Abstract
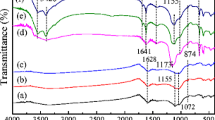
Cadmium (Cd) and lead (Pb) contaminated soils that are used for food production can lead to metal bioaccumulation in the food chain and eventually affect human health. In these agroecosystems, means by which Cd and Pb bioavailability can be reduced are desperately required, with biochar as a proxy for bioavailability reductions. Molecular Cd and Pb sorption mechanisms within short- (0–2 years) or long-term (8–10 years) time periods following biochar application to a contaminated rice paddy soil were investigated. A combination of Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and soft X-ray imaging was utilized to discern potential metal sorption mechanisms. Following both short- and long-term biochar applications, soil Cd and Pb bioavailable fractions shifted partially towards metal (hydr)oxide and carbonate precipitates, and partially towards biochar-organic function group associations; oxygen-containing groups, such as C=O and O–H, appeared to bind Cd and Pb. Soft X-ray imaging results suggested that heavy metals were primarily sorbed on biochar exterior surfaces, yet given time and particle disintegration, metals sorbed onto biochar interior pore walls. Findings suggest that biochar may play a pivotal role in reducing long-term bioavailable Cd and Pb in contaminated soils. Observations also support previous findings that suggest biochar use can lead to reduced heavy metal transfer to plants and potentially to reduced heavy metal consumption by humans.




Similar content being viewed by others
References
Akpomie K, Dawodu F, Adebowale K (2015) Mechanism on the sorption of heavy metals from binary-solution by a low cost montmorillonite and its desorption potential. Alexandria Eng J 54:757–767. https://doi.org/10.1016/j.aej.2015.03.025
Awan S, Ippolito J, Ullman J, Ansari K, Cui L, Siyal A (2021) Biochars reduce irrigation water sodium adsorption ratio. Biochar 3:77–87. https://doi.org/10.1007/s42773-020-00073-z
Chen D, Wang X, Wang X, Feng K, Su J, Dong J (2020) The mechanism of cadmium sorption by sulphur-modified wheat straw biochar and its application cadmium-contaminated soil. Sci Total Environ 714:136550. https://doi.org/10.1016/j.scitotenv.2020.136550
Cui L, Chen T, Yin C, Yan J, Ippolito J, Hussain Q (2019a) Mechanism of adsorption of cadmium and lead ions by iron-activated biochar. BioResources 14:842–857
Cui L, Noerpel M, Scheckel K, Ippolito J (2019b) Wheat straw biochar reduces environmental cadmium bioavailability. Environ Int 126:69–75. https://doi.org/10.1016/j.envint.2019.02.022
Cui L, Li L, Bina R, Yan J, Quan G, Liu Y, Ippolito J, Wang H (2020) Short- and long-term biochar cadmium and lead immobilization mechanisms. Environments 7(7):53. https://doi.org/10.3390/environments7070053
Dong X, Ma L, Zhu Y, Li Y, Gu B (2013) Mechanistic investigation of mercury sorption by Brazilian pepper biochars of different pyrolytic temperatures based on X-ray photoelectron spectroscopy and flow calorimetry. Environ Sci Technol 47:12156–12164. https://doi.org/10.1021/es4017816
Fan J, Zhao G, Sun J, Hu Y, Wang T (2019) Effect of humic acid on Se and Fe transformations in soil during waterlogged incubation. Sci Total Environ 684:476–485. https://doi.org/10.1016/j.scitotenv.2019.05.246
He L, Zhong H, Liu G, Dai Z, Brookes P, Xu J (2019) Remediation of heavy metal contaminated soils by biochar: mechanisms, potential risks and applications in China. Environ Pollut 252:846–855. https://doi.org/10.1016/j.envpol.2019.05.151
Hemavathy R, Kumar P, Kanmani K, Jahnavi N (2020) Adsorptive separation of Cu(II) ions from aqueous medium using thermally/chemically treated Cassia fistula based biochar. J Cleaner Prod 249:119390. https://doi.org/10.1016/j.jclepro.2019.119390
Huang Y, Tang J, Gai L, Gong Y, Guan H, He R, Lyu H (2017) Different approaches for preparing a novel thiol-functionalized graphene oxide/Fe-Mn and its application for aqueous methylmercury removal. Chem Eng J 319:229–239. https://doi.org/10.1016/j.cej.2017.03.015
Ippolito J, Strawn D, Scheckel K, Novak J, Ahmedna M, Niandou M (2012) Macroscopic and molecular investigations of copper sorption by a steam-activated biochar. J Environ Qual 41:1150–1156. https://doi.org/10.2134/jeq2011.0113
Ippolito J, Berry C, Strawn D, Novak J, Levine J, Harley A (2017) Biochars reduce mine land soil bioavailable metals. J Environ Qual 46:411–419. https://doi.org/10.2134/jeq2016.10.0388
Khan Z, Gao M, Qiu W, Islam M, Song Z (2020) Mechanisms for cadmium adsorption by magnetic biochar composites in an aqueous solution. Chemosphere 246:125701. https://doi.org/10.1016/j.chemosphere.2019.125701
Knicker H, Hilscher A, Gonzalezvila F, Almendros G (2008) A new conceptual model for the structural properties of char produced during vegetation fires. Org Geochem 39:935–939. https://doi.org/10.1016/j.orggeochem.2008.03.021
Lehmann J, Rillig M, Thies J, Masiello C, Hockaday W, Crowley D (2011) Biochar effects on soil biota—a review. Soil Biol Biochem 43:1812–1836. https://doi.org/10.1016/j.soilbio.2011.04.022
Li X, Wang C, Zhang J, Liu J, Liu B, Chen G (2020) Preparation and application of magnetic biochar in water treatment: a critical review. Sci Total Environ 711:134847. https://doi.org/10.1016/j.scitotenv.2019.134847
Lian F, **ng B (2017) Black carbon (biochar) in water/soil environments: molecular structure, sorption, stability, and potential risk. Environ Sci Technol 51:13517–13532. https://doi.org/10.1021/acs.est.7b02528
Lin D, Tian X, Li T, Zhang Z, He X, **ng B (2012) Surface-bound humic acid increased Pb2+ sorption on carbon nanotubes. Environ Pollut 167:138–147. https://doi.org/10.1016/j.envpol.2012.03.044
Lu R (2000) Methods of inorganic pollutants analysis. Soil and agro-chemical analysis methods. Agricultural Science and Technology Press, Bei**g, pp 205–266
Mujtaba Munir M, Liu G, Yousaf B, Ali M, Cheema A, Rashid M, Rehman A (2020) Bamboo-biochar and hydrothermally treated-coal mediated geochemical speciation, transformation and uptake of Cd, Cr, and Pb in a polymetal(iod)s-contaminated mine soil. Environ Pollut 265:114816. https://doi.org/10.1016/j.envpol.2020.114816
Nguyen T, Cho H, Poster D, Ball W (2007) Evidence for a pore-filling mechanism in the adsorption of aromatic hydrocarbons to a natural wood char. Environ Sci Technol 41:1212–1217. https://doi.org/10.1021/es0617845
Novak J, Ippolito J, Watts D, Sigua G, Ducey T, Johnson M (2019) Biochar compost blends facilitate switchgrass growth in mine soils by reducing Cd and Zn bioavailability. Biochar 1:97–114. https://doi.org/10.1007/s42773-019-00004-7
Quan G, Fan Q, Sun J, Cui L, Wang H, Gao B, Yan J (2020) Characteristics of organo-mineral complexes in contaminated soils with long-term biochar application. J Hazard Mater 384:121265. https://doi.org/10.1016/j.jhazmat.2019.121265
Soares da Silva L, Constantino I, Bento L, Tadini A, Bisinoti M, Boscolo M, Ferreira O, Mounier S, Piccolo A, Spaccini R, Cornélio M, Moreira A (2020) Humic extracts from hydrochar and Amazonian anthrosol: molecular features and metal binding properties using EEM-PARAFAC and 2D FTIR correlation analyses. Chemosphere 256:127110. https://doi.org/10.1016/j.chemosphere.2020.127110
Spokas K, Novak J, Masiello C, Johnson M, Colosky E, Ippolito J, Trigo C (2014) Physical disintegration of biochar: an overlooked process. Environ Sci Technol Lett 1:326–332. https://doi.org/10.1021/ez500199t
Tan L, Sun C, Wang Y, Wang T, Wu G, He H, Zheng J (2020) Changes in biochar properties in typical loess soil under a 5-year field experiment. J Soils Sediments 20:340–351. https://doi.org/10.1007/s11368-019-02398-0
**e S, Wang L, Xu Y, Lin D, Sun Y, Zheng S (2020) Performance and mechanisms of immobilization remediation for Cd contaminated water and soil by hydroxy ferric combined acid-base modified sepiolite (HyFe/ABsep). Sci Total Environ 740:140009. https://doi.org/10.1016/j.scitotenv.2020.140009
Yang F, Zhao L, Gao B, Xu X, Cao X (2016) The interfacial behavior between biochar and soil minerals and its effect on biochar stability. Environ Sci Technol 50(5):2264–2271. https://doi.org/10.1021/acs.est.5b03656
Zhao L, Nan H, Kan Y, Xu X, Qiu H, Cao X (2019) Infiltration behavior of heavy metals in runoff through soil amended with biochar as bulking agent. Environ Pollut 254:113114. https://doi.org/10.1016/j.envpol.2019.113114
Zhao M, Dai Y, Zhang M, Feng C, Qin B, Zhang W, Zhao N, Li Y, Ni Z, Xu Z, Tsang D, Qiu R (2020) Mechanisms of Pb and/or Zn adsorption by different biochars: biochar characteristics, stability, and binding energies. Sci Total Environ 717:136894. https://doi.org/10.1016/j.scitotenv.2020.136894
Zhong Y, Igalavithana A, Zhang M, Li X, Rinklebe J, Hou D, Tack F, Alessi D, Tsang D, Ok Y (2020) Effects of aging and weathering on immobilization of trace metals/metalloids in soils amended with biochar. Environ Sci Processes Impacts 22(9):1790–1808. https://doi.org/10.1039/d0em00057d
Acknowledgements
This study was partially supported by, the National Natural Science Foundation of China under a grant number of 41501339, 21677119, Jiangsu Province Science Foundation for Youths under a grant number of BK20140468 and sponsored by the QingLan Project.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MPG 4221 KB)
Supplementary file2 (MPG 5082 KB)
Supplementary file3 (MPG 9244 KB)
Rights and permissions
About this article
Cite this article
Cui, L., Li, L., Bian, R. et al. Physicochemical disintegration of biochar: a potentially important process for long-term cadmium and lead sorption. Biochar 3, 511–518 (2021). https://doi.org/10.1007/s42773-021-00108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42773-021-00108-z