Abstract
In the literature, there is growing evidence that subfertile patients who conceived after infertility treatments have an increased risk of pregnancy and perinatal complications and this is particularly true for patients who conceived through use of high technology infertility treatments. Moreover, high technology infertility treatments include many concomitant clinical and biological risk factors. This review aims to summarize in a systematic fashion the current evidence regarding the relative effect of the different procedures for high technology infertility treatments on the risk of adverse pregnancy and perinatal outcome. A literature search up to August 2016 was performed in IBSS, SocINDEX, Institute for Scientific Information, PubMed, Web of Science and Google Scholar and an evidence-based hierarchy was used to determine which articles to include and analyze. Data on prepregnancy maternal factors, low technology interventions, specific procedures for male factor, ovarian tissue/ovary and uterus transplantation, and chromosomal abnormalities and malformations of the offspring were excluded. The available evidences were analyzed assessing the level and the quality of evidence according to the Oxford Centre for Evidence-Based Medicine guidelines and the Grading of Recommendations Assessment, Development, and Evaluation system, respectively. Current review highlights that every single procedure of high technology infertility treatments can play a crucial role in increasing the risk of pregnancy and perinatal complications. Due to the suboptimal level and quality of the current evidence, further well-designed studies are needed.
Similar content being viewed by others
Background
Throughout the years, it has always been clear to scientists that the primary endpoint in reproductive medicine was the healthy baby and that all other endpoints would be considered only a surrogate [1, 2]. The published infertility clinical trials have rarely reported clear data about the possible harm of the medical, surgical and biological procedures for enhancing fertility [3, 4], as well as giving very little relevance to long-term effects of those procedures on maternal and offspring health. Only 4.8 % and 5.7 % of randomized controlled trials (RCTs) on infertility treatments reported on perinatal and maternal outcome [5]. This is probably because obstetrical and infant care are delivered by other providers and patients are lost to follow-up.
Notwithstanding these limitations, more and more data available in the literature seem to demonstrate that pregnancy following infertility treatments are at higher risk of adverse pregnancy and perinatal outcomes when compared with those after natural conception (NC) independently from scientific approach [6], and this is particularly true for pregnancies achieved thanks to high technology infertility treatments [5]. The majority of this risk is a “pure” iatrogenic risk due to the high rates of multiple births, i.e. 41.1 % of the United States (US) infants conceived with assisted reproductive technologies (ART) were born as multiple-birth infants compared with only 3.5 % of infants among the general birth population [7]. The rate of multiple deliveries following ART represents about 18.7 % of total multiple-birth infants [7]. However, because also singleton infants conceived with ART are at higher risk of preterm birth (PTB) and low birthweight (LBW) [7], other determinants cannot be excluded. Patients’ characteristics including the infertility state [8, 9] and many preconception risk factors for subfertility [10–12] can largely increase the absolute and relative risk of obstetric morbidity.
Although systematic reviews with and/or without meta-analysis have been published on specific topics, at the moment no comprehensive review is available in the literature, discussing the impact on maternal and perinatal outcome of each element and/or clinical/biological choice which comprise the high technology infertility treatments. Based on these considerations, the aim of the current document was to comprehensively review in a systematic fashion the hitherto published evidences regarding the effects of high technology infertility treatments on the obstetric risk of patients with female and couple infertility. The effects on the risk of chromosomal abnormalities and malformations of the offspring was not a study aim.
Materials and methods
The methodology used for the current systematic review consisted of searching all available articles for each specific issue to explore the relationship between high technology infertility treatments and pregnancy and perinatal complications. High technology infertility treatments were considered as all interventions for fertility enhancement including manipulation of female gametes. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement [13] was followed but after comprehensive search all the authors agreed to prepare the document in a narrative fashion in consideration of the multifaceted aspects to discuss.
Multiple strategies were used to search and identify relevant demographic, epidemiological, clinical and experimental studies. Sociological online libraries (IBSS, SocINDEX), Institute for Scientific Information, PubMed, Web of Science and Google Scholar were consulted. Only articles written in English were considered. The search was conducted independently by two authors (S.P. and S.S.). The literature available up to August 2016 was captured, including all available studies which reported data about the relationship between each fertility technique and the related obstetric and perinatal complications, matching every intervention with every potential obstetric disorder and perinatal health impact, as shown in Table 1. Additional journal articles were identified from the bibliography of the studies included. At study design, all the authors agreed to exclude from final analysis data of pregnancy and perinatal complications related to: 1. prepregnancy maternal factors; 2. low technology interventions (intervention aimed to enhance fertility without any manipulation of female gametes, ie. lifestyle intervention programs, insulin sensitizing drugs, ovulation inductors, macro- and micro-supplements, intrauterine insemination, etc.); 3. specific high technology infertility treatments for male factor [including intracytoplasmic sperm injection (ICSI), specific techniques for sperm selection and/or retrieval, etc.]; 4. ovarian tissue/ovary and uterus transplantation; 5. chromosomal abnormalities and malformations of the offspring. The choice to consider a study relevant in order to be included in the current review was arbitrarily taken by each author, even if an evidence-based hierarchy was used. Exploratory studies on mechanisms of action and/or pathogenesis of any complication were included only in absence of available clinical data. Data on the efficacy of each procedure were reported only as necessary for the study aim. Any disagreement or uncertainty was resolved by discussion to reach a consensus.
The available evidence about the relationship between high technology infertility treatments and adverse obstetric and perinatal outcomes was analyzed assessing the level of evidence according to the Oxford Centre for Evidence-Based Medicine (OCEM)-Levels of Evidence 2011 guidelines [14] and the quality of evidence according to the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system [15].
Results
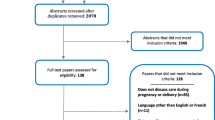
Figure 1 is the flow diagram of the systematic review including the numbers of studies screened, assessed for eligibility, and included in the review, with reasons for exclusions at each stage [13]. Table 2 summarizes the main risks for obstetric and perinatal adverse outcomes in women who receive ART and specific procedure for high technology infertility treatments according to the level [14] and the quality [15] of available evidence.
PRISMA 2009 [13] flow diagram
Overall ART-related complications
Singleton pregnancies
Many data from systematic reviews with and without meta-analyses have demonstrated that ART singletons are at increased risk of pregnancy and perinatal complications. In 2004, an initial systematic review of controlled studies found a relative risk of 3.27 (95 % CI 2.03 to 5.28) for early-preterm birth (EPTB) (< 32 weeks) in singleton pregnancies from assisted conceptions [16]. Singleton pregnancies resulting from in vitro fertilization (IVF) presented higher rates of worse obstetric outcome, compared with NC singletons of couples matched for maternal age, showing an increase in perinatal mortality [odd ratio (OR) 2.40, 95 % confidence interval (CI) 1.59 to 3.63], PTB at < 33 weeks’ gestation (OR 2.99, 95 % CI 1.54 to 5.80), PTB at < 37 weeks’ gestation (OR 1.93, 95 % CI 1.36 to 2.74), very-low preterm birth (VLBW) (<1500 g) (OR 3.78, 95 % CI 4.29 to 5.75), and SGA (OR 1.59, 95 % CI 1.20 to 2.11) [17]. It is noteworthy that this review included in the final analysis studies in which the control arm was composed of NCs and after ovulation induction strategies. In 2012, an extensive systematic review with meta-analysis of 20 matched and 10 unmatched cohort studies, most having high quality and adjusted for important confounders, concluded that singleton IVF/intracytoplasmic sperm injection (ICSI) pregnancies are associated with higher risks of antepartum hemorrhage [relative risk (RR) 2.49, 95 % CI 2.30 to 2.69], pregnancy-induced hypertension (PIH)/preeclampsia (PE) (RR 1.49, 95 % CI 1.39 to 1.59), gestational diabetes mellitus (GDM) (RR 1.48, 95 % CI 1.33 to 1.66), caesarean section (RR 1.56, 95 % CI 1.51 to 1.60), PTB (RR 1.54, 95 % CI 1.47 to 1.62), LBW (RR 1.65, 95 % CI 1.56 to 1.75), SGA (RR 1.39, 95 % CI 1.27 to 1.53), admission to neonatal intensive care unit (NICU) (RR 1.58, 95 % CI 1.42 to 1.77), and perinatal mortality (RR 1.87, 95 % CI 1.48 to 2.37) than NC [18]. Also in this last meta-analysis, only in some of the included studies were the pregnancies resulting from ovulation induction excluded from non-IVF/ICSI conceptions [18].
The most recently published meta-analysis about the risk of pregnancy-related complications and adverse perinatal outcomes in singleton pregnancies obtained with ART involves 50 cohort studies for a total of 161,370 ART singleton compared with 2,280,241 NC singleton pregnancies [19]. This meta-analysis revealed that the singleton ART pregnancies experienced a significantly increased risk of placenta previa (RR 3.71, 95 % CI 2.67 to 5.16), placental abruption (RR 1.83, 95 % CI 1.49 to 2.24), antepartum hemorrhage (RR 2.11, 95 % CI 1.86 to 2.38), and postpartum hemorrhage (RR 1.29, 95 % CI 1.06 to 1.57). The increased risk for PIH, GDM, caesarean sections, PTB, LBW, SGA and perinatal mortality was confirmed and resulted not different from previous meta-analytic data [19]. Of note, the risk of EPTB (RR 2.12, 95 % CI 1.73 to 2.59) and of VLBW (RR 2.12, 95 % CI 1.84 to 2.43) was two-fold higher in ART conceptions than in NC [19]. The results of this study are consistent with those of the previous reviews but it presents important strengths, such as the large sample size, 64 % of the included studies were considered of high methodological quality and the association between ART and obstetric risk persisted and remained statistically significant in sensitivity analysis based on various exclusion criteria [19]. Nevertheless, as relevant biases, patients who achieved a pregnancy with ovulation induction and/or intrauterine insemination (IUI) were included in the NC category, resulting in an underestimation of the association between ART and adverse outcomes. When data were restricted to studies that did not include these patients in the NC group, the risk of GDM, placental abruption, PTB, EPTB, LBW, VLBW, and perinatal mortality resulted in a further increase. Unfortunately, more than half of the included studies did not specify whether they were included, thereby restricting this subgroup analysis [19]. Finally, recent retrospective studies confirmed in ART pregnancies a risk of PIH/PE about 20 % higher (aOR 1.17, 95 % CI 1.10 to 1.24) [20] and demonstrated a risk of peripartum hysterectomy about six-fold increased (OR 5.98 95 % CI 2.18 to 16.40) in comparison with non-ART pregnancies [21].
Multiple pregnancies
Initial systematic reviews with meta-analyses [16, 17] demonstrated that ART twins had a higher risk of PTB compared with NC twins but the perinatal mortality, contrary to singletons, was unchanged [17] or reduced [16].
A recent meta-analysis including 39 cohort studies explored the risk of adverse pregnancy outcomes in ART conceptions compared with NC in a sample of 146,008 multiple births [153]. Also the odds for SGA (OR 1.81, 95 % CI 1.26 to 2.60), PTB (OR 1.34, 95 % CI 1.08 to 1.66), and caesarean section (OR 2.71, 95 % CI 2.23 to 3.30) were increased [153]. Meta-regression for the covariate of age suggested that risk was independent of age [153]. Similarly, also the fourth meta-analysis [154] including 11 retrospective and prospective cohort studies confirmed that OD increase the risk of PE (in comparison with homologous IVF cycles) of about 70 % and that neither multiple pregnancies nor patient age can explain that effect by meta-regression analysis.
Finally, the last systematic review with meta-analysis [155] included, after search for original studies reporting at least five OD pregnancies with a control group of pregnancies conceived by conventional IVF/ICSI or NC and case series with > 500 cases, 35 studies reporting one or more pregnancy and perinatal complications were analyzed. The risk of PIH (aOR 2.30, 95 % CI 1.60 to 3.32), PE (aOR 2.11, 95 % CI 1.42 to 3.15), LBW (aOR 1.53, 95 % CI 1.16 to 2.01), PTB (aOR 1.75, 95 % CI 1.39 to 2.20) and CS (aORs 2.20, 95 % CI 1.85 to 2.60) was higher in OD than in IVF singleton pregnancies, whereas in multiple pregnancies only the incidence of PIH (aOR 2.45, 95 % CI 1.53 to 3.93) and PE (aOR 3.31, 95 % CI 1.61 to 6.80) was increased [155]. The risk of PE (aOR 2.94, 95 % CI 2.29 to 3.76), PTB (aOR 2.30, 95 % CI 1.09 to 4.87), LBW (aOR 1.94, 95 % CI 1.10 to 3.41) and CS (aOR 2.38, 95 % CI 2.01 to 2.81) was also increased in OD vs. NC singleton pregnancies [155]. Postpartum hemorrhage resulted increased in OD vs. IVF both in singleton (aOR 2.40, 95 % CI 1.49 to 3.88) and multiple (aOR 4.91, 95 % CI 1.22 to 19.83) pregnancies. No difference was detected in terms of GDM [155].
One of the largest and better controlled cohort study published on perinatal outcomes of children born after OD included 375 OD babies, and clinical data compared with three control cohorts of children, i.e. NC (33,852 babies matched by date and year of birth) and born after either IVF (11,060 singletons, and 6,532 twins) or ICSI (5,866 singletons, and 3,101 twins) [156]. An increased risk of PTB (aORs 1.8, 95 % CI 1.2 to 2.3; aOR 2.5, 95 % CI 1.7 to 3.6; and aOR 3.4, 95 % CI 2.3 to 4.9, respectively) and LBW (aOR 1.4, 95 % CI 0.9 to 2.2; aOR 1.8, 95 % CI 1.2 to 2.8; and aOR 2.6, 95 % CI 1.7 to 4.0, respectively) was detected in OD pregnancies vs. control pregnancies [156]. Of note, the risk of PE was also increased three-fold in OD pregnancies (aOR 2.9, 95 % CI 1.8 to 4.6; aOR 2.8, 95 % CI 1.7 to 4.5; and aOR 3.1, 95 % CI 1.9 to 4.9) [156]. The risk remained higher also after adjusting for maternal characteristics and after sub-analysis for twin pregnancies. Moreover, when the perinatal risk was adjusted for maternal PE the results improved, demonstrating no direct effect of OD on perinatal outcomes [156]. These data have been recently confirmed by a systematic review [157] showing that OD is an independent risk factor for PIH/PE, especially in twin pregnancies, and that its effect on fetal birthweight or growth is minimal after adjusting for obstetric complications. Finally, a very recent national registry study confirmed that OD recipients are more likely to have PTB (aOR 1.28, 95 % CI 1.12 to 1.46) and VPTB (aOR 1.30, 95 % CI 1.03 to 1.64) when compared with autologous patients, whereas the risk of having a SGA baby (aOR 0.72, 95 % CI 0.58 to 0.89) and of perinatal death (aOR 0.29, 95 % CI 0.09 to 0.94) was lower after adjusting data for gestational age [158].
Sperm donation
Sparse available data in the literature seems to suggest an increased risk of hypertensive disorders during pregnancy, with a specific increase of the PE risk, in nulliparous and in multiparous pregnancies with changed paternity [159, 160]. In this view, it could be hypothesized that the exposure to the paternal semen before conception has a protective effect, whereas the use of donor insemination after a previous pregnancy with paternal semen could increase the risk with an immune mechanism similar to hypertensive disorders seen in OD pregnancies. Unfortunately, at the moment data on sperm donation regard low technology interventions [11], and showed that the use of donor sperm in IUI cycles is associated with a risk of perinatal complications lower to those of the children born after partner sperm IUI and comparable to those of the NC children [44].
Embryo donation
Very little is known about the relationship between embryo donation and pregnancy and perinatal complications. In fact, available data can be extrapolated from infertile populations who conceived after mixed procedures of gamete and embryo donations [159]. At the moment, it very difficult to draw conclusions on the obstetric risk in women who had an embryo donation because the number of confounders and biases is so frequent and the detrimental effect of the double gamete donation can be only supposed. Commonly, the recipients are highly selected women with few medical comorbidities but who had had probably many previous ART failed attempts and a longer time-to-pregnancy. In addition, embryos donated are almost always frozen embryos. Finally, because embryo donation is more cost-effective than oocyte donation in case of male factor [161], the comparison in terms of pregnancy complication can be favorable for pregnancies obtained after embryo donation.
Surrogate pregnancy
A systematic review on gestational surrogacy has been very recently published, and the pregnancy and perinatal outcomes of gestational carriers compared, when possible, with those of standard IVF and OD cycles [162]. The incidence of PIH ranged from 4.3 to 10 % and from 2.9 and 7.4 %, and the incidence of placenta previa and/or placental abruption from 1.1 to 7.9 % and from 1.1 to 3.7 %, respectively, in singleton and twin surrogate pregnancies [162], and resulted not different from those observed in IVF pregnancies and lower than that usually reported in OD pregnancies (ranged from 16 to 40 %) [163]. Three cases of hysterectomies related to delivery were also reported in gestational carriers and were due to uterine atony, placenta accreta and uterine rupture [162]. In surrogate singletons, the incidence of PTB and of LBW resulted, respectively, ranging from 0 to 11.5 % and from 0 to 11.1 % [162]. When compared to control groups, the risk of PTB and LBW was not different from IVF singletons (incidence of PTB of 14 % and of 13.6–14.0 %, respectively) and the risk of LBW was also not different from OD singletons (incidence of 14.0 %) [162]. However, a very recent US cohort study [164] underlined that the increased risk of PTB (aRR 1.14, 95 % CI 1.05 to 1.23) observed in gestational carriers is significantly influenced not only by multiple pregnancies but also by OD.
Discussion
Despite the level and the quality of the current evidence it is generally suboptimal due to the presence of biases, confounders and limitations in study design (Table 3), current comprehensive review confirms that subfertile women who conceived after the use of high technology infertility treatments have an overall increased risk of pregnancy and perinatal complications, and highlights that every single step and/or procedure can play an independent and crucial role. In addition, several concomitant risk factors are frequently present in the same woman and influence the clinical and biological strategy of treatment. Thus, it is virtually impossible to define the weight of each reproductive treatment’s phase determining the whole patient’s risk. In fact, the infertility condition represents a bias per se in every study dealing with infertility treatments [11] and the presence of many confounding factors cannot be always adequately controlled through multivariate analysis because in many studies they are not clinically available, missing, or not collected.
Ideally, the knowledge of the pathogenesis of the increased risk of pregnancy and perinatal complications in women who receive high technology infertility treatments and of the specific mechanisms of action could be crucial for preventing them. Unfortunately, few data are available regarding the biological explanations of that increased risk. Many mechanisms have been hypothesized and regard the alterations of the early placentation including not only alterations in endometrial receptivity, genetic and epigenetic mechanisms of implantation, invasion and growth of the trophoblast but also genetic and/or epigenetic alterations of oocyte/embryos due to biological manipulations (extended culture, culture media, techniques of cryopreservation, etc.), and immunological intolerance in case of OD because the fetal genome is allogenic to the carrier [11, 90, 127, 142, 165–167]. Thus, in the next future, still remains the need and an effort should be made to understand the reasons of these risks in order to minimize or prevent them.
However, from the clinical point of view, the priority is not to precisely estimate the amount of the obstetric risk but to recognize the presence of one or more risk factors (infertility and subfertility causes, patient’s characteristics and specific ARTs-associated risks), to correct those modifiable and to strictly follow the resulting pregnancies with appropriate prenatal cares. In fact, the delay in receiving prenatal care increased the PTB risk, while more-frequent use of prenatal care significantly improved the birth weight among pregnancies at high risk including subfertile women [168]. Recently, a large nationwide population-based study demonstrated that an adequate and intensive prenatal care reduces the risk of adverse pregnancy outcomes in women with history of infertility [169]. Specifically, less than six prenatal visits (compared with equal or more than six prenatal visits) and prenatal visits performed after the 12th week of gestation (compared with prenatal visits performed at or before the 12th week of gestation) are related with a risk lower of VLBW neonates (aOR15.1, 95 % CI 8.8 to 25.8; aOR 2.1, 95 % CI 1.2 to 3.8; respectively), LBW neonates (aOR 2.1, 95 % CI 1.5 to 2.5; aOR 1.6, 95 % CI 1.3 to 1.9; respectively), and preterm birth (aOR 2.2, 95 % CI 1.9 to 2.6; aOR 1.1, 95 % CI 0.9 to 1.3; respectively) [169]. That data, however, were limited to singleton pregnancies.
At the moment, well established strategies to identify, follow and manage infertile patients and/or patients who have receive an infertility treatment are lacking and only few papers suggest potential strategies of management consisting in a generic close pregnancy monitoring and diagnostic testing [137, 157]. Physicians should assess the pregestational risk of infertile women before start any fertility enhancement treatment and discuss with the couples the increased risk for maternal and perinatal complications in a view of risk to benefit ratio and the potential alternatives, suggesting also to avoid any medical intervention in case of high-risk patients [137, 170], such as in case of women of very advanced reproductive age (> 55 years) or advanced reproductive age (> 45 years) with medical conditions [171].
Conclusion
Subfertile women who conceived after the use of high technology infertility treatments are at increased risk of pregnancy complications, and every single/specific step and/or procedure can play an independent and crucial role. Thus, all infertile patients scheduled for high technology infertility treatments should be clearly informed of that increased obstetric and perinatal risk in case of pregnancy, regardless of multiple pregnancy. A careful preconceptional counselling aimed to optimize the general health status of the pre-pregnant women is needed (to stop smoking, reduce BMI in overweight/obese patients, and so on), identifying and treating modifiable reproductive disorders [11] and, finally, an effort should be made to optimize the infertility treatments (milder stimulation, OHSS prevention, elective SET) in order to prevent or reduce the risk of pregnancy complications in these infertile women. Finally, further large cohort prospective studies are required to clarify the contribution of each single factor on pregnancy and perinatal outcomes.
Abbreviations
- aOR:
-
Adjusted odd ratio
- aRR:
-
Adjusted relative risk
- ART:
-
Assisted reproductive technology
- ASRM:
-
American society of reproductive medicine
- BMI:
-
Body mass index
- CC:
-
Clomiphene citrate
- CI:
-
Confidence interval
- COH:
-
Controlled ovarian hyperstimulation
- DET:
-
Double embryo transfer
- E2:
-
Estradiol
- EPTB:
-
Early-preterm birth
- ESHRE:
-
European society of human reproduction and embryology
- ET:
-
Embryo transfer
- FSH:
-
Follicle stimulating hormone
- GnRH:
-
Gonadotropin-releasing hormone
- GRADE:
-
Grading of recommendations assessment, development, and evaluation
- hCG:
-
Human chorionic gonadotropin
- ICSI:
-
Intracytoplasmic sperm injection
- IUGR:
-
Intrauterine growth restriction
- IUI:
-
Intrauterine insemination
- IVF:
-
In vitro fertilization
- IVM:
-
In vitro maturation
- LBW:
-
Low birth weight
- LGA:
-
Large-for-gestational age
- NC:
-
Natural conception
- NICU:
-
Neonatal intensive care unit
- OCEM:
-
Oxford centre for evidence-based medicine
- OD:
-
Oocyte donation
- OGTT:
-
Oral glucose tolerance test
- OHSS:
-
Ovarian hyperstimulation syndrome
- OR:
-
Odd ratio
- PCOS:
-
Polycystic ovarian syndrome
- PE:
-
Preeclampsia
- PGD:
-
Pre-implantation genetic diagnosis
- PIH:
-
Pregnancy-induced hypertension
- PRISMA:
-
Preferred reporting items for systematic reviews and meta-analyses
- PTB:
-
Preterm birth
- RCTs:
-
Randomized controlled trials
- RR:
-
Relative risk
- SART:
-
Society for assisted reproductive technology
- SET:
-
Single embryo transfer
- SGA:
-
Small for gestational age
- UK:
-
United Kingdom
- VLBW:
-
Very-low preterm birth
- VTS:
-
Vanishing twin syndrome
References
Legro RS, Myers E. Surrogate end-points or primary outcomes in clinical trials in women with polycystic ovary syndrome? Hum Reprod. 2004;19:1697–704.
Barnhart KT. Live birth is the correct outcome for clinical trials evaluating therapy for the infertile couple. Fertil Steril. 2014;101:1205–8.
Barnhart KT. Assisted reproductive technologies and perinatal morbidity: interrogating the association. Fertil Steril. 2013;99:299–302.
Bhattacharya S, Vanderpoel S, Bhattacharya S, Evers JL, Ng EH, Niederberger C, et al. Improving the Reporting of Clinical Trials of Infertility Treatments (IMPRINT): modifying the CONSORT statement. Fertil Steril. 2014;102:952–9.
Braakhekke M, Kamphuis EI, Van Rumste MM, Mol F, van der Veen F, Mol BW. How are neonatal and maternal outcomes reported in randomised controlled trials (RCTs) in reproductive medicine? Hum Reprod. 2014;29:1211–7.
Dhalwani NN, Boulet SL, Kissin DM, Zhang Y, McKane P, Bailey MA, et al. Assisted reproductive technology and perinatal outcomes: conventional versus discordant-sibling design. Fertil Steril. 2016. doi:10.1016/j.fertnstert.2016.04.038.
Sunderam S, Kissin DM, Crawford SB, Folger SG, Jamieson DJ, Warner L, Centers for Disease Control and Prevention (CDC), et al. Assisted Reproductive Technology Surveillance-United States, 2013. MMWR Surveill Summ. 2015;64:1–25.
Messerlian C, Maclagan L, Basso O. Infertility and the risk of adverse pregnancy outcomes: a systematic review and meta-analysis. Hum Reprod. 2013;28:125–37.
Pinborg A, Wennerholm UB, Romundstad LB, Loft A, Aittomaki K, Soderstrom-Anttila V, et al. Why do singletons conceived after assisted reproduction technology have adverse perinatal outcome? Systematic review and meta-analysis. Hum Reprod Update. 2013;19:87–104.
Ensing S, Abu-Hanna A, Roseboom TJ, Rep** S, van der Veen F, Mol BW, et al. Risk of poor neonatal outcome at term after medically assisted reproduction: a propensity score-matched study. Fertil Steril. 2015;104:384–90.e1.
Palomba S, Santagni S, Gibbins K, La Sala GB, Silver RM. Pregnancy complications in spontaneous and assisted conceptions of women with infertility and factors of subfertility. A comprehensive review. Reprod Biomed Online. 2016; doi: 10.1016/j.rbmo.2016.08.007
Seggers J, Pontesilli M, Ravelli AC, Painter RC, Hadders-Algra M, Heineman MJ, et al. Effects of in vitro fertilization and maternal characteristics on perinatal outcomes: a population-based study using siblings. Fertil Steril. 2016;105:590–8.
Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. J Clin Epidemiol. 2009;62:1006–12.
Howick J, Glasziou P, Aronson JK. Evidence-based mechanistic reasoning. J R Soc Med. 2010;103:433–41.
Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64:383–94.
Helmerhorst FM, Perquin DA, Donker D, Keirse MJ. Perinatal outcome of singletons and twins after assisted conception: a systematic review of controlled studies. BMJ. 2004;328:261–5.
McDonald S, Murphy K, Beyene J, Ohlsson A. Perinatal outcomes of in vitro fertilization twins: a systematic review and meta-analyses. Am J Obstet Gynecol. 2005;193:141–52.
Pandey S, Shetty A, Hamilton M, Bhattacharya S, Maheshwari A. Obstetric and perinatal outcomes in singleton pregnancies resulting from IVF/ICSI: a systematic review and meta-analysis. Hum Reprod Update. 2012;18:485–503.
Qin J, Liu X, Sheng X, Wang H, Gao S. Assisted reproductive technology and the risk of pregnancy-related complications and adverse pregnancy outcomes in singleton pregnancies: a meta-analysis of cohort studies. Fertil Steril. 2016;105:73–85.
Wang YA, Chugtai AA, Farquhar CM, Pollock W, Lui K, Sullivan EA. Increased incidence of gestational hypertension and preeclampsia after assisted reproductive technology treatment. Fertil Steril. 2016;105:920–926.e2.
Cromi A, Candeloro I, Marconi N, Casarin J, Serati M, Agosti M, et al. Risk of peripartum hysterectomy in births after assisted reproductive technology. Fertil Steril. 2016. doi:10.1016/j.fertnstert.2016.05.005.
Qin J, Wang H, Sheng X, Liang D, Tan H, **a J. Pregnancy-related complications and adverse pregnancy outcomes in multiple pregnancies resulting from assisted reproductive technology: a meta-analysis of cohort studies. Fertil Steril. 2015;103:1492–508.
Qin JB, Wang H, Sheng X, **e Q, Gao S. Assisted reproductive technology and risk of adverse obstetric outcomes in dichorionic twin pregnancies: a systematic review and meta-analysis. Fertil Steril. 2016;105:1180–92.
Declercq E, Luke B, Belanoff C, Cabral H, Diop H, Gopal D, et al. Perinatal outcomes associated with assisted reproductive technology: the Massachusetts Outcomes Study of Assisted Reproductive Technologies (MOSART). Fertil Steril. 2015;103:888–95.
Jauniaux E, Ben-Ami I, Maymon R. Do assisted-reproduction twin pregnancies require additional antenatal care? Reprod Biomed. 2013;26:107–19.
Simões T, Queirós A, Marujo AT, Valdoleiros S, Silva P, Blickstein I. Outcome of monochorionic twins conceived by assisted reproduction. Fertil Steril. 2015;104:629–32.
Bensdorp AJ, Hukkelhoven CW, van der Veen F, Mol BW, Lambalk CB, Van Wely M. Dizygotic twin pregnancies after medically assisted reproduction and after natural conception: maternal and perinatal outcomes. Fertil Steril. 2016;106:371–7.
McLernon DJ, Harrild K, Bergh C, Davies MJ, De Neubourg D, Dumoulin JC, et al. Clinical effectiveness of elective single versus double embryo transfer: meta-analysis of individual patient data from randomised trials. BMJ. 2010;341:c6945.
Kissin DM, Kulkarni AD, Kushnir VA, Jamieson DJ, National ART Surveillance System Group. Number of embryos transferred after in vitro fertilization and good perinatal outcome. Obstet Gynecol. 2014;123:239–47.
De Sutter P, Delbaere I, Gerris J, Verstraelen H, Goetgeluk S, Van der Elst J, et al. Birthweight of singletons after assisted reproduction is higher after single- than after double-embryo transfer. Hum Reprod. 2006;21:2633–7.
Wang YA, Sullivan EA, Healy DL, Black DA. Perinatal outcomes after assisted reproductive technology treatment in Australia and New Zealand: single versus double embryo transfer. Med J Aust. 2009;190:234–7.
Poikkeus P, Gissler M, Unkila-Kallio L, Hyden-Granskog C, Tiitinen A. Obstetric and neonatal outcome after single embryo transfer. Hum Reprod. 2007;22:1073–9.
Sazonova A, Källen K, Thurin-Kjellberg A, Wennerholm UB, Bergh C. Obstetric outcome after in vitro fertilization with single or double embryo transfer. Hum Reprod. 2011;26:442–50.
Levi S. Ultrasonic assessment of the high rate of human multiple pregnancy in the first trimester. J Clin Ultrasound. 1976;4:3–5.
Márton V, Zádori J, Kozinszky Z, Keresztúri A. Prevalences and pregnancy outcome of vanishing twin pregnancies achieved by in vitro fertilization versus natural conception. Fertil Steril. 2016. doi:10.1016/j.fertnstert.2016.07.1098.
Pinborg A, Lidegaard O, La Cour Freiesleben N, Nyboe Andersen A. Consequences of vanishing twins in IVF/ICSI pregnancies. Hum Reprod. 2005;20:2821–9.
La Sala GB, Villani MT, Nicoli A, Gallinelli A, Nucera G, Blickstein I. Effect of the mode of assisted reproductive technology conception on obstetric outcomes for survivors of the vanishing twin syndrome. Fertil Steril. 2006;86:247–9.
Luke B, Brown MB, Grainger DA, Stern JE, Klein N, Cedars MI. The effect of early fetal losses on singleton assisted-conception pregnancy outcomes. Fertil Steril. 2009;91:2578–85.
Pinborg A, Lidegaard O, Freiesleben N, Andersen AN. Vanishing twins: a predictor of small-for-gestational age in IVF singletons. Hum Reprod. 2007;22:2707–14.
Shebl O, Ebner T, Sommergruber M, Sir A, Tews G. Birth weight is lower for survivors of the vanishing twin syndrome: a case–control study. Fertil Steril. 2008;90:310–4.
Van Oppenraaij RH, Jauniaux E, Christiansen OB, Horcajadas JA, Farquharson RG, Exalto N, ESHRE Special Interest Group for Early Pregnancy (SIGEP). Predicting adverse obstetric outcome after early pregnancy events and complications: a review. Hum Reprod. 2009;15:409–21.
Evron E, Sheiner E, Friger M, Sergienko R, Harlev A. Vanishing twin syndrome: is it associated with adverse perinatal outcome? Fertil Steril. 2015;103:1209–14.
Nakashima A, Araki R, Tani H, Ishihara O, Kuwahara A, Irahara M, et al. Implications of assisted reproductive technologies on term singleton birth weight: an analysis of 25,777 children in the national assisted reproduction registry of Japan. Fertil Steril. 2013;99:450–5.
Malchau SS, Loft A, Henningsen AK, Nyboe Andersen A, Pinborg A. Perinatal outcomes in 6,338 singletons born after intrauterine insemination in Denmark, 2007 to 2012: the influence of ovarian stimulation. Fertil Steril. 2014;102:1110–6.
Griesinger G, Kolibianakis EM, Diedrich K, Ludwig M. Ovarian stimulation for IVF has no quantitative association with birth weight: a registry study. Hum Reprod. 2008;23:2549–54.
Budinetz TH, Mann JS, Griffin DW, Benadiva CA, Nulsen JC, Engmann LL. Maternal and neonatal outcomes after gonadotropin-releasing hormone agonist trigger for final oocyte maturation in patients undergoing in vitro fertilization. Fertil Steril. 2014;102:753–8.
Lainas GT, Kolibianakis EM, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas TG, et al. Pregnancy and neonatal outcomes following luteal GnRH antagonist administration in patients with severe early OHSS. Hum Reprod. 2013;28:1929–42.
Weiss A, Beck-Fruchter R, Golan J, Lavee M, Geslevich Y, Shalev E. Ectopic pregnancy risk factors for ART patients undergoing the GnRH antagonist protocol: a retrospective study. Reprod Biol Endocrinol. 2016;14:12.
Sunkara SK, La Marca A, Seed PT, Khalaf Y. Increased risk of preterm birth and low birthweight with very high number of oocytes following IVF: an analysis of 65 868 singleton live birth outcomes. Hum Reprod. 2015;30:1473–80.
Kalra SK, Ratcliffe SJ, Coutifaris C, Molinaro T, Barnhart KT. Ovarian stimulation and low birth weight in newborns conceived through in vitro fertilization. Obstet Gynecol. 2011;118:863–71.
Farhi J, Ben-Haroush A, Andrawus N, Pinkas H, Sapir O, Fisch B, et al. High serum oestradiol concentrations in IVF cycles increase the risk of pregnancy complications related to abnormal placentation. Reprod Biomed. 2010;21:331–7.
Imudia AN, Awonuga AO, Doyle JO, Kaimal AJ, Wright DL, Toth TL, et al. Peak serum estradiol level during controlled ovarian hyperstimulation is associated with increased risk of small for gestational age and preeclampsia in singleton pregnancies after in vitro fertilization. Fertil Steril. 2012;97:1374–9.
Imudia AN, Awonuga AO, Kaimal AJ, Wright DL, Styer AK, Toth TL. Elective cryopreservation of all embryos with subsequent cryothaw embryo transfer in patients at risk for ovarian hyperstimulation syndrome reduces the risk of adverse obstetric outcomes: a preliminary study. Fertil Steril. 2013;99:168–73.
Wiser A, Levron J, Kreizer D, Achiron R, Shrim A, Schiff E, et al. Outcome of pregnancies complicated by severe ovarian hyperstimulation syndrome (OHSS): a follow-up beyond the second trimester. Hum Reprod. 2005;20:910–4.
Courbiere B, Oborski V, Braunstein D, Desparoir A, Noizet A, Gamerre M. Obstetric outcome of women with in vitro fertilization pregnancies hospitalized for ovarian hyperstimulation syndrome: a case–control study. Fertil Steril. 2011;95:1629–32.
Haas J, Baum M, Meridor K, Hershko-Klement A, Elizur S, Hourvitz A, et al. Is severe OHSS associated with adverse pregnancy outcomes? Evidence from a case–control study. Reprod Biomed. 2014;29:216–21.
Calhoun KC, Fritz MA, Steiner AZ. Examining the relationship between ovarian reserve, as measured by basal FSH levels, and the risk of poor obstetric outcome in singleton IVF gestations. Hum Reprod. 2011;26:3424–30.
Haas J, Yinon Y, Meridor K, Orvieto R. Pregnancy outcome in severe OHSS patients following ascitic/plerural fluid drainage. J Ovarian Res. 2014;7:56.
Maheshwari A, Kalampokas T, Davidson J, Bhattacharya S. Obstetric and perinatal outcomes in singleton pregnancies resulting from the transfer of blastocyst-stage versus cleavage-stage embryos generated through in vitro fertilization treatment: a systematic review and meta-analysis. Fertil Steril. 2013;100:1615–21.
Dar S, Lazer T, Shah PS, Librach CL. Neonatal outcomes among singleton births after blastocyst versus cleavage stage embryo transfer: a systematic review and meta-analysis. Hum Reprod. 2014;20:439–48.
Kalra SK, Ratcliffe SJ, Barnhart KT, Coutifaris C. Extended embryo culture and an increased risk of preterm delivery. Obstet Gynecol. 2012;120:69–75.
Oron G, Sokal-Arnon T, Son WY, Demirtas E, Buckett W, Zeadna A, et al. Extended embryo culture is not associated with increased adverse obstetric or perinatal outcome. Am J Obstet Gynecol. 2014;211:165.e1–7.
Oron G, Nayot D, Son WY, Holzer H, Buckett W, Tulandi T. Obstetric and perinatal outcome from single cleavage transfer and single blastocyst transfer: a matched case–control study. Gynecol Endocrinol. 2015;31:469–72.
Chambers GM, Chughtai AA, Farquhar CM, Wang YA. Risk of preterm birth after blastocyst embryo transfer: a large population study using contemporary registry data from Australia and New Zealand. Fertil Steril. 2015;104:997–1003.
Mäkinen S, Söderström-Anttila V, Vainio J, Suikkari AM, Tuuri T. Does long in vitro culture promote large for gestational age babies? Hum Reprod. 2013;28:828–34.
Ginström Ernstad E, Bergh C, Khatibi A, Källén KB, Westlander G, Nilsson S, et al. Neonatal and maternal outcome after blastocyst transfer: a population-based registry study. Am J Obstet Gynecol. 2016;214:378.e1–378.e10.
Grace KS, Sinclair KD. Assisted reproductive technology, epigenetics, and long-term health: a developmental time bomb still ticking. Semin Reprod Med. 2009;27:409–16.
Chason RJ, Csokmay J, Segars JH, DeCherney AH, Armant DR. Environmental and epigenetic effects upon preimplantation embryo metabolism and development. Trends Endocrinol Metab. 2011;22:412–20.
Rizos D, Lonergan P, Boland MP, Arroyo-García R, Pintado B, de la Fuente J, et al. Analysis of differential messenger RNA expression between bovine blastocysts produced in different culture systems: implications for blastocyst quality. Biol Reprod. 2002;66:589–95.
Chang HJ, Lee JR, Jee BC, Suh CS, Kim SH. Impact of blastocyst transfer on offspring sex ratio and the monozygotic twinning rate: a systematic review and meta-analysis. Fertil Steril. 2009;91:2381–90.
Shiozaki A, Yoneda S, Nakabayashi M, Takeda Y, Takeda S, Sugimura M, et al. Multiple pregnancy, short cervix, part-time worker, steroid use, low educational level and male fetus are risk factors for preterm birth in Japan: a multicenter, prospective study. J Obstet Gynaecol Res. 2014;40:53–61.
Maheshwari A, Hamilton M, Bhattacharya S. Should we be promoting embryo transfer at blastocyst stage? Reprod Biomed. 2016;32:142–6.
Lemmen JG, Pinborg A, Rasmussen S, Ziebe S. Birthweight distribution in ART singletons resulting from embryo culture in two different culture media compared with the national population. Hum Reprod. 2014;29:2326–32.
Zhu J, Li M, Chen L, Liu P, Qiao J. The protein source in embryo culture media influences birthweight: a comparative study between G1 v5 and G1-PLUS v5. Hum Reprod. 2014;29:1387–92.
De Vos A, Janssens R, Vande Velde H, Haentjens P, Bonduelle M, Tournaye H, et al. The type of culture medium and the duration of in vitro culture do not influence birthweight of ART singletons. Hum Reprod. 2015;30:20–7.
Roque M, Lattes K, Serra S, Sola I, Geber S, Carreras R, et al. Fresh embryo transfer versus frozen embryo transfer in in vitro fertilizationcycles: a systematic review and meta-analysis. Fertil Steril. 2013;99:156–62.
Evans J, Hannan NJ, Edgell TA, Vollenhoven BJ, Lutjen PJ, Osianlis T, et al. Fresh versus frozen embryo transfer: backing clinical decisions with scientific and clinical evidence. Hum Reprod. 2014;20:808–21.
Londra L, Moreau C, Strobino D, Garcia J, Zacur H, Zhao Y. Ectopic pregnancy after in vitro fertilization: differences between fresh and frozen-thawed cycles. Fertil Steril. 2015;104:110–8.
Fang C, Huang R, Wei LN, Jia L. Frozen-thawed day 5 blastocyst transfer is associated with a lower risk of ectopic pregnancy than day 3 transfer and fresh transfer. Fertil Steril. 2015;103:655–61.
Wennerholm UB, Soderstrom-Anttila V, Bergh C, Aittomaki K, Hazekamp J, Nygren KG, et al. Children born after cryopreservation of embryos or oocytes: a systematic review of outcome data. Hum Reprod. 2009;24:2158–72.
Maheshwari A, Pandey S, Shetty A, Hamilton M, Bhattacharya S. Obstetric and perinatal outcomes in singleton pregnancies resulting from the transfer of frozen thawed versus fresh embryos generated through in vitro fertilization treatment: a systematic review and meta-analysis. Fertil Steril. 2012;98:368–77.
Kalra SK, Ratcliffe SJ, Milman L, Gracia CR, Coutifaris C, Barnhart KT. Perinatal morbidity after in vitro fertilization is lower with frozen embryo transfer. Fertil Steril. 2011;95:548–53.
Wennerholm UB, Henningsen AK, Romundstad LB, Bergh C, Pinborg A, Skjaerven R, et al. Perinatal outcomes of children born after frozen-thawed embryo transfer: a Nordic cohort study from the CoNARTaS group. Hum Reprod. 2013;28:2545–53.
Sazonova A, Källen K, Thurin-Kjellberg A, Wennerholm UB, Bergh C. Obstetric outcome in singletons after in vitro fertilization with cryopreserved/thawed embryos. Hum Reprod. 2012;27:1343–50.
Pelkonen S, Koivunen R, Gissler M, Nuojua-Huttunen S, Suikkari AM, Hyden-Granskog C, et al. Perinatal outcome of children born after frozen and fresh embryo transfer: the Finnish cohort study 1995–2006. Hum Reprod. 2010;25:914–23.
Pinborg A, Loft A, Aaris Henningsen AK, Rasmussen S, Andersen AN. Infant outcome of 957 singletons born after frozen embryo replacement: the Danish National Cohort Study 1995–2006. Fertil Steril. 2010;94:1320–7.
Kato O, Kawasaki N, Bodri D, Kuroda T, Kawachiya S, Kato K, et al. Neonatal outcome and birth defects in 6623 singletons born following minimal ovarian stimulation and vitrified versus fresh single embryo transfer. Eur J Obstet Gynecol Reprod Biol. 2012;161:46–50.
Mansour R, Ishihara O, Adamson GD, Dyer S, De Mouzon J, Nygren KG, et al. International Committee for Monitoring Assisted Reproductive Technologies world report: Assisted Reproductive Technology 2006. Hum Reprod. 2014;29:1536–51.
Ishihara O, Araki R, Kuwahara A, Itakura A, Saito H, Adamson GD. Impact of frozen-thawed single-blastocyst transfer on maternal and neonatal outcome: an analysis of 277,042 single-embryo transfer cycles from 2008 to 2010 in Japan. Fertil Steril. 2014;101:128–33.
Kaser DJ, Melamed A, Bormann CL, Myers DE, Missmer SA, Walsh BW, et al. Cryopreserved embryo transfer is an independent risk factor for placenta accreta. Fertil Steril. 2015;103:1176–84.e2.
Opdahl S, Henningsen AA, Tiitinen A, Bergh C, Pinborg A, Romundstad PR, et al. Risk of hypertensive disorders in pregnancies following assisted reproductive technology: a cohort study from the CoNARTaS group. Hum Reprod. 2015;30:1724–31.
Chen ZJ, Shi Y, Sun Y, Zhang B, Liang X, Cao Y, et al. Fresh versus Frozen Embryos for Infertility in the Polycystic Ovary Syndrome. N Engl J Med. 2016;375:523–33.
Nakashima A, Araki R, Tani H, Ishihara O, Kuwahara A, Irahara M, et al. Implications of assisted reproductive technologies on term singleton birth weight: an analysis of 25,777 children in the national assisted reproduction registry of Japan. Fertil Steril. 2012;282:2246–7.
Dumoulin JC, Land JA, Van Montfoort AP, Nelissen EC, Coonen E, Derhaag JG, et al. Effect of in vitro culture of human embryos on birthweight of newborns. Hum Reprod. 2010;25:605–12.
Nelissen EC, Van Montfoort AP, Coonen E, Derhaag JG, Geraedts JP, Smits LJ, et al. Further evidence that culturemedia affect perinatal outcome: findings after transfer of fresh and cryopreserved embryos. Hum Reprod. 2012;27:1966–76.
Vergouw CG, Kostelijk EH, Doejaaren E, Hompes PG, Lambalk CB, Schats R. The influence of the type of embryo culture medium on neonatal birthweight after single embryo transfer in IVF. Hum Reprod. 2012;27:2619–26.
De Geyter C, De Geyter M, Steimann S, Zhang H, Holzgreve W. Comparative birth weights of singletons born after assisted reproduction and natural conception in previously infertile women. Hum Reprod. 2006;21:705–12.
Pinborg A, Henningsen AA, Loft A, Malchau SS, Forman J, Andersen AN. Large baby syndrome in singletons born after frozen embryo transfer (FET): is it due to maternal factors or the cryotechnique? Hum Reprod. 2014;29:618–27.
Maheshwari A, Raja EA, Bhattacharya S. Obstetric and perinatal outcomes after either fresh or thawed frozen embryo transfer: an analysis of 112,432 singleton pregnancies recorded in the Human Fertilisation and Embryology Authority anonymized dataset. Fertil Steril. 2016;24.
Li Z, Sullivan EA, Chapman M, Farquhar C, Wang YA. Risk of ectopic pregnancy lowest with transfer of single frozen blastocyst. Hum Reprod. 2015;30:2048–54.
Mateizel I, Santos-Ribeiro S, Done E, Van Landuyt L, Van de Velde H, Tournaye H, et al. Do ARTs affect the incidence of monozygotic twinning? Hum Reprod. 2016 Sep 22.
Vajta G, Holm P, Greve T, Callesen H. Overall efficiency of in vitro embryo production and vitrification in cattle. Theriogenology. 1996;45:683–9.
Kuwayama M, Vajta G, Kato O, Leibo SP. Highly efficient vitrification method for cryopreservation of human oocytes. Reprod Biomed. 2005;11:300–8.
Vajta G, Kuwayama M. Improving cryopreservation systems. Theriogenology. 2006;65:236–44.
Chian RC, Huang JY, Gilbert L, Son WY, Holzer H, Cui SJ, et al. Obstetric outcomes following vitrification of in vitro and in vivo matured oocytes. Fertil Steril. 2009;91:2391–8.
Levi Setti PE, Albani E, Morenghi E, Morreale G, Delle Piane L, Scaravelli G, et al. Comparative analysis of fetal and neonatal outcomes of pregnancies from fresh and cryopreserved/thawed oocytes in the same group of patients. Fertil Steril. 2013;100:396–401.
Cobo A, Serra V, Garrido N, Olmo I, Pellicer A, Remohì J. Obstetric and perinatal outcome of babies born from vitrified oocytes. Fertil Steril. 2014;102:1006–15.
Takahashi K, Mukaida T, Goto T, Oka C. Perinatal outcome of blastocyst transfer with vitrification using cryoloop: a 4-year follow-up study. Fertil Steril. 2005;84:88–92.
Rama Raju GA, Jaya Prakash G, Murali Krishna K, Madan K. Neonatal outcome after vitrified day 3 embryo transfers: a preliminary study. Fertil Steril. 2009;92:143–8.
Desai N, Abdelhafez F, Bedaiwy MA, Goldberg J, Falcone T, Goldfarb J. Clinical pregnancy and live births after transfer of embryos vitrified on day 3. Reprod Biomed. 2010;20:808–13.
Shi W, Xue X, Zhang S, Zhao W, Liu S, Zhou H, et al. Perinatal and neonatal outcomes of 494 babies delivered from 972 vitrified embryo transfers. Fertil Steril. 2012;97:1338–42.
Liu SY, Teng B, Fu J, Li X, Zheng Y, Sun XX. Obstetric and neonatal outcomes after transfer of vitrified early cleavage embryos. Hum Reprod. 2013;28:2093–100.
Kaartinen N, Kananen K, Huhtala H, Keränen S, Tinkanen H. The freezing method of cleavage stage embryos has no impact on the weight of the newborns. J Assist Reprod Genet. 2016;33:393–9.
Wikland M, Hardarson T, Hillensjo T, Westin C, Westlander G, Wood M, et al. Obstetric outcomes after transfer of vitrified blastocysts. Hum Reprod. 2010;25:1699–707.
Li Z, Wang YA, Ledger W, Edgar DH, Sullivan EA. Clinical outcomes following cryopreservation of blastocysts by vitrification or slow freezing: a population-based cohort study. Hum Reprod. 2014;29:2794–801.
Roy TK, Bradley CK, Bowman MC, McArthur SJ. Single-embryo transfer of vitrified-warmed blastocysts yields equivalent live-birth rates and improved neonatal outcomes compared with fresh transfers. Fertil Steril. 2014;101:1294–301.
Ozgur K, Berkkanoglu M, Bulut H, Humaidan P, Coetzee K. Perinatal outcomes after fresh versus vitrified-warmed blastocyst transfer: retrospective analysis. Fertil Steril. 2015;104:899–907.
Belva F, Bonduelle M, Roelants M, Verheyen G, Van Landuyt L. Neonatal health including congenital malformation risk of 1072 children born after vitrified embryo transfer. Hum Reprod. 2016;31:1610–20.
Coticchio G, Dal Canto M, Mignini Renzini M, Guglielmo MC, Brambillasca F, Turchi D, et al. Oocyte maturation: gamete-somatic cells interactions, meiotic resumption, cytoskeletal dynamics and cytoplasmic reorganization. Hum Reprod. 2015;21:427–54.
Siristatidis CS, Vrachnis N, Creatsa M, Maheshwari A, Bhattacharya S. In vitro maturation in subfertile women with polycystic ovarian syndrome undergoing assisted reproduction. Cochrane Database Syst Rev. 2013. doi:10.1002/14651858.CD006606.pub3.
Cha KY, Chung HM, Lee DR, Kwon H, Chung MK, Park LS, et al. Obstetric outcome of patients with polycystic ovary syndrome treated by in vitro maturation and in vitro fertilization-embryo transfer. Fertil Steril. 2005;83:1461–5.
Soderstrom-Anttila V, Makinen S, Tuuri T, Suikkari AM. Favourable pregnancy results with insemination of in vitro matured oocytes from unstimulated patients. Hum Reprod. 2005;20:1534–40.
Buckett WM, Chian RC, Holzer H, Dean N, Usher R, Tan SL. Obstetric outcomes and congenital abnormalities after in vitro maturation, in vitro fertilization, and intracytoplasmic sperm injection. Obstet Gynecol. 2007;110:885–91.
Buckett WM, Chian RC, Dean N, Sylvestre C, Holzer H, Tan SL. Pregnancy loss in pregnancies conceived after in vitro oocyte maturation, conventional in vitro fertilization, and intracytoplasmic sperm injection. Fertil Steril. 2008;110:885–91.
Fadini R, Mignini Renzini M, Guarnieri T, Dal Canto M, De Ponti E, Sutcliffe A, et al. Comparison of the obstetric and perinatal outcomes of children conceived from in vitro or in vivo matured oocytes in vitro maturation treatments with births from conventional ICSI cycles. Hum Reprod. 2012;27:3601–8.
Anckaert E, De Rycke M, Smitz J. Culture of oocytes and risk of imprinting defects. Hum Reprod. 2013;19:52–66.
Palomba S, De Wilde MA, Falbo A, Koster MPH, La Sala GB, Fauser BCJM. Pregnancy complications in women with polycystic ovary syndrome: new clinical and pathophysiologic insight. Hum Reprod. 2015;21:575–92.
Hagemann AR, Lanzendorf SE, Jungheim ES, Chang AS, Ratts VS, Odem RR. A prospective, randomized, double-blinded study of assisted hatching in women younger than 38 years undergoing in vitro fertilization. Fertil Steril. 2010;93:586–91.
Zhou H, Zao W, Zhang W, Shi J, Shi W. No adverse effects were identified on the perinatal outcomes after laser-assisted hatching treatment. Reprod Biomed. 2014;29:692–8.
Knopman JM, Krey LC, Oh C, Lee J, McCaffrey C, Noyes N. What makes them split? Identifying risk factors that lead to monozygotic twins after in vitro fertilization. Fertil Steril. 2014;102:82–9.
Harper JC, Wilton L, Traeger-Synodinos J, Goossens V, Moutou C, Sengupta SB, et al. The ESHRE PGD Consortium: 10 years of data collection. Hum Reprod. 2012;18:234–47.
Desmyttere S, De Rycke M, Staessen C, Liebaers I, De Schrijver F, Verpoest W, et al. Neonatal follow-up of 995 consecutively born children after embryo biopsy for PGD. Hum Reprod. 2012;27:288–93.
Eldar-Geva T, Srebnik N, Altarescu G, Varshaver I, Brooks B, Levy-Lahad E, et al. Neonatal outcome after preimplantation genetic diagnosis. Fertil Steril. 2014;102:1016–21.
Chang J, Boulet SL, Jeng G, Flowers L, Kissin DM. Outcomes of in vitro fertilization with preimplantation genetic diagnosis: an analysis of the United States Assisted Reproductive Technology Surveillance Data, 2011–2012. Fertil Steril. 2016;105:394–400.
Bay B, Ingerslev HJ, Lemmen JG, Degn B, Rasmussen IA, Kesmodel US. Preimplantation genetic diagnosis: a national multicenter obstetric and neonatal follow-up study. Fertil Steril. 2016. doi:10.1016/j.fertnstert.2016.07.1092.
**g S, Luo K, He H, Lu C, Zhang S, Tan Y, et al. Obstetric and neonatal outcomes in blastocyst-stage biopsy with frozen embryo transfer and cleavage-stage biopsy with fresh embryo transfer after preimplantation genetic diagnosis/screening. Fertil Steril. 2016;106:105–112.e4.
Society of Obstetricians and Gynaecologists of Canada, Okun N, Sierra S. Pregnancy outcomes after assisted human reproduction. J Obstet Gynaecol Can. 2014;36:64–83.
Sheffer-Miouni G, Mashiach S, Dor J, Levran D, Seidman DS. Factors influencing the obstetric and perinatal outcome after oocyte donation. Hum Reprod. 2002;17:2636–40.
Rodríguez-González M, Serra V, Garcia-Velasco JA, Pellicer A, Remohí J. The “vanishing embryo” phenomenon in an oocyte donation programme. Hum Reprod. 2002;17:798–802.
Wiggins DA, Main E. Outcomes of pregnancies achieved by donor egg in vitro fertilization-a comparison with standard in vitro fertilization pregnancies. Am J Obstet Gynecol. 2005;192:2002–8.
Keegan DA, Krey LC, Chang HC, Noyes N. Increased risk of pregnancy induced hypertension in young recipients of donated oocytes. Fertil Steril. 2007;87:776–81.
Gundogan F, Bianchi DW, Scherjon SA, Roberts DJ. Placental pathology in egg donor pregnancies. Fertil Steril. 2010;93:397–404.
Zegers-Hochschild F, Masoli D, Schwarze JE, Iaconelli A, Borges E, Pacheco IM. Reproductive performance in oocyte donors and their recipients: comparative analysis from implantation to birth and lactation. Fertil Steril. 2010;93:2210–5.
Soderstrom-Anttila V, Tiitinen A, Foudila T, Hovatta O. Obstetric and perinatal outcome after oocyte donation: comparison with in-vitro fertilization pregnancies. Hum Reprod. 1998;13:483–90.
Klatsky PC, Delaney SS, Caughey AB, Tran ND, Scattman GL, Rosenwaks Z. The role of embryonic origin in preeclampsia. Obstet Gynecol. 2010;116:1387–92.
Le Ray C, Scherier S, Anselem O, Marszalek A, Tsatsaris V, Cabrol D, et al. Association between oocyte donation and maternal and perinatal outcomes in women aged 43 years or older. Hum Reprod. 2012;27:896–901.
Yaron Y, Ochshorn Y, Amit A, Kogosowski A, Yovel I, Lessing JB, et al. Oocyte donation in Israel: a study of 1001 initiated cycles. Hum Reprod. 1998;13:1819–24.
Levron Y, Dviri M, Segol I, Yerushalmi GM, Hourvitz A, Orvieto R, et al. The ‘immunologic theory’ of preeclampsia revisited: a lesson from donor oocyte gestations. Am J Obstet Gynecol. 2014;211:383.e1–5.
Jeve YB, Potdar N, Opoku A, Khare M. Three-arm age-matched retrospective cohort study of obstetric outcomes of donor oocyte pregnancies. Int J Gynaecol Obstet. 2016;133:156–8.
Gibbons WE, Cedars M, Ness RB. Toward understanding obstetrical outcome in advanced assisted reproduction: varying sperm, oocyte, and uterine source and diagnosis. Fertil Steril. 2011;95:1645–9.
Pecks U, Maass N, Neulen J. Oocyte donation: a risk factor for pregnancy-induced hypertension: a meta-analysis and case series. Dtsch Arztebl Int. 2011;108:23–31.
Adams DH, Clark RA, Davies MJ, De Lacey S. A meta-analysis of neonatal health outcomes from oocyte donation. J Dev Orig Health Dis. 2015;27:1–16.
Jeve YB, Potdar N, Opoku A, Khare M. Donor oocyte conception and pregnancy complications: a systematic review and meta-analysis. BJOG. 2016;123:1471–80.
Blázquez A, García D, Rodríguez A, Vassena R, Figueras F, Vernaeve V. Is oocyte donation a risk factor for preeclampsia? A systematic review and meta-analysis. J Assist Reprod Genet. 2016;33:855–63.
Storgaard M, Loft A, Bergh C, Wennerholm UB, Söderström-Anttila V, Romundstad LB, et al. Obstetric and neonatal complications in pregnancies conceived after oocyte donation - a systematic review and meta-analysis. BJOG. 2016. doi:10.1111/1471-0528.14257.
Malchau SS, Loft A, Larsen EC, Aaris Henningsen AK, Rasmussen S, Andersen AN, et al. Perinatal outcomes in 375 children born after oocyte donation: a Danish national cohort study. Fertil Steril. 2013;99:1637–43.
Savasi VM, Mandia L, Laoreti A, Cetin I. Maternal and fetal outcomes in oocyte donation pregnancies. Hum Reprod. 2016;22:620–33.
Dude AM, Yeh JS, Muasher SJ. Donor oocytes are associated with preterm birth when compared to fresh autologous in vitro fertilization cycles in singleton pregnancies. Fertil Steril. 2016. doi:10.1016/j.fertnstert.2016.05.029.
Salha O, Sharma V, Dada T, Nugent D, Rutherford AJ, Tomlinson AJ, et al. The influence of donated gametes on the incidence of hypertensive disorders of pregnancy. Hum Reprod. 1999;14:2268–77.
Dekker GA, Sibai BM. Etiology and pathogenesis of preeclampsia: current concepts. Am J Obstet Gynecol. 1998;179:1359–75.
Finger R, Sommerfelt C, Freeman M, Wilson CK, Wade A, Daly D. A cost-effectiveness comparison of embryo donation with oocyte donation. Fertil Steril. 2010;93:379–81.
Söderström-Anttila V, Wennerholm UB, Loft A, Pinborg A, Aittomäki K, Romundstad LB, Bergh C. Surrogacy: outcomes for surrogate mothers, children and the resulting families-a systematic review. Hum Reprod. 2016;22:260–76.
Dar S, Lazer T, Swanson S, Silverman J, Wasser C, Moskovtsev SI, et al. Assisted reproduction involving gestational surrogacy: an analysis of the medical, psychosocial and legal issues: experience from a large surrogacy program. Hum Reprod. 2015;30:345–52.
Perkins KM, Boulet SL, Jamieson DJ, Kissin DM, National Assisted Reproductive Technology Surveillance System (NASS) Group. Trends and outcomes of gestational surrogacy in the United States. Fertil Steril. 2016;106:435–42.
Turan N, Katari S, Gerson LF, Chalian R, Foster MW, Gaughan JP, et al. Inter- and intra-individual variation in allele-specific DNA methylation and gene expression in children conceived using assisted reproductive technology. PLoS Genet. 2010. doi:10.1371/journal.pgen.1001033.
Van der Hoorn MLP, Lashley EELO, Biannchi DW, Claas FHJ, Schonkerren CMC, Scherjon SA. Clinical and immunologic aspects of egg donation pregnancies: a systematic review. Hum Reprod. 2010;16:704–12.
Palomba S, Russo T, Falbo A, Di Cello A, Tolino A, Tucci L, et al. Macroscopic and microscopic findings of the placenta in women with polycystic ovary syndrome. Hum Reprod. 2013;28:2838–47.
Wehby GL, Murray JC, Castilla EE, Lopez-Camelo JS, Ohsfeldt RL. Prenatal care effectiveness and utilization in Brazil. Health Policy Plan. 2009;24:175–88.
Alibekova R, Huang JP, Chen YH. Adequate prenatal care reduces the risk of adverse pregnancy outcomes in women with history of infertility: a nationwide population-based study. PLoS One. 2013;8, e84237.
Ethics Committee of the American Society for Reproductive Medicine. Provision of fertility services for women at increased risk of complications during fertility treatment or pregnancy: an Ethics Committee opinion. Fertil Steril. 2016; doi:10.1016/j.fertnstert.2016.08.015.
Ethics Committee of the American Society for Reproductive Medicine. Oocyte or embryo donation to women of advanced reproductive age: an Ethics Committee opinion. Fertil Steril. 2016; doi:10.1016/j.fertnstert.2016.07.002.
Acknowledgements
No personal acknowledgement is declared.
Funding
No specific funding was sought for the study. Departmental funds from the Center of Reproductive Medicine and Surgery-ASMN-IRCCS of Reggio Emilia (Italy); Homerton Fertility Unit, Homerton University Hospital, Homerton Row, London (UK); and Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv (Israel) were used to support the Authors throughout the study period and manuscript preparation.
Availability of data and materials
Please contact author for data requests.
Authors’ contributions
SP conceived the study, performed the literature search and selected the papers, analyzed and interpreted data, wrote the manuscript and approved its final version; RH interpreted data, drafted the manuscript, revised the manuscript for intellectual content and approved its final version; SS performed the literature search and selected the papers, drafted the manuscript and approved its final version; GBLS analyzed data, revised the manuscript for intellectual content and approved its final version; RO interpreted data, drafted the manuscript, revised the manuscript for intellectual content and approved its final version.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Palomba, S., Homburg, R., Santagni, S. et al. Risk of adverse pregnancy and perinatal outcomes after high technology infertility treatment: a comprehensive systematic review. Reprod Biol Endocrinol 14, 76 (2016). https://doi.org/10.1186/s12958-016-0211-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12958-016-0211-8