D. J Immunol. 2005;175(4):2144–51.
Lanca T, Correia DV, Moita CF, Raquel H, Neves-Costa A, Ferreira C, et al. The MHC class Ib protein ULBP1 is a nonredundant determinant of leukemia/lymphoma susceptibility to gammadelta T-cell cytotoxicity. Blood. 2010;115(12):2407–11.
Kabelitz D, Wesch D. Features and functions of gamma delta T lymphocytes: focus on chemokines and their receptors. Crit Rev Immunol. 2003;23(5-6):339–70.
Li B, Bassiri H, Rossman MD, Kramer P, Eyuboglu AF, Torres M, et al. Involvement of the Fas/Fas ligand pathway in activation-induced cell death of mycobacteria-reactive human gamma delta T cells: a mechanism for the loss of gamma delta T cells in patients with pulmonary tuberculosis. J Immunol. 1998;161(3):1558–67.
Gogoi D, Chiplunkar SV. Targeting gamma delta T cells for cancer immunotherapy: bench to bedside. Indian J Med Res. 2013;138(5):755–61.
Gober HJ, Kistowska M, Angman L, Jeno P, Mori L, De Libero G. Human T cell receptor gammadelta cells recognize endogenous mevalonate metabolites in tumor cells. J Exp Med. 2003;197(2):163–8.
Uchida R, Ashihara E, Sato K, Kimura S, Kuroda J, Takeuchi M, et al. Gamma delta T cells kill myeloma cells by sensing mevalonate metabolites and ICAM-1 molecules on cell surface. Biochem Biophys Res Commun. 2007;354(2):613–8.
Li J, Herold MJ, Kimmel B, Muller I, Rincon-Orozco B, Kunzmann V, et al. Reduced expression of the mevalonate pathway enzyme farnesyl pyrophosphate synthase unveils recognition of tumor cells by Vgamma9Vdelta2 T cells. J Immunol. 2009;182(12):8118–24.
Zheng BJ, Chan KW, Im S, Chua D, Sham JS, Tin PC, et al. Anti-tumor effects of human peripheral gammadelta T cells in a mouse tumor model. Int J Cancer. 2001;92(3):421–5.
Bennouna J, Bompas E, Neidhardt EM, Rolland F, Philip I, Galea C, et al. Phase-I study of Innacell gammadelta, an autologous cell-therapy product highly enriched in gamma9delta2 T lymphocytes, in combination with IL-2, in patients with metastatic renal cell carcinoma. Cancer Immunol Immunother. 2008;57(11):1599–609.
Nakajima J, Murakawa T, Fukami T, Goto S, Kaneko T, Yoshida Y, et al. A phase I study of adoptive immunotherapy for recurrent non-small-cell lung cancer patients with autologous gammadelta T cells. Eur J Cardiothorac Surg. 2010;37(5):1191–7.
Meraviglia S, Eberl M, Vermijlen D, Todaro M, Buccheri S, Cicero G, et al. In vivo manipulation of Vgamma9Vdelta2 T cells with zoledronate and low-dose interleukin-2 for immunotherapy of advanced breast cancer patients. Clin Exp Immunol. 2010;161(2):290–7.
Nicol AJ, Tokuyama H, Mattarollo SR, Hagi T, Suzuki K, Yokokawa K, et al. Clinical evaluation of autologous gamma delta T cell-based immunotherapy for metastatic solid tumours. Br J Cancer. 2011;105(6):778–86.
Cavazzana-Calvo M, Payen E, Negre O, Wang G, Hehir K, Fusil F, et al. Transfusion independence and HMGA2 activation after gene therapy of human beta-thalassaemia. Nature. 2010;467(7313):318–22.
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack MP, Wulffraat N, Leboulch P, et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science. 2003;302(5644):415–9.
Merten OW, Hebben M, Bovolenta C. Production of lentiviral vectors. Mol Ther Methods Clin Dev. 2016;3:16017.
Kumar M, Keller B, Makalou N, Sutton RE. Systematic determination of the packaging limit of lentiviral vectors. Hum Gene Ther. 2001;12(15):1893–905.
al Yacoub N, Romanowska M, Haritonova N, Foerster J. Optimized production and concentration of lentiviral vectors containing large inserts. J Gene Med. 2007;9(7):579-584.
Hacein-Bey-Abina S, Garrigue A, Wang GP, Soulier J, Lim A, Morillon E, et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J Clin Invest. 2008;118(9):3132–42.
Braun CJ, Boztug K, Paruzynski A, Witzel M, Schwarzer A, Rothe M, et al. Gene therapy for Wiskott-Aldrich syndrome—long-term efficacy and genotoxicity. Sci Transl Med. 2014;6(227):227ra33.
Barrett DM, Zhao Y, Liu X, Jiang S, Carpenito C, Kalos M, et al. Treatment of advanced leukemia in mice with mRNA engineered T cells. Hum Gene Ther. 2011;22(12):1575–86.
Yoon SH, Lee JM, Cho HI, Kim EK, Kim HS, Park MY, et al. Adoptive immunotherapy using human peripheral blood lymphocytes transferred with RNA encoding Her-2/neu-specific chimeric immune receptor in ovarian cancer xenograft model. Cancer Gene Ther. 2009;16(6):489–97.
Beatty GL, Haas AR, Maus MV, Torigian DA, Soulen MC, Plesa G, et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol Res. 2014;2(2):112–20.
Svoboda J, Rheingold SR, Gill SI, Grupp SA, Lacey SF, Kulikovskaya I, et al. Nonviral RNA chimeric antigen receptor-modified T cells in patients with Hodgkin lymphoma. Blood. 2018;132(10):1022–6.
Naso MF, Tomkowicz B, Perry WL 3rd, Strohl WR. Adeno-Associated Virus (AAV) as a Vector for Gene Therapy. BioDrugs. 2017;31(4):317–34.
Penaud-Budloo M, Le Guiner C, Nowrouzi A, Toromanoff A, Cherel Y, Chenuaud P, et al. Adeno-associated virus vector genomes persist as episomal chromatin in primate muscle. J Virol. 2008;82(16):7875–85.
Duan D, Sharma P, Yang J, Yue Y, Dudas L, Zhang Y, et al. Circular intermediates of recombinant adeno-associated virus have defined structural characteristics responsible for long-term episomal persistence in muscle tissue. J Virol. 1999;72(11):8568–77.
Wu P, Phillips MI, Bui J, Terwilliger EF. Adeno-associated virus vector-mediated transgene integration into neurons and other nondividing cell targets. J Virol. 1998;72(7):5919–26.
Smith RH. Adeno-associated virus integration: virus versus vector. Gene Ther. 2008;15(11):817–22.
Li H, Malani N, Hamilton SR, Schlachterman A, Bussadori G, Edmonson SE, et al. Assessing the potential for AAV vector genotoxicity in a murine model. Blood. 2011;117(12):3311–9.
Mietzsch M, Broecker F, Reinhardt A, Seeberger PH, Heilbronn R. Differential adeno-associated virus serotype-specific interaction patterns with synthetic heparins and other glycans. J Virol. 2014;88(5):2991–3003.
Agbandje-McKenna M, Kleinschmidt J. AAV capsid structure and cell interactions. Methods Mol Biol. 2011;807:47–92.
Song L, Li X, Jayandharan GR, Wang Y, Aslanidi GV, Ling C, et al. High-efficiency transduction of primary human hematopoietic stem cells and erythroid lineage-restricted expression by optimized AAV6 serotype vectors in vitro and in a murine xenograft model in vivo. PLoS One. 2013;8(3):e58757.
Song L, Kauss MA, Kopin E, Chandra M, Ul-Hasan T, Miller E, et al. Optimizing the transduction efficiency of capsid-modified AAV6 serotype vectors in primary human hematopoietic stem cells in vitro and in a xenograft mouse model in vivo. Cytotherapy. 2013;15(8):986–98.
Zincarelli C, Soltys S, Rengo G, Rabinowitz JE. Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol Ther. 2008;16(6):1073–80.
Ciceri F, Bonini C, Marktel S, Zappone E, Servida P, Bernardi M, et al. Antitumor effects of HSV-TK-engineered donor lymphocytes after allogeneic stem-cell transplantation. Blood. 2007;109(11):4698–707.
Tiberghien P, Ferrand C, Lioure B, Milpied N, Angonin R, Deconinck E, et al. Administration of herpes simplex-thymidine kinase-expressing donor T cells with a T-cell-depleted allogeneic marrow graft. Blood. 2001;97(1):63–72.
Bonini C, Ferrari G, Verzeletti S, Servida P, Zappone E, Ruggieri L, et al. HSV-TK gene transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science. 1997;276(5319):1719–24.
Ciceri F, Bonini C, Stanghellini MTL, Bondanza A, Traversari C, Salomoni M, et al. Infusion of suicide-gene-engineered donor lymphocytes after family haploidentical haemopoietic stem-cell transplantation for leukaemia (the TK007 trial): a non-randomised phase I–II study. The Lancet Oncology. 2009;10(5):489–500.
Di Stasi A, Tey SK, Dotti G, Fujita Y, Kennedy-Nasser A, Martinez C, et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N Engl J Med. 2011;365(18):1673–83.
Zhou X, Di Stasi A, Tey SK, Krance RA, Martinez C, Leung KS, et al. Long-term outcome after haploidentical stem cell transplant and infusion of T cells expressing the inducible caspase 9 safety transgene. Blood. 2014;123(25):3895–905.
Griffioen M, van Egmond EH, Kester MG, Willemze R, Falkenburg JH, Heemskerk MH. Retroviral transfer of human CD20 as a suicide gene for adoptive T-cell therapy. Haematologica. 2009;94(9):1316–20.
Introna M, Barbui AM, Bambacioni F, Casati C, Gaipa G, Borleri G, et al. Genetic modification of human T cells with CD20: a strategy to purify and lyse transduced cells with anti-CD20 antibodies. Hum Gene Ther. 2000;11(4):611–20.
Saif MA, Borrill R, Bigger BW, Lee H, Logan A, Poulton K, et al. In vivo T-cell depletion using alemtuzumab in family and unrelated donor transplantation for pediatric non-malignant disease achieves engraftment with low incidence of graft vs. host disease. Pediatr Transplant. 2015;19(2):211–8.
Greco R, Oliveira G, Stanghellini MT, Vago L, Bondanza A, Peccatori J, et al. Improving the safety of cell therapy with the TK-suicide gene. Front Pharmacol. 2015;6:95.
Riddell SR, Elliott M, Lewinsohn DA, Gilbert MJ, Wilson L, Manley SA, et al. T-cell mediated rejection of gene-modified HIV-specific cytotoxic T lymphocytes in HIV-infected patients. Nat Med. 1996;2(2):216–23.
Zhang E, Xu H. A new insight in chimeric antigen receptor-engineered T cells for cancer immunotherapy. J Hematol Oncol. 2017;10(1):1.
Marin V, Cribioli E, Philip B, Tettamanti S, Pizzitola I, Biondi A, et al. Comparison of different suicide-gene strategies for the safety improvement of genetically manipulated T cells. Hum Gene Ther Methods. 2012;23(6):376–86.
Diaconu I, Ballard B, Zhang M, Chen Y, West J, Dotti G, et al. Inducible Caspase-9 Selectively Modulates the Toxicities of CD19-Specific Chimeric Antigen Receptor-Modified T Cells. Mol Ther. 2017;25(3):580–92.
Tey SK, Dotti G, Rooney CM, Heslop HE, Brenner MK. Inducible caspase 9 suicide gene to improve the safety of allodepleted T cells after haploidentical stem cell transplantation. Biol Blood Marrow Transplant. 2007;13(8):913–24.
Hoyos V, Savoldo B, Quintarelli C, Mahendravada A, Zhang M, Vera J, et al. Engineering CD19-specific T lymphocytes with interleukin-15 and a suicide gene to enhance their anti-lymphoma/leukemia effects and safety. Leukemia. 2010;24(6):1160–70.
Budde LE, Berger C, Lin Y, Wang J, Lin X, Frayo SE, et al. Combining a CD20 chimeric antigen receptor and an inducible caspase 9 suicide switch to improve the efficacy and safety of T cell adoptive immunotherapy for lymphoma. PLoS One. 2013;8(12):e82742.
Petrov JC, Wada M, Pinz KG, Yan LE, Chen KH, Shuai X, et al. Compound CAR T-cells as a double-pronged approach for treating acute myeloid leukemia. Leukemia. 2018;32(6):1317–26.
Tasian SK, Kenderian SS, Shen F, Ruella M, Shestova O, Kozlowski M, et al. Optimized depletion of chimeric antigen receptor T cells in murine xenograft models of human acute myeloid leukemia. Blood. 2017;129(17):2395–407.
Serafini M, Manganini M, Borleri G, Bonamino M, Imberti L, Biondi A, et al. Characterization of CD20-transduced T lymphocytes as an alternative suicide gene therapy approach for the treatment of graft-versus-host disease. Hum Gene Ther. 2004;15(1):63–76.
Di Gaetano N, Cittera E, Nota R, Vecchi A, Grieco V, Scanziani E, et al. Complement activation determines the therapeutic activity of rituximab in vivo. J Immunol. 2003;171(3):1581–7.
Philip B, Kokalaki E, Mekkaoui L, Thomas S, Straathof K, Flutter B, et al. A highly compact epitope-based marker/suicide gene for easier and safer T-cell therapy. Blood. 2014;124(8):1277–87.
Wang X, Chang WC, Wong CW, Colcher D, Sherman M, Ostberg JR, et al. A transgene-encoded cell surface polypeptide for selection, in vivo tracking, and ablation of engineered cells. Blood. 2011;118(5):1255–63.
Koneru M, O'Cearbhaill R, Pendharkar S, Spriggs DR, Brentjens RJ. A phase I clinical trial of adoptive T cell therapy using IL-12 secreting MUC-16(ecto) directed chimeric antigen receptors for recurrent ovarian cancer. J Transl Med. 2015;13:102.
Wu CY, Roybal KT, Puchner EM, Onuffer J, Lim WA. Remote control of therapeutic T cells through a small molecule-gated chimeric receptor. Science. 2015;350(6258):aab4077.
Acknowledgments
None.
Funding
This work is supported by grants from the National Institutes of Health (F31CA221002, K12HD072245), Hyundai Hope of Wheels, Curing Kids Cancer, and Children’s Healthcare of Atlanta.
Author information
Authors and Affiliations
Contributions
LCF wrote the initial manuscript draft. HTS and SSR reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Fleischer, L.C., Spencer, H.T. & Raikar, S.S. Targeting T cell malignancies using CAR-based immunotherapy: challenges and potential solutions. J Hematol Oncol 12, 141 (2019). https://doi.org/10.1186/s13045-019-0801-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13045-019-0801-y