Abstract
Background
The biologic heterogeneity of soft tissue sarcomas (STS), even within histological subtypes, complicates treatment. In earlier studies, gene expression patterns that distinguish two subsets of clear cell renal carcinoma (RCC), serous ovarian carcinoma (OVCA), and aggressive fibromatosis (AF) were used to separate 73 STS into two or four groups with different probabilities of develo** metastatic disease (PrMet). This study was designed to confirm our earlier observations in a larger independent data set.
Methods
We utilized these gene sets, hierarchical clustering (HC), and Kaplan-Meier analysis, to examine 309 STS, using Affymetrix chip expression profiling.
Results
HC using the combined AF-, RCC-, and OVCA-gene sets identified subsets of the STS samples. Analysis revealed differences in PrMet between the clusters defined by the first branch point of the clustering dendrogram (p = 0.048), and also among the four different clusters defined by the second branch points (p < 0.0001). Analysis also revealed differences in PrMet between the leiomyosarcomas (LMS), dedifferentiated liposarcomas (LipoD), and undifferentiated pleomorphic sarcomas (UPS) (p = 0.0004). HC of both the LipoD and UPS sample sets divided the samples into two groups with different PrMet (p = 0.0128, and 0.0002, respectively). HC of the UPS samples also showed four groups with different PrMet (p = 0.0007). HC found no subgroups of the LMS samples.
Conclusions
These data confirm our earlier studies, and suggest that this approach may allow the identification of more than two subsets of STS, each with distinct clinical behavior, and may be useful to stratify STS in clinical trials and in patient management.
Similar content being viewed by others
Background
Soft tissue sarcomas (STS) represent a diverse group of malignancies with different clinical behaviors. Adult STS can be grouped into two broad categories. One category has simple genomic profiles and specific cytogenetic changes, such as a point mutation or translocation (for example SYT-SSX in synovial sarcoma). The second category is comprised of tumors with more complex genomic patterns characterized by multiple gains and losses, including many leiomyosarcomas (LMS), pleomorphic liposarcomas, and undifferentiated pleomorphic sarcomas (UPS) (previously termed malignant fibrous histiocytomas) [1–5]. Although UPS may represent a distinct tumor entity, many UPS have mRNA expression profiles that are similar to other well defined subtypes of STS, including LMS and liposarcoma, although they are not easily recognized as such based on histology (http://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb5/bb5-classifsofttissue.pdf) [6–10].
While some differences in behavior generally correlate with histologic diagnosis and grade, significant heterogeneity of tumor biology exists even within histologic subsets. The heterogeneity of biological behavior complicates clinical care of patients with STS. One clinically important variable is whether a tumor will metastasize or not.
Gene expression patterns may be useful in the subclassification of STS, both for diagnosis and for prediction of clinical behavior [2, 7–16]. In some cases, gene expression patterns may correlate better with biological behavior than histology, and some studies have suggested that gene expression patterns may correlate with metastatic potential in some high-grade STS [11, 12, 14, 17]. A recent study identified a set of 67 genes involved in mitosis and chromosome integrity, termed the complexity index in sarcomas (CINSARC), that can predict metastasis outcome in non-translocation dependent STS [11] and also synovial sarcoma [18].
In earlier studies, we described gene expression profiles that identified two general subgroups in a set of clear cell renal cell carcinomas (ccRCC-gene set), a set of ovarian carcinomas (OVCA-gene set), and a set of aggressive fibromatosis samples (AF-gene set) [19–22]. We recently reported the use of a gene set derived from these three studies to separate 73 high grade STS into 2 or 4 groups with different propensity of metastasis [14]. Because the expression data for the STS sample set was limited since it was from a different platform than the Affymetrix system, we pooled the ccRCC-, OVCA-, and AF-gene sets for the earlier study.
In this study we confirmed the results from our earlier studies with an independent data set. We utilized our three gene sets to examine a larger group of 309 non-translocation associated STS using Affymetrix chip based expression profiling, in data sets in which all probes utilized in our earlier studies were represented. These gene sets successfully separated the STS samples into subsets with different probabilities of develo** metastases.
Methods
Samples
Three hundred nine STS samples were obtained from patients who had surgical resection of STS as previously described [11]. The samples were from the FSG database, part of the Conticabase (http://www.conticabse.org), and were treated in one of 11 centers. In each case, the pathology was reviewed by the pathologist subgroup and classified according to the 2002 WHO classification. For the current study, STS with no recurrent chromosomal translocation were selected. According to French law at the time of the study, experiments were performed in agreement with the Bioethics Law 2004 800 and the Ethics Charter from the National Institute of Cancer; all subjects signed a non-opposition statement for research use of their sample.
cDNA microarrays
Total RNA was extracted from each frozen tumor sample, and analyzed on Human Genome U133 Plus 2.0 array (Affymetrix) as previously described [11].
Gene sets
Three different gene sets with limited overlap, that identified two general subgroups of a series of ccRCC samples (ccRCC-gene set), two subgroups of AF (AF-gene set), and distinguished borderline from invasive serous OVCA (OVCA-gene set) have been previously described, including 138, 161, and 173 known genes respectively [19, 21, 22]. For the current study, these three gene sets were pooled resulting in a set of 533 probes.
Hierarchical clustering and fold-change analysis
The ccRCC-, AF-, and OVCA- gene sets were used individually or combined, to cluster the 309 primary high-grade STS samples. For clustering, genes were median centered, normalized, and then clustered by complete hierarchical clustering using uncentered correlation with the Eisen clustering software and viewed using the TreeView software (http://rana.lbl.gov). The geometric means of the expression intensities of the relevant gene fragments were computed using Genedata Expressionist (Genedata, Basel, Switzerland), and the ratio was reported as the fold change (up or down). Confidence intervals and p-values on the fold change were calculated using a two-sided Welch modified two-sample t-test. Differences were considered significant when p</=0.05.
Analysis of time to metastasis
For each data set, we used the Kaplan-Meier (K-M) method to calculate the metastasis-free survival probabilities, and the cumulative probabilities of metastasis (i.e. one minus survival probabilities) at critical time points (2, 3, 5, and 10 years). The p-values were calculated by using the Wilcoxon-Gehan test for comparing different groups. The Tukey-Kramer method was used to adjust for multiple pair-wise comparisons when there were more than two groups. P-values </=0.05 were considered statistically significant. Analyses were performed in SAS® 9.3 (SAS Institute, Cary, NC).
Results
Analysis of all STS samples using the combined gene set
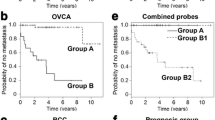
Hierarchical clustering of the 309 STS samples was first performed with the set of 533 probes of the combined gene set (ccRCC-gene set, OVCA-gene set, and AF-gene set) (Figure 1 top). Kaplan-Meier analysis was performed using the two sample sets defined by the first branch point (Groups A and B). The time to the development of metastasis was significantly different between the two sample sets (p = 0.048), with the probability of not develo** a metastasis by 3 years of 0.69 for Group A vs. 0.55 for Group B (Figure 1 middle, Additional file 1: Table S1A).
Top: clustering of gene expression in the 309 high-grade STS samples using the Eisen clustering software Cluster. The samples were clustered by complete-linkage hierarchical clustering with uncentered correlation using the Eisen clustering software Cluster and the 533 probes present in the pooled gene set as described in the text. Groups A and B are defined by the first branch point in the clustering. Groups 1–4 are defined by the second branch points. Middle and bottom: Kaplan-Meier analysis of the time to development of metastases of the two groups (groups A and B) and four groups (groups 1–4), defined by the first and second break points, respectively, of the hierarchical clustering. The time to development of metastasis differed between groups A and B (p = 0.048), and groups 1–4 (p < 0.0001) (see Additional file 1: Table S1).
Kaplan-Meier analysis was also performed using the four sample sets defined by the first and second branch points in the hierarchical clustering analysis (Groups 1–4 in Figure 1 bottom). The time to the development of metastasis differed among the four groups (p < 0.0001), with the probability of remaining free of metastasis by 5 years being 0.15, 0.77, 0.40, and 0.61 for groups 1, 2, 3, and 4, respectively (Figure 1 bottom, Additional file 1: Table S1B). Group comparisons by the Tukey-Kramer method revealed significant differences between groups 1 and 2 (p < 0.0001), 1 and 4 (p = 0.0237), 2 and 3 (p < 0.0001), and 2 and 4 (p = 0.0314).
Analysis of histologic subtypes
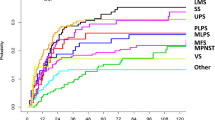
In examining Figure 1 bottom, we noticed that leiomyosarcoma (LMS) samples were preferentially enriched in group 1, and questioned the impact of differential mixtures of histologic diagnoses to the observed results. We therefore questioned whether there was a difference in probability of metastasis between the LMS, dedifferentiated liposarcoma (LipoD), undifferentiated pleomorphic sarcomas (UPS), and other sarcoma samples (OTH) (which include all other diagnoses as described in reference 11) in our sample set. Analysis revealed that LMS had a higher incidence of metastasis than the other three diagnostic groups (p = 0.0004) (Figure 2, Additional file 1: Table S1C). Group comparisons by the Tukey-Kramer method showed significant differences between LMS and LipoD (p = 0.0004), OTH (p = 0.0016), and UPS (p = 0.0138).
Kaplan-Meier analysis of the time to development of metastases of the four groups of LMS, LipoD, UPS, and OTH samples. The time to development of metastasis differed between the groups, p = 0.0004 (see Additional file 1: Table S1).
Analysis of histologic subtypes using the combined gene set
We next performed hierarchical clustering of the four individual histologic types of STS (LipoD, UPS, LMS, and OTH). Hierarchical clustering of the 62 LipoD samples with the pooled probe set divided the samples into two groups (A and B) with different probabilities of metastasis (Figure 3 top, Additional file 1: Table S1D) (p = 0.0128).
Kaplan-Meier analysis of the time to development of metastases of the two groups of LipoD samples (groups A and B), defined by the first break point of the hierarchical clustering with the pooled probe set (top panel). The time to development of metastasis differed between groups A and B, p = 0.0128. Analysis of the time to development of metastases of the two groups (groups A and B) (middle panel), and four groups (groups 1–4) of UPS samples (bottom panel) defined by the first and second break points, respectively, of the hierarchical clustering with the pooled probe set. The time to development of metastasis differed between groups A and B, p = 0.0002, and groups 1–4, p = 0.0007 (see Additional file 1: Table S1).
Hierarchical clustering of the 136 UPS samples with the pooled probe set divided the samples into two groups (A and B) with different probabilities of metastasis (Figure 3 middle, Additional file 1: Table S1E) (p = 0.0002). Analysis as four groups (1–4) also revealed statistically significant differences (p = 0.0007) (Figure 3 bottom, Additional file 1: Table S1F), suggesting further heterogeneity in this tumor subset. Group comparisons by the Tukey-Kramer method revealed significant differences between groups 1 and 3 (p = 0.0012), and between groups 2 and 3 (p = 0.0051), while the other group comparisons did not reach statistical significance.
In contrast, when the 84 LMS samples were analyzed in the same manner, analysis of the groups formed by the first 2 or 4 clusters defined by the hierarchical clustering found no statistically significant differences (not shown). Similarly, no difference was observed when a similar analysis was performed using the 27 miscellaneous samples in the OTH subgroup (not shown). Factors possibly contributing to the inability to identify groups with different outcomes in the LMS and OTH sets could include the small number of samples in these two subgroups, and the relative mix of “low” and “high” metastatic samples in these two sample sets.
Analysis of histologic subtypes using the individual RCC-, OVCA-, and AF-gene sets
We also analyzed the STS samples with the individual AF-, OVCA-, and RCC-probe sets. Analysis of the UPS subset with the RCC-gene set identified differences in time to metastasis when the UPS samples were analyzed as two groups (p = 0.0002) or four groups (p < 0.0007)(Figure 4, top row, Additional file 1: Table S1 G,H). Group comparisons by the Tukey-Kramer method revealed significant differences between groups 1 and 4 (p = 0.0159) and 2 and 4 (p = 0.0005) with a trend between 2 and 3 (p = 0.0971). Analysis of the UPS samples with the AF-gene set revealed differences in time to metastasis when analyzed as two groups (p = 0.0007) or four groups (p = 0.008) (Figure 4, second row, Additional file 1: Table S1 I,J). Group comparisons by the Tukey-Kramer method revealed significant differences between groups 1 and 3 (p = 0.0500), and 2 and 3 (p = 0.0422), with a trend between groups 2 and 4 (p = 0.1196) and 1 and 4 (p = 0.1451). Analysis of the UPS samples with the OVCA-gene set found differences in time to metastasis when the samples were analyzed as two (p = 0.004), or five subgroups (Figure 4, third row, Additional file 1: Table S1, K,L) (p = 0.0009). Group comparisons by the Tukey-Kramer method revealed significant differences between groups 1 and 3 (p = 0.0303) and 2 and 3 (p = 0.0004).
Top panel: Kaplan-Meier analysis of the time to development of metastases of the two groups (groups A and B) and the four groups (groups 1–4) of UPS samples, defined by the first and second break points of the hierarchical clustering with the RCC-gene set. The time to development of metastasis differed between groups A and B, p = 0.0002 and groups 1–4, p < 0.0007 (see Additional file 1: Table S1). Second row: Kaplan-Meier analysis of the time to development of metastases of the two (groups A and B) and four groups (groups 1–4) of UPS samples defined by the first and second break points, respectively, of the hierarchical clustering with the AF-gene set. The time to development of metastasis differed between groups A and B, p = 0.0007, and groups 1–4, p = 0.008 (see Additional file 1: Table S1). Third row: Kaplan-Meier analysis of the time to development of metastases of the two groups (groups A and B), and five groups (groups 1–5) of the UPS samples, defined by the first break point, and visual separation of the break points, respectively, of the hierarchical clustering with the OVCA-gene set. The time to development of metastasis differed between groups A and B, p = 0.004, and between groups 1–5, p = 0.0009 (see Additional file 1: Table S1). Bottom row: Kaplan-Meier analysis of the time to development of metastases of the four groups of LipoD samples defined by the second break points of the hierarchical clustering with the AF-gene set, p = 0.011 (see Additional file 1: Table S1).
When the AF-gene set was used to cluster the LipoD samples and analyzed as two groups the difference in time to metastasis did not reach statistical significance (not shown). Although when analyzed as four subgroups, statistically significant differences in time to metastasis were observed (Figure 4 bottom, Additional file 1: Table S1 M) (p = 0.011). Group comparisons by the Tukey-Kramer method revealed significant differences between groups 2 and 4 (p = 0.0384) and 3 and 4 (p = 0.0220). When either the RCC-gene set or the OVCA-gene set was used to cluster the LipoD samples, no significant difference in time to metastasis was noted when analyzed as 2, 4, or 5 groups, although a trend was suggested (not shown). None of the individual AF-, RCC-, or OVCA- gene sets separated the LMS or OTH samples into groups of different metastatic propensity.
Effect of tumor grade on probability of develo** metastases
Tumor grade is correlated with survival in STS, and we therefore questioned whether this might contribute to our observations. We performed Kaplan-Meier analyses of the LipoD and UPS subgroups defined by clustering with the total gene set as in Figure 3 top and middle, but excluded the 7 LipoD and 7 UPS samples that were histologically grade 1 tumors. In the case of the LipoD samples, the samples present in the first two groups defined by clustering still differed in the probability of develo** metastases (p = 0.0545) (not shown). Similarly, when the UPS samples were analyzed without grade 1 tumors, a difference in probability of metastasis was observed when analyzed as two groups (p = 0.0004) or four groups (p = 0.001) (not shown). Group comparisons using the Tukey-Kramer method found significant differences between groups 1 and 3 (p = 0.002) and 2 and 3 (p = 0.006). Therefore, the subgroups identified by clustering in Figure 3 were not accounted for by differences in content of grade 1 tumors.
Effect of stage on probability of develo** metastasis
Since clinical grade is correlated with survival in STS, we questioned whether this might contribute to our observations. When the LipoD samples were analyzed by AJC (American Joint Commission) stage, no significant difference in the probability of develo** metastases was seen between stage III (n = 17) vs IIA (n = 2), III vs IIB (n = 26), or IIA vs IIB, though the difference between stage III and IIA trended toward significance at p = 0.15 by Tukey-Kramer, and the rank order was as expected. When the UPS samples were analyzed by stage (IIA, n = 23; IIB, n = 30; III, n = 65), the three stages also did not differ in risk of metastases, though the difference between stage IIA and III and that between stage IIB and III also trended to significance, both at p = 0.07, and the rank order was again as expected. The lack of good correlation between stage and metastases seen here may reflect the limited number of cases examined in the different stages, and the fact that most cases were either IIA, IIB, or III.
Correlations between AJC stage and clustering
For the UPS samples, AJC stage was significantly correlated with both the two groups and four groups defined by clustering (Figure 3, middle and bottom) (p = 0.03 and 0.02, respectively). For the LipoD samples, AJC stage was not significantly correlated with the two groups formed by clustering (Figure 3, top).
Differential gene expression between subgroups
We next examined the most differentially expressed genes of the total detected in the Affymetrix U133 Plus chip set, in the first two subsets (groups A and B) of LipoD and UPS (Figure 3, top and middle). Fold change analysis was performed as described in the Methods, and the genes most over- or under-expressed between the groups are shown for LipoD in Additional file 2: Table S2 and for UPS in Additional file 3: Table S3.
When the global gene expression of the LipoD-A subset was compared with the LipoD-B subset (Figure 3 top), many differences were found (Additional file 2: Table S2). For example, in the LipoD-A subset (the better prognosis group) several genes that would be expected to be expressed in adipocytes, including: ADIPOQ, PLIN1, FABP4, LPL, PPARG, and THRSP were over-expressed compared with the LipoD-B subset, as well as ALDH1, ADH1B, and NTRK2 (Additional file 2: Table S2A). This may correlate with a less “de-differentiated” state. In contrast, genes over-expressed in the LipoD-B subset (the worse prognosis group) compared to the LipoD-A subset included: RUNX2, CDC20, TOP2A, ANLN, multiple members of the kinesin family, CDK1, AURKA, CCNB1, CDKN3, LOX, CKS2, CCNA2, PDPN, AURKB, CCNE2, and the extracellular matrix genes FN1 and CTHRC1 (Additional file 2: Table S2B).
The most over-expressed gene in the UPS-A subset (the good prognosis group) was SCARA5, scavenger receptor class A, member 5, a gene that may act as a tumor suppressor in some models [30]. Podoplanin (PDPN) is a mucin-like protein expressed in a variety of cells. Notably, PDPN is a specific marker for lymphatic endothelial cells, and has been shown to be over-expressed in a variety of tumors. ANLN (actin binding protein anillin) binds actin, but is also localized to the nucleus in some cancer cells, and has been reported to be over-expressed in hormone resistant prostate cancer and squamous cell head and neck cancer. Lysyl oxidase (LOX) is a copper containing enzyme that catalyzes the formation of aldehydes from lysine, especially in collagen and elastin [31]. LOX is regulated by hypoxia-inducible factors (HIFs) and has been reported to be upregulated in a number of cancers. In a mouse model, LOX inhibitors decreased metastasis [32] and such inhibitors could eventually be clinically useful [33]. Many of these regulatory proteins could serve as drug targets.
Extracellular matrix genes were also differentially expressed in the two LipoD subsets, with FN1 and CTHRC1 over-expressed in LipoD-B. Differential expression of extracellular matrix proteins in different subsets of malignancies appears to be a common theme. The recognition of a desmoplastic response to cancer has long been recognized [34–36], and tumor-associated fibroblasts may play a role in cancer growth and development [37–42]. The expression of several extracellular matrix and collagen genes has been related to invasion and survival in other tumors [43–45], and studies in breast cancer described two types of stromal responses: a fibromatosis-like stromal gene signature and a CSF-1 macrophage stromal gene signature [46].
Genes over-expressed in UPS-A (the good prognosis group) compared to UPS-B included: SCARA5, TNXB, ALDH1A3, ADH1B, MFAP5, AKAP12, DEFB1, TGFBR3, GAS7, IL17D, IL33, and CD34, as well as the growth factors NEGR1, PDGFD, FGF18, NTRK2, and IGFBP6, a regulator of IGF1. SCARA5 was expressed 34-fold higher in the UPS-A subgroup, and may function as a tumor suppressor gene. Over-expression of SCARA5 suppressed some malignant behaviors in hepatoma cells, and SCARA5 knockdown was associated with activation of MMP9 [ Soft tissue sarcomas Renal cell carcinoma Ovarian cancer Aggressive fibromatosis Hierarchical clustering Probability of develo** metastatic disease Leiomyosarcoma Dedifferentiated liposarcoma Undifferentiated pleomorphic sarcoma. Helias-Rodzewicz Z, Pedeutour F, Coindre JM, Terrier P, Aurias A: Selective elimination of amplified CDK4 sequences correlates with spontaneous adipocytic differentiation in liposarcoma. Genes Chromosomes Cancer. 2009, 48 (11): 943-952. Ladanyi M, Bridge JA: Contribution of molecular genetic data to the classification of sarcomas. Hum Pathol. 2000, 31 (5): 532-538. Mitelman F: Recurrent chromosome aberrations in cancer. Mutat Res. 2000, 462 (2–3): 247-253. Skubitz KM, D’Adamo DR: Sarcoma. Mayo Clin Proc. 2007, 82 (11): 1409-1432. van de Rijn M, Fletcher JA: Genetics of soft tissue tumors. Annu Rev Pathol. 2006, 1: 435-466. Fletcher CD, Gustafson P, Rydholm A, Willen H, Akerman M: Clinicopathologic re-evaluation of 100 malignant fibrous histiocytomas: prognostic relevance of subclassification. J Clin Oncol. 2001, 19 (12): 3045-3050. Nakayama R, Nemoto T, Takahashi H, Ohta T, Kawai A, Seki K, Yoshida T, Toyama Y, Ichikawa H, Hasegawa T: Gene expression analysis of soft tissue sarcomas: characterization and reclassification of malignant fibrous histiocytoma. Mod Pathol. 2007, 20 (7): 749-759. Nielsen TO, West RB, Linn SC, Alter O, Knowling MA, O’Connell JX, Zhu S, Fero M, Sherlock G, Pollack JR, Brown PO, Botstein D, van de Rijn M: Molecular characterisation of soft tissue tumours: a gene expression study. Lancet. 2002, 359 (9314): 1301-1307. Segal NH, Pavlidis P, Antonescu CR, Maki RG, Noble WS, DeSantis D, Woodruff JM, Lewis JJ, Brennan MF, Houghton AN, Cordon-Cardo C: Classification and subtype prediction of adult soft tissue sarcoma by functional genomics. Am J Pathol. 2003, 163 (2): 691-700. Skubitz KM, Skubitz AP: Characterization of sarcomas by means of gene expression. J Lab Clin Med. 2004, 144 (2): 78-91. Chibon F, Lagarde P, Salas S, Perot G, Brouste V, Tirode F, Lucchesi C, de Reynies A, Kauffmann A, Bui B, Terrior P, Bonvalot S, LeCesne A, Vince-Panchere D, Blay JY, Collin F, Guillou L, Leroux A, Coindre JM, Aurias A: Validated prediction of clinical outcome in sarcomas and multiple types of cancer on the basis of a gene expression signature related to genome complexity. Nat Med. 2010, 16 (7): 781-787. Francis P, Namlos HM, Muller C, Eden P, Fernebro J, Berner JM, Bjerkehagen B, Akerman M, Bendahl PO, Isinger A, Rydholm A, Myklebost O, Nilbert M: Diagnostic and prognostic gene expression signatures in 177 soft tissue sarcomas: hypoxia-induced transcription profile signifies metastatic potential. BMC Genomics. 2007, 8: 73- Skubitz KM, Cheng EY, Clohisy DR, Thompson RC, Skubitz AP: Differential gene expression in liposarcoma, lipoma, and adipose tissue. Cancer Invest. 2005, 23 (2): 105-118. Skubitz KM, Francis P, Skubitz AP, Luo X, Nilbert M: Gene expression identifies heterogeneity of metastatic propensity in high-grade soft tissue sarcomas. Cancer. 2012, 118 (17): 4235-4243. Skubitz KM, Pambuccian S, Manivel JC, Skubitz AP: Identification of heterogeneity among soft tissue sarcomas by gene expression profiles from different tumors. J Transl Med. 2008, 6: 23- Skubitz KM, Skubitz AP: Differential gene expression in leiomyosarcoma. Cancer. 2003, 98 (5): 1029-1038. Hoffmann AC, Danenberg KD, Taubert H, Danenberg PV, Wuerl P: A three-gene signature for outcome in soft tissue sarcoma. Clin Cancer Res. 2009, 15 (16): 5191-5198. Lagarde P, Przybyl J, Brulard C, Perot G, Pierron G, Delattre O, Sciot R, Wozniak A, Schoffski P, Terrier P, Neuville A, Coindre JM, Italiano A, Orbach D, Debiec-Rychter M, Chibon F: Chromosome instability accounts for reverse metastatic outcomes of pediatric and adult synovial sarcomas. J Clin Oncol. 2013, 31 (5): 608-615. Skubitz AP, Pambuccian SE, Argenta PA, Skubitz KM: Differential gene expression identifies subgroups of ovarian carcinoma. Transl Res. 2006, 148 (5): 223-248. Skubitz KM, Skubitz AP: Differential gene expression in renal-cell cancer. J Lab Clin Med. 2002, 140 (1): 52-64. Skubitz KM, Skubitz AP: Gene expression in aggressive fibromatosis. J Lab Clin Med. 2004, 143 (2): 89-98. Skubitz KM, Zimmermann W, Kammerer R, Pambuccian S, Skubitz AP: Differential gene expression identifies subgroups of renal cell carcinoma. J Lab Clin Med. 2006, 147 (5): 250-267. Huang J, Zheng DL, Qin FS, Cheng N, Chen H, Wan BB, Wang YP, **ao HS, Han ZG: Genetic and epigenetic silencing of SCARA5 may contribute to human hepatocellular carcinoma by activating FAK signaling. J Clin Invest. 2010, 120 (1): 223-241. Crago AM, Socci ND, DeCarolis P, O’Connor R, Taylor BS, Qin LX, Antonescu CR, Singer S: Copy number losses define subgroups of dedifferentiated liposarcoma with poor prognosis and genomic instability. Clin Cancer Res. 2012, 18 (5): 1334-1340. Lara-Castro C, Fu Y, Chung BH, Garvey WT: Adiponectin and the metabolic syndrome: mechanisms mediating risk for metabolic and cardiovascular disease. Curr Opin Lipidol. 2007, 18 (3): 263-270. Matsuzawa Y, Funahashi T, Kihara S, Shimomura I: Adiponectin and metabolic syndrome. Arterioscler Thromb Vasc Biol. 2004, 24 (1): 29-33. Cunningham BA, Moncur JT, Huntington JT, Kinlaw WB: “Spot 14” protein: a metabolic integrator in normal and neoplastic cells. Thyroid. 1998, 8 (9): 815-825. Kinlaw WB, Quinn JL, Wells WA, Roser-Jones C, Moncur JT: Spot 14: A marker of aggressive breast cancer and a potential therapeutic target. Endocrinology. 2006, 147 (9): 4048-4055. Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M, Jacquemier J, Viens P, Kleer CG, Liu S, Schott A, Hayes D, Birnbaum D, Wicha WS, Dontu G: ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007, 1 (5): 555-567. Burgess DJ, Doles J, Zender L, Xue W, Ma B, McCombie WR, Hannon GJ, Lowe SW, Hemann MT: Topoisomerase levels determine chemotherapy response in vitro and in vivo. Proc Natl Acad Sci U S A. 2008, 105 (26): 9053-9058. Csiszar K: Lysyl oxidases: a novel multifunctional amine oxidase family. Prog Nucleic Acid Res Mol Biol. 2001, 70: 1-32. Erler JT, Giaccia AJ: Lysyl oxidase mediates hypoxic control of metastasis. Cancer Res. 2006, 66 (21): 10238-10241. Rodriguez C, Rodriguez-Sinovas A, Martinez-Gonzalez J: Lysyl oxidase as a potential therapeutic target. Drug News Perspect. 2008, 21 (4): 218-224. Ferrini FS, Rossi MA, Neto MM, Soares FA: Schirrous invasive ductal carcinoma of the breast overexpress p53 oncoprotein. Sao Paulo Med J. 2001, 119 (1): 4-6. Gamgee S: On the removal of scirrhous tumors of the breast. Br Med J. 1877, 844: 255- Underwood JC: A morphometric analysis of human breast carcinoma. Br J Cancer. 1972, 26 (3): 234-237. Chang HY, Sneddon JB, Alizadeh AA, Sood R, West RB, Montgomery K, Chi JT, van de Rijn M, Botstein D, Brown PO: Gene expression signature of fibroblast serum response predicts human cancer progression: similarities between tumors and wounds. PLoS Biol. 2004, 2 (2): E7- Dvorak HF: Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med. 1986, 315 (26): 1650-1659. Elenbaas B, Weinberg RA: Heterotypic signaling between epithelial tumor cells and fibroblasts in carcinoma formation. Exp Cell Res. 2001, 264 (1): 169-184. Iacobuzio-Donahue CA, Argani P, Hempen PM, Jones J, Kern SE: The desmoplastic response to infiltrating breast carcinoma: gene expression at the site of primary invasion and implications for comparisons between tumor types. Cancer Res. 2002, 62 (18): 5351-5357. Olumi AF, Grossfeld GD, Hayward SW, Carroll PR, Tlsty TD, Cunha GR: Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 1999, 59 (19): 5002-5011. West RB, Nuyten DS, Subramanian S, Nielsen TO, Corless CL, Rubin BP, Montgomery K, Zhu S, Patel R, Hernandez-Boussard T, Goldblum JR, Brown PO, van de Vijver M, van de Rijn M: Determination of stromal signatures in breast carcinoma. PLoS Biol. 2005, 3 (6): e187- Baba Y, Iyama K, Ikeda K, Ishikawa S, Hayashi N, Miyanari N, Sado Y, Ninomiya Y, Baba H: The expression of type IV collagen alpha6 chain is related to the prognosis in patients with esophageal squamous cell carcinoma. Ann Surg Oncol. 2008, 15 (2): 555-565. Bauer R, Ratzinger S, Wales L, Bosserhoff A, Senner V, Grifka J, Grassel S: Inhibition of collagen XVI expression reduces glioma cell invasiveness. Cell Physiol Biochem. 2011, 27 (3–4): 217-226. Moh MC, Shen S: The roles of cell adhesion molecules in tumor suppression and cell migration: a new paradox. Cell Adhes Migr. 2009, 3 (4): 334-336. Wu JM, Beck AH, Pate LL, Witten D, Zhu S, Montgomery KD, Allison KH, van de Rijn M, West RB: Endogenous versus tumor-specific host response to breast carcinoma: a study of stromal response in synchronous breast primaries and biopsy site changes. Clin Cancer Res. 2011, 17 (3): 437-446. Vaghjiani RJ, Talma S, Murphy CL: Six-transmembrane epithelial antigen of the prostate (STEAP1 and STEAP2)-differentially expressed by murine and human mesenchymal stem cells. Tissue Eng A. 2009, 15 (8): 2073-2083. Grunewald TG, Ranft A, Esposito I, da Silva-Buttkus P, Aichler M, Baumhoer D, Schaefer KL, Ottaviano L, Poremba C, Jundt G, Jurgens H, Dirksen U, Richter GH, Burdach S: High STEAP1 expression is associated with improved outcome of Ewing’s sarcoma patients. Ann Oncol. 2012, 23 (8): 2185-2190. Grunewald TG, Diebold I, Esposito I, Plehm S, Hauer K, Thiel U, da Silva-Buttkus P, Neff F, Unland R, Muller-Tidow C, Zobywalski C, Lohrig K, Lewandrowski U, Sickmann A, Prazeres da Costa O, Gorlach A, Cossarizza A, Butt E, Richter GH, Burdach S: STEAP1 is associated with the invasive and oxidative stress phenotype of Ewing tumors. Mol Cancer Res. 2012, 10 (1): 52-65. Zhang K, Hu S, Wu J, Chen L, Lu J, Wang X, Liu X, Zhou B, Yen Y: Overexpression of RRM2 decreases thrombspondin-1 and increases VEGF production in human cancer cells in vitro and in vivo: implication of RRM2 in angiogenesis. Mol Cancer. 2009, 8: 11- Supported in part by the Karen Wyckoff Rein in Sarcoma Foundation, NIH P30 CA77598 for utilizing the support of the Biostatistics and Bioinformatics Core of the University of Minnesota Masonic Cancer Center shared resources, and the University of Minnesota Supercomputing Institute. Presented in part at the 18th annual meeting of the Connective Tissue Oncology Society, November 14–17, 2012, Prague, and the 30th meeting of the American Society of Clinical Oncology, Chicago, IL, May 31 - June 4, 2013. The authors declare that they have no competing interests. KMS participated in study design, data analysis, and helped draft the manuscript. APNS participated in study design, data analysis, and helped draft the manuscript. WX participated in data analysis, and helped draft the manuscript. XL participated in data analysis, and helped draft the manuscript. PL participated in data acquisition and analysis, and helped draft the manuscript. JMC participated in data acquisition and analysis, and helped draft the manuscript. FC participated in data acquisition and analysis, and helped draft the manuscript. All authors read and approved the final manuscript. Below are the links to the authors’ original submitted files for images.
This article is published under an open access license.
Please check the 'Copyright Information' section either on this page or in the PDF
for details of this license and what re-use is permitted.
If your intended use exceeds what is permitted by the license or if
you are unable to locate the licence and re-use information,
please contact the Rights and
Permissions team.
Skubitz, K.M., Skubitz, A.P., Xu, W.W. et al. Gene expression identifies heterogeneity of metastatic behavior among high-grade non-translocation associated soft tissue sarcomas.
J Transl Med 12, 176 (2014). https://doi.org/10.1186/1479-5876-12-176 Received: Accepted: Published: DOI: https://doi.org/10.1186/1479-5876-12-176Abbreviations
References
Acknowledgements
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
Authors’ contributions
Electronic supplementary material
Authors’ original submitted files for images
Rights and permissions
About this article
Cite this article
Keywords