Abstract
Background and objectives
Recent literature on multiple sclerosis (MS) demonstrates the growing implementation of optical coherence tomography–angiography (OCT-A) to discover potential qualitative and quantitative changes in the retina and optic nerve. In this review, we analyze OCT-A studies in patients with MS and examine its utility as a surrogate or precursor to changes in central nervous system tissue.
Methods
PubMed and EMBASE were systematically searched to identify articles that applied OCT-A to evaluate the retinal microvasculature measurements in patients with MS. Quantitative data synthesis was performed on all measurements which were evaluated in at least two unique studies with the same OCT-A devices, software, and study population compared to controls. A fixed-effects or random-effects model was applied for the meta-analysis based on the heterogeneity level.
Results
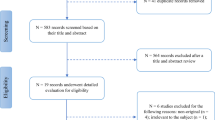
The study selection process yielded the inclusion of 18 studies with a total of 1552 evaluated eyes in 673 MS-associated optic neuritis (MSON) eyes, 741 MS without optic neuritis (MSNON eyes), and 138 eyes without specification for the presence of optic neuritis (ON) in addition to 1107 healthy control (HC) eyes. Results indicated that MS cases had significantly decreased whole image superficial capillary plexus (SCP) vessel density when compared to healthy control subjects in the analyses conducted on Optovue and Topcon studies (both P < 0.0001). Likewise, the whole image vessel densities of deep capillary plexus (DCP) and radial peripapillary capillary (RPC) were significantly lower in MS cases compared to HC (all P < 0.05). Regarding optic disc area quadrants, MSON eyes had significantly decreased mean RPC vessel density compared to MSNON eyes in all quadrants except for the inferior (all P < 0.05). Results of the analysis of studies that used prototype Axsun machine revealed that MSON and MSNON eyes both had significantly lower ONH flow index compared to HC (both P < 0.0001).
Conclusions
This systematic review and meta-analysis of the studies reporting OCT-A measurements of people with MS confirmed the tendency of MS eyes to exhibit reduced vessel density in the macular and optic disc areas, mainly in SCP, DCP, and RPC vessel densities.
Similar content being viewed by others
Introduction
Multiple sclerosis (MS) is an inflammatory auto-immune disease causing demyelination and axonal loss in the central nervous system (CNS) with varying geographical prevalence and incidence rates [1, 2]. The presence of myelin-reactive T cells in MS plaques and the effective response of MS patients to immunomodulatory drugs targeting these cells supports the theory that MS primarily results from dysregulation in the cellular immune system [3]. Aside from immune-based etiologies, there is now growing attention to discovering possible metabolic and vascular elements contributing to the pathogenesis of this disorder [4]. Because of a higher incidence among young adults, this condition can result in considerable disability for affected individuals [5, 6]. Therefore, the importance of finding clinically useful, non-invasive, and objective biomarkers to detect MS in the early stages is important.
The eye offers one gateway to studying the CNS tissue. The retina and brain share the same diencephalic origin and have analogous neuronal layers, including a ganglion cell layer (GCL), a retinal nerve fiber layer (RNFL) in addition to a vascular supply [7, 8]. Thus, the retina can be highly influenced by pathologies, such as Alzheimer’s disease, migraine, MS, neuromyelitis optica spectrum disorder (NMOSD), and myelin oligodendrocyte glycoprotein-associated disease (MOGAD), which are primarily known to also involve other parts of the CNS [9,10,11,12]. Optic neuritis (ON), characterized by acute visual loss and eye pain, is a common manifestation of MS [13, 14]. Approximately half of MS patients experience ON during the course of their disease [15].
The location of the eye makes the retina a highly accessible structure for non-invasive imaging, and it may serve as a surrogate for brain pathology [8]. Optical coherence tomography (OCT), which uses low-coherence light to capture cross-sectional and high-resolution images from the retinal and choroidal layers, has been broadly employed [16].
Application of OCT to examine the retinal layers in MS-associated optic neuritis (MSON) and MS without optic neuritis (MSNON) has revealed thinning and neurodegeneration in the peripapillary retinal nerve fiber layer (pRNFL), macular ganglion cell layer and inner plexiform layer (GCIPL) of the patients compared to the healthy controls [10]. Furthermore, investigating the role of vascular abnormalities in the establishment and progression of MS lesions is gaining popularity. These abnormalities can stem from cerebral endothelial cell dysfunction and may lead to hypoperfusion and hypoxia of CNS tissue [17]. Likewise, the anterior visual pathway requires a high blood flow rate to meet the supply and demand characteristics of this highly active metabolic system [18]. A recent study showed an abnormal retinal microcirculation in patients with relapsing–remitting multiple sclerosis (RRMS) [19]. In addition, blood flow in the ocular vascular system may be diminished in association with ON, potentially resulting in impaired visual acuity [20].
As a promising novel imaging technique, OCT–angiography (OCT-A) provides in-vivo depth-resolved images of the retinal and choroidal microvasculature [21]. The results of a meta-analysis on the application of OCT-A in dementia revealed a significant increase in the foveal avascular zone (FAZ) area in patients with Alzheimer’s disease [9].
To date, a considerable number of studies have examined retinal vasculature in MS patients using different OCT-A devices and methods, unveiling significant changes compared to healthy controls (HC) and correlations with several variables including disability scores, highlighting the promising capability of this technology to improve our current understanding of this disorder [8, 22]. MS, NMOSD, and MOGAD, which have different prognoses and treatments, may have overlap** presentations, such as ON, making it difficult to distinguish them in the acute clinical setting [23,24,25,26]. Qualitative differences in retinal microvasculature captured by OCT-A together with regular OCT may help to mitigate the challenge in the differentiation of these disorders [27,28,70, 71] or field of view, may act as sources of bias and explain the dissimilarity of findings.
Conclusion
OCT-A provides a quantitative tool to explore alterations in the retinal and optic nerve vascular networks that might be disrupted by optic neuritis and MS. This systematic review and meta-analysis of the studies reporting OCT-A measurements of patients with MS confirmed the tendency of MS eyes to exhibit reduced vessel density in the macular and optic disc areas. As the current OCT-A techniques produce two-dimensional pictures, they are unable to truly differentiate between constriction, shrinkage, or loss of vasculature. Future technological advancements should address this problem. Further studies with larger populations, longitudinal designs, and standardized segmentation and imaging analysis protocols are required to better understand the temporality and chronology of vascular alterations occurring in MS eyes. Such advances will make the application of OCT-A more practical while potentially offering a better understanding of the pathogenesis of MS.
Availability of data and materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Filippi M, Bar-Or A, Piehl F, Preziosa P, Solari A, Vukusic S, et al. Multiple sclerosis. Nat Rev Dis Prim. 2018;4:43.
Goodin DS. The epidemiology of multiple sclerosis: insights to disease pathogenesis. Handb Clin Neurol. 2014;122:231–66.
Weiner HL. Multiple sclerosis is an inflammatory T-cell-mediated autoimmune disease. Arch Neurol. 2004;61:1613–5.
Martinez Sosa S, Smith KJ. Understanding a role for hypoxia in lesion formation and location in the deep and periventricular white matter in small vessel disease and multiple sclerosis. Clin Sci (Lond). 2017;131:2503–24.
Alonso A, Hernán MA. Temporal trends in the incidence of multiple sclerosis: a systematic review. Neurology. 2008;71:129–35.
Papp V, Magyari M, Aktas O, Berger T, Broadley SA, Cabre P, et al. Worldwide incidence and prevalence of neuromyelitis optica. Neurology. 2021;96:59–77.
London A, Benhar I, Schwartz M. The retina as a window to the brain-from eye research to CNS disorders. Nat Rev Neurol. 2013;9:44–53.
Kleerekooper I, Houston S, Dubis AM, Trip SA, Petzold A. Optical coherence tomography angiography (OCTA) in multiple sclerosis and neuromyelitis optica spectrum disorder. Front Neurol. 2020;11: 604049.
Rifai OM, McGrory S, Robbins CB, Grewal DS, Liu A, Fekrat S, et al. The application of optical coherence tomography angiography in Alzheimer’s disease: a systematic review. Alzheimer’s Dement (Amsterdam, Netherlands). 2021;13: e12149.
Petzold A, Balcer LJ, Calabresi PA, Costello F, Frohman TC, Frohman EM, et al. Retinal layer segmentation in multiple sclerosis: a systematic review and meta-analysis. Lancet Neurol. 2017;16:797–812.
Oertel FC, Specovius S, Zimmermann HG, Chien C, Motamedi S, Bereuter C, et al. Retinal optical coherence tomography in neuromyelitis optica. Neurol Neuroimmunol Neuroinflamm. 2021;8:e1068.
Oertel FC, Sotirchos ES, Zimmermann HG, Motamedi S, Specovius S, Asseyer ES, et al. Longitudinal retinal changes in MOGAD. Ann Neurol. 2022;92:476–85.
de Seze J. Inflammatory optic neuritis: from multiple sclerosis to neuromyelitis optica. Neuroophthalmology. 2013;37:141–5.
Soelberg K, Jarius S, Skejoe H, Engberg H, Mehlsen JJ, Nilsson AC, et al. A population-based prospective study of optic neuritis. Mult Scler. 2017;23:1893–901.
Balcer LJ. Clinical practice. Optic neuritis. N Engl J Med. 2006;354:1273–80.
Podoleanu AG. Optical coherence tomography. J Microsc. 2012;247:209–19.
Plumb J, McQuaid S, Mirakhur M, Kirk J. Abnormal endothelial tight junctions in active lesions and normal-appearing white matter in multiple sclerosis. Brain Pathol. 2002;12:154–69.
Kleerekooper I, Petzold A, Trip SA. Anterior visual system imaging to investigate energy failure in multiple sclerosis. Brain. 2020;143:1999–2008.
Jiang H, Delgado S, Tan J, Liu C, Rammohan KW, DeBuc DC, et al. Impaired retinal microcirculation in multiple sclerosis. Mult Scler. 2016;22:1812–20.
Chen T-C, Yeh C-Y, Lin C-W, Yang C-M, Yang C-H, Lin I-H, et al. Vascular hypoperfusion in acute optic neuritis is a potentially new neurovascular model for demyelinating diseases. PLoS ONE. 2017;12: e0184927.
Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G. Optical coherence tomography angiography. Prog Retin Eye Res. 2018;64:1–55.
Murphy OC, Kwakyi O, Iftikhar M, Zafar S, Lambe J, Pellegrini N, et al. Alterations in the retinal vasculature occur in multiple sclerosis and exhibit novel correlations with disability and visual function measures. Mult Scler. 2020;26:815–28.
Marignier R, Hacohen Y, Cobo-Calvo A, Pröbstel A-K, Aktas O, Alexopoulos H, et al. Myelin-oligodendrocyte glycoprotein antibody-associated disease. Lancet Neurol. 2021;20:762–72.
Denis M, Woillez J-P, Smirnov VM, Drumez E, Lannoy J, Boucher J, et al. Optic nerve lesion length at the acute phase of optic neuritis is predictive of retinal neuronal loss. Neurol Neuroimmunol Neuroinflamm. 2022;9: e1135.
Akaishi T, Takahashi T, Misu T, Abe M, Ishii T, Fujimori J, et al. Progressive patterns of neurological disability in multiple sclerosis and neuromyelitis optica spectrum disorders. Sci Rep. 2020;10:13890.
Chen JJ, AbouChehade JE, Iezzi RJ, Leavitt JA, Kardon RH. Optical coherence angiographic demonstration of retinal changes from chronic optic neuropathies. Neuroophthalmology. 2017;41:76–83.
Graves JS, Oertel FC, Van der Walt A, Collorone S, Sotirchos ES, Pihl-Jensen G, et al. Leveraging visual outcome measures to advance therapy development in neuroimmunologic disorders. Neurol Neuroimmunol Neuroinflamm. 2022;9: e1126.
Oertel FC, Zimmermann H, Paul F, Brandt AU. Optical coherence tomography in neuromyelitis optica spectrum disorders: potential advantages for individualized monitoring of progression and therapy. EPMA J. 2018;9:21–33.
Liu C, **ao H, Zhang X, Zhao Y, Li R, Zhong X, et al. Optical coherence tomography angiography helps distinguish multiple sclerosis from AQP4-IgG-seropositive neuromyelitis optica spectrum disorder. Brain Behav. 2021;11: e02125.
Pache F, Zimmermann H, Mikolajczak J, Schumacher S, Lacheta A, Oertel FC, et al. MOG-IgG in NMO and related disorders: a multicenter study of 50 patients. Part 4: afferent visual system damage after optic neuritis in MOG-IgG-seropositive versus AQP4-IgG-seropositive patients. J Neuroinflamm. 2016;13:1–10.
Lanzillo R, Cennamo G, Criscuolo C, Carotenuto A, Velotti N, Sparnelli F, et al. Optical coherence tomography angiography retinal vascular network assessment in multiple sclerosis. Mult Scler. 2018;24:1706–14.
Tewarie P, Balk L, Costello F, Green A, Martin R, Schippling S, et al. The OSCAR-IB consensus criteria for retinal OCT quality assessment. PLoS ONE. 2012;7: e34823.
Petzold A, Albrecht P, Balcer L, Bekkers E, Brandt AU, Calabresi PA, et al. Artificial intelligence extension of the OSCAR-IB criteria. Ann Clin Transl Neurol. 2021;8:1528–42.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editors. Cochrane handbook for systematic reviews of interventions version 6.1 (updated September 2020). Cochrane. 2020. www.training.cochrane.org/handbook.
Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, Comi G, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17:162–73.
Khader SA, Nawar AE, Ghali AA, Ghoneim AM. Evaluation of optical coherence tomography angiography findings in patients with multiple sclerosis. Indian J Ophthalmol. 2021;69:1457–63.
Durbin MK, An L, Shemonski ND, Soares M, Santos T, Lopes M, et al. Quantification of retinal microvascular density in optical coherence tomographic angiography images in diabetic retinopathy. JAMA Ophthalmol. 2017;135:370–6.
Lin Y, Jiang H, Liu Y, Rosa Gameiro G, Gregori G, Dong C, et al. Age-related alterations in retinal tissue perfusion and volumetric vessel density. Invest Ophthalmol Vis Sci. 2019;60:685–93.
Camino A, Jia Y, Guo Y, Huang D. Compensation of OCTA flow index dependence on OCT signal strength. Invest Ophthalmol Vis Sci. 2021;62:1774.
Grading diabetic retinopathy from stereoscopic color fundus photographs—an extension of the modified Airlie House classification. ETDRS report number 10. Early treatment diabetic retinopathy study research group. Ophthalmology. 1991;98:786–806.
Rogaczewska M, Michalak S, Stopa M. Macular vessel density differs in multiple sclerosis and neuromyelitis optica spectrum disorder: an optical coherence tomography angiography study. PLoS ONE. 2021;16: e0253417.
Rogaczewska M, Michalak S, Stopa M. Differentiation between multiple sclerosis and neuromyelitis optica spectrum disorder using optical coherence tomography angiography. Sci Rep. 2021;11:10697.
Rogaczewska M, Michalak S, Stopa M. Optical coherence tomography angiography of peripapillary vessel density in multiple sclerosis and neuromyelitis optica spectrum disorder: a comparative study. J Clin Med. 2021;10:609.
Lanzillo R, Cennamo G, Moccia M, Criscuolo C, Carotenuto A, Frattaruolo N, et al. Retinal vascular density in multiple sclerosis: a 1-year follow-up. Eur J Neurol. 2019;26:198–201.
Yilmaz H, Ersoy A, Icel E. Assessments of vessel density and foveal avascular zone metrics in multiple sclerosis: an optical coherence tomography angiography study. Eye (Lond). 2020;34:771–8.
Wang X, Jia Y, Spain R, Potsaid B, Liu JJ, Baumann B, et al. Optical coherence tomography angiography of optic nerve head and parafovea in multiple sclerosis. Br J Ophthalmol. 2014;98:1368–73.
Ulusoy MO, Horasanlı B, Işık-Ulusoy S. Optical coherence tomography angiography findings of multiple sclerosis with or without optic neuritis. Neurol Res. 2020;42:319–26.
Spain RI, Liu L, Zhang X, Jia Y, Tan O, Bourdette D, et al. Optical coherence tomography angiography enhances the detection of optic nerve damage in multiple sclerosis. Br J Ophthalmol. 2018;102:520–4.
Murphy OC, Kalaitzidis G, Vasileiou E, Filippatou AG, Lambe J, Ehrhardt H, et al. Optical coherence tomography and optical coherence tomography angiography findings after optic neuritis in multiple sclerosis. Front Neurol. 2020;11: 618879.
Lee G-I, Park K-A, Oh SY, Min J-H, Kim BJ. Differential patterns of parafoveal and peripapillary vessel density in multiple sclerosis and neuromyelitis optica spectrum disorder. Mult Scler Relat Disord. 2021;49: 102780.
Jesus J, Soares R, Geraldes R, Matias M, Chibante J. Analysis of choroidal vessel density in patients with multiple sclerosis. Biomark Neuropsychiatry. 2021;5: 100040.
Farci R, Carta A, Cocco E, Frau J, Fossarello M, Diaz G. Optical coherence tomography angiography in multiple sclerosis: a cross-sectional study. PLoS ONE. 2020;15: e0236090.
Cordon B, Vilades E, Orduna E, Satue M, Perez-Velilla J, Sebastian B, et al. Angiography with optical coherence tomography as a biomarker in multiple sclerosis. PLoS ONE. 2020;15: e0243236.
Cennamo G, Carotenuto A, Montorio D, Petracca M, Moccia M, Melenzane A, et al. Peripapillary vessel density as early biomarker in multiple sclerosis. Front Neurol. 2020;11:542.
Ava S, Tamam Y, Hazar L, Karahan M, Erdem S, Dursun ME, et al. Relationship between optical coherence tomography angiography and visual evoked potential in patients with multiple sclerosis. Indian J Ophthalmol. 2022;70:873–8.
Aly L, Strauß E-M, Feucht N, Weiß I, Berthele A, Mitsdoerffer M, et al. Optical coherence tomography angiography indicates subclinical retinal disease in neuromyelitis optica spectrum disorders. Mult Scler. 2022;28:522–31.
Jiang H, Gameiro GR, Liu Y, Lin Y, Hernandez J, Deng Y, et al. Visual function and disability are associated with increased retinal volumetric vessel density in patients with multiple sclerosis. Am J Ophthalmol. 2020;213:34–45.
Koustenis AJ, Harris A, Gross J, Januleviciene I, Shah A, Siesky B. Optical coherence tomography angiography: an overview of the technology and an assessment of applications for clinical research. Br J Ophthalmol. 2017;101:16–20.
Sinnecker T, Bozin I, Dörr J, Pfueller CF, Harms L, Niendorf T, et al. Periventricular venous density in multiple sclerosis is inversely associated with T2 lesion count: a 7 Tesla MRI study. Mult Scler. 2013;19:316–25.
Park JJ, Soetikno BT, Fawzi AA. Characterization of the middle capillary plexus using optical coherence tomography angiography in healthy and diabetic eyes. Retina. 2016;36:2039–50.
Paul F, Calabresi PA, Barkhof F, Green AJ, Kardon R, Sastre-Garriga J, et al. Optical coherence tomography in multiple sclerosis: a 3-year prospective multicenter study. Ann Clin Transl Neurol. 2021;8:2235–51.
Green AJ, McQuaid S, Hauser SL, Allen IV, Lyness R. Ocular pathology in multiple sclerosis: retinal atrophy and inflammation irrespective of disease duration. Brain. 2010;133:1591–601.
D’haeseleer M, Cambron M, Vanopdenbosch L, De Keyser J. Vascular aspects of multiple sclerosis. Lancet Neurol. 2011;10:657–66.
Zhang X, Guo X, Zhang N, Cai H, Sun J, Wang Q, et al. Cerebral blood flow changes in multiple sclerosis and neuromyelitis optica and their correlations with clinical disability. Front Neurol. 2018;9:305.
Paling D, Thade Petersen E, Tozer DJ, Altmann DR, Wheeler-Kingshott CAM, Kapoor R, et al. Cerebral arterial bolus arrival time is prolonged in multiple sclerosis and associated with disability. J Cereb Blood Flow Metab. 2014;34:34–42.
Inglese M, Adhya S, Johnson G, Babb JS, Miles L, Jaggi H, et al. Perfusion magnetic resonance imaging correlates of neuropsychological impairment in multiple sclerosis. J Cereb Blood Flow Metab. 2008;28:164–71.
Holland CM, Charil A, Csapo I, Liptak Z, Ichise M, Khoury SJ, et al. The relationship between normal cerebral perfusion patterns and white matter lesion distribution in 1,249 patients with multiple sclerosis. J Neuroimaging. 2012;22:129–36.
Bambo MP, Garcia-Martin E, Perez-Olivan S, Sigut J, Fumero F, Fuentes JL, et al. Diagnostic ability of a new method for measuring haemoglobin levels in the optic nerve head in multiple sclerosis patients. Br J Ophthalmol. 2013;97:1543–8.
Spaide RF, Klancnik JMJ, Cooney MJ. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol. 2015;133:45–50.
Hao H, Sasongko MB, Wong TY, Che Azemin MZ, Aliahmad B, Hodgson L, et al. Does retinal vascular geometry vary with cardiac cycle? Invest Ophthalmol Vis Sci. 2012;53:5799–805.
Alnawaiseh M, Lahme L, Treder M, Rosentreter A, Eter N. Short-term effects of exercise on optic nerve and macular perfusion measured by optical coherence tomography angiography. Retina. 2017;37:1642–6.
Acknowledgements
Not applicable.
Funding
No funding for this research was done.
Author information
Authors and Affiliations
Contributions
SM did the literature search, developed the protocol, and contributed to screening and data extraction. MG wrote the original draft. MAS designed the project, performed the review and contributed to the protocol development and final manuscript. HH designed and developed the figures. JFA, SLG, EMF, TCF, SS, NS, and FP encouraged and supervised the project and reviewed the manuscript. All authors have contributed significantly, and agree with its submission to this journal. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Shiv Saidha has received consulting fees from Medical Logix for the development of CME programs in neurology and has served on scientific advisory boards for Biogen, Novartis, Genentech Corporation, TG therapeutics, Rewind therapeutics & Bristol Myers Squibb. He has performed consulting for Novartis, Genentech Corporation, JuneBrain LLC, and Lapix therapeutics. He is the PI of investigator-initiated studies funded by Genentech Corporation, Novartis, and Biogen. He previously received support from the Race to Erase MS foundation. He has received equity compensation for consulting from JuneBrain LLC and Lapix therapeutics. He was also the site investigator of trials sponsored by MedDay Pharmaceuticals, Clene Pharmaceuticals, and is the site investigator of a trial sponsored by Novartis. Dr. Sattarnezhad has received Sylvia Lawry Physician Fellowship award from National Multiple Sclerosis Society (NMSS). Elliot Frohman has received consulting and speaker fees from Biogen, Genzyme, Novartis, Alexion, Horizon, and Janssen. Soheil Mohammadi, Mahdi Gouravani, Mohammad Amin Salehi, J. Fernando Arevalo, Steven L. Galetta, Hamid Harandi, Teresa C Frohman, and Friedemann Paul report no disclosures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
OCT-A metrics and regions assessed in the included studies with term definitions.
Additional file 2: Table S2.
Summary of fields of view and dimensions used to define retinal regions in included studies.
Additional file 3: Table S3.
Exclusion criteria and additional information of included studies.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Mohammadi, S., Gouravani, M., Salehi, M.A. et al. Optical coherence tomography angiography measurements in multiple sclerosis: a systematic review and meta-analysis. J Neuroinflammation 20, 85 (2023). https://doi.org/10.1186/s12974-023-02763-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12974-023-02763-4