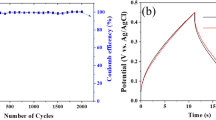
Abstract
Development of cheap, abundant and noble-metal-free materials as high efficient oxygen reduction electrocatalysts is crucial for future energy storage system. Here, one-dimensional (1D) MnO N-doped carbon nanofibers (MnO-NCNFs) were successfully developed by electrospinning combined with high temperature pyrolysis. The MnO-NCNFs exhibit promising electrochemical performance, methanol tolerance, and durability in alkaline medium. The outstanding electrocatalytic activity is mainly attributed to several issues. First of all, the uniform 1D fiber structure and the conductive network could facilitate the electron transport. Besides, the introduction of Mn into the precursor can catalyze the transformation of amorphous carbon to graphite carbon, while the improved graphitization means better conductivity, beneficial for the enhancement of catalytic activity for oxygen reduction reaction (ORR). Furthermore, the porous structure and high surface area can effectively decrease the mass transport resistance and increase the exposed ORR active sites, thus improve utilization efficiency and raise the quantity of exposed ORR active sites. The synergistic effect of MnO and NCNFs matrix, which enhances charge transfer, adsorbent transport, and delivers efficiency in the electrolyte solution, ensures the high ORR performance of MnO-NCNFs.
摘要
廉价、 储量丰富的非贵金属材料作为高效氧还原电催化剂是未来能量存储系统实际应用的关键. 基于此, 我们通过静电纺丝法并结合相应热处理制备了一维氮掺杂碳纳米纤维包覆一氧化锰(MnO-NCNFs). 该材料在碱性体系中表现出良好的电化学性能, 耐甲醇腐蚀性和稳定性. 其优异的电催化活性主要归结于以下几点: (1) 均一的一维纤维结构和导电网络能够促进反应过程中快速的电子转移; (2) 过渡金属锰的引入能够促进无定型碳向石墨碳的转变, 同时石墨化程度的提高意味着更好的导电性, 有利于氧还原活性的提高; (3) 多孔结构和高比表面积能够有效降低传质阻力并提高暴露的氧还原活性位点数量, 进而提高物质利用效率.
Similar content being viewed by others
References
Aijaz A, Masa J, Rösler C, et al. Co@Co3O4 encapsulated in carbon nanotube-grafted nitrogen-doped carbon polyhedra as an advanced bifunctional oxygen electrode. Angew Chem Int Ed, 2016, 55: 4087–4091
Liu R, Zhang H, Liu S, et al. Shrimp-shell derived carbon nanodots as carbon and nitrogen sources to fabricate three-dimensional N-doped porous carbon electrocatalysts for the oxygen reduction reaction. Phys Chem Chem Phys, 2016, 18: 4095–4101
Wu G, More KL, Johnston CM, et al. High-performance electro-catalysts for oxygen reduction derived from polyaniline, iron, and cobalt. Science, 2011, 332: 443–447
Koh K, Meng Y, Huang X, et al. N- and O-doped mesoporous carbons derived from rice grains: efficient metal-free electro-catalysts for hydrazine oxidation. Chem Commun, 2016, 52: 13588–13591
Meng Y, Voiry D, Goswami A, et al. N-, O-, and S-tridoped nanoporous carbons as selective catalysts for oxygen reduction and alcohol oxidation reactions. J Am Chem Soc, 2014, 136: 13554–13557
Chen Z, Zhao H, Zhang J, et al. IrNi nanoparticle-decorated flower-shaped NiCo2O4 nanostructures: controllable synthesis and enhanced electrochemical activity for oxygen evolution reaction. Sci China Mater, 2016, 60: 119–130
Lu S, Zhuang Z. Electrocatalysts for hydrogen oxidation and evolution reactions. Sci China Mater, 2016, 59: 217–238
Bu L, Zhang N, Guo S, et al. Biaxially strained PtPb/Pt core/shell nanoplate boosts oxygen reduction catalysis. Science, 2016, 354: 1410–1414
Shen K, Chen X, Chen J, et al. Development of MOF-derived carbon-based nanomaterials for efficient catalysis. ACS Catal, 2016, 6: 5887–5903
Huang H, Du C, Wu S, et al. Thermolytical entrapment of ultrasmall MoC nanoparticles into 3D frameworks of nitrogen-rich graphene for efficient oxygen reduction. J Phys Chem C, 2016, 120: 15707–15713
Meng Y, Zou X, Huang X, et al. Polypyrrole-derived nitrogen and oxygen Co-doped mesoporous carbons as efficient metal-free electrocatalyst for hydrazine oxidation. Adv Mater, 2014, 26: 6510–6516
Fu QQ, Li HH, Ma SY, et al. A mixed-solvent route to unique PtAuCu ternary nanotubes templated from Cu nanowires as efficient dual electrocatalysts. Sci China Mater, 2016, 59: 112–121
Yin P, Yao T, Wu Y, et al. Single cobalt atoms with precise Ncoordination as superior oxygen reduction reaction catalysts. Angew Chem Int Ed, 2016, 55: 10800–10805
**a BY, Yan Y, Li N, et al. A metal-organic framework-derived bifunctional oxygen electrocatalyst. Nat Energy, 2016, 1: 15006
Wei J, Liang Y, Hu Y, et al. Hydrothermal synthesis of metal-polyphenol coordination crystals and their derived metal/N-doped carbon composites for oxygen electrocatalysis. Angew Chem Int Ed, 2016, 55: 12470–12474
Zhou T, Du Y, Yin S, et al. Nitrogen-doped cobalt phosphate@-nanocarbon hybrids for efficient electrocatalytic oxygen reduction. Energy Environ Sci, 2016, 9: 2563–2570
Wang MQ, Ye C, Bao SJ, et al. Platanus hispanica-inspired design of Co-carbon nanotube frameworks through chemical vapor deposition: a highly integrated hierarchical electrocatalyst for oxygen reduction reactions. Chem Commun, 2016, 52: 12992–12995
Wu ZY, Xu XX, Hu BC, et al. Iron carbide nanoparticles encapsulated in mesoporous Fe-N-doped carbon nanofibers for efficient electrocatalysis. Angew Chem Int Ed, 2015, 54: 8179–8183
Singh KP, Bae EJ, Yu JS. Fe-P: a new class of electroactive catalyst for oxygen reduction reaction. J Am Chem Soc, 2015, 137: 3165–3168
Li W, Yu A, Higgins DC, et al. Biologically inspired highly durable iron phthalocyanine catalysts for oxygen reduction reaction in polymer electrolyte membrane fuel cells. J Am Chem Soc, 2010, 132: 17056–17058
Wang S, Cui Z, Cao M. A template-free method for preparation of cobalt nanoparticles embedded in N-doped carbon nanofibers with a hierarchical pore structure for oxygen reduction. Chem Eur J, 2015, 21: 2165–2172
Tao L, Wang Q, Dou S, et al. Edge-rich and dopant-free graphene as a highly efficient metal-free electrocatalyst for the oxygen reduction reaction. Chem Commun, 2016, 52: 2764–2767
Li PC, Hu CC, You TH, et al. Development and characterization of bi-functional air electrodes for rechargeable zinc-air batteries: effects of carbons. Carbon, 2017, 111: 813–821
Delmondo L, Salvador GP, Muñoz-Tabares JA, et al. Nanostructured MnxOy for oxygen reduction reaction (ORR) catalysts. Appl Surf Sci, 2016, 388: 631–639
Sumboja A, Ge X, Zheng G, et al. Durable rechargeable zinc-air batteries with neutral electrolyte and manganese oxide catalyst. J Power Sources, 2016, 332: 330–336
Zhang F, Shi Y, Xue T, et al. In situ electrochemically converting Fe2O3-Ni(OH)2 to NiFe2O4-NiOOH: a highly efficient electrocatalyst towards water oxidation. Sci China Mater, 2017, 60: 324–334
Calegaro ML, Lima FHB, Ticianelli EA. Oxygen reduction reaction on nanosized manganese oxide particles dispersed on carbon in alkaline solutions. J Power Sources, 2006, 158: 735–739
Mao L. Mechanistic study of the reduction of oxygen in air electrode with manganese oxides as electrocatalysts. ElectroChim Acta, 2003, 48: 1015–1021
Cheng H, Xu K, **ng L, et al. Manganous oxide nanoparticles encapsulated in few-layer carbon as an efficient electrocatalyst for oxygen reduction in alkaline media. J Mater Chem A, 2016, 4: 11775–11781
Yuan H, He Z. Graphene-modified electrodes for enhancing the performance of microbial fuel cells. Nanoscale, 2015, 7: 7022–7029
Chen G, Sunarso J, Zhu Y, et al. Highly active carbon/α-MnO2 hybrid oxygen reduction reaction electrocatalysts. ChemElectroChem, 2016, 3: 1760–1767
Cao S, Han N, Han J, et al. Mesoporous hybrid shells of carbonized polyaniline/Mn2O3 as non-precious efficient oxygen reduction reaction catalyst. ACS Appl Mater Interfaces, 2016, 8: 6040–6050
Aricò AS, Bruce P, Scrosati B, et al. Nanostructured materials for advanced energy conversion and storage devices. Nat Mater, 2005, 4: 366–377
Tang Q, Jiang L, Liu J, et al. Effect of surface manganese valence of manganese oxides on the activity of the oxygen reduction reaction in alkaline media. ACS Catal, 2014, 4: 457–463
Chen R, Yan J, Liu Y, et al. Three-dimensional nitrogen-doped graphene/MnO nanoparticle hybrids as a high-performance catalyst for oxygen reduction reaction. J Phys Chem C, 2015, 119: 8032–8037
Dou S, Tao L, Huo J, et al. Etched and doped Co9S8/ graphene hybrid for oxygen electrocatalysis. Energy Environ Sci, 2016, 9: 1320–1326
Zhao X, Du Y, ** L, et al. Membranes of MnO beading in carbon nanofibers as flexible anodes for high-performance lithium-ion batteries. Sci Rep, 2015, 5: 14146
Liu B, Hu X, Xu H, et al. Encapsulation of MnO nanocrystals in electrospun carbon nanofibers as high-performance anode materials for lithium-ion batteries. Sci Rep, 2014, 4: 4229
Yuan C, Li J, Hou L, et al. Ultrathin Mesoporous NiCo2O4 nanosheets supported on Ni foam as advanced electrodes for supercapacitors. Adv Funct Mater, 2012, 22: 4592–4597
Wu J, Park HW, Yu A, et al. Facile synthesis and evaluation of nanofibrous iron-carbon based non-precious oxygen reduction reaction catalysts for Li-O2 battery applications. J Phys Chem C, 2012, 116: 9427–9432
Wu TX, Wang GZ, Zhang X, et al. Transforming chitosan into Ndoped graphitic carbon electrocatalysts. Chem Commun, 2015, 51: 1334–1337
**e S, Huang S, Wei W, et al. Chitosan waste-derived Co and N Co-doped carbon electrocatalyst for efficient oxygen reduction reaction. ChemElectroChem, 2015, 2: 1806–1812
Lin L, Zhu Q, Xu AW. Noble-metal-free Fe-N/C catalyst for highly efficient oxygen reduction reaction under both alkaline and acidic conditions. J Am Chem Soc, 2014, 136: 11027–11033
Yin J, Qiu Y, Yu J. Onion-like graphitic nanoshell structured Fe-N/C nanofibers derived from electrospinning for oxygen reduction reaction in acid media. Electrochem Commun, 2013, 30: 1–4
Zamani P, Higgins D, Hassan F, et al. Electrospun iron-polyaniline-polyacrylonitrile derived nanofibers as non-precious oxygen reduction reaction catalysts for PEM fuel cells. Electrochim Acta, 2014, 139: 111–116
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21671096 and 21603094), the Natural Science Foundation of Guangdong Province (2016A030310376), Shenzhen Key Laboratory Project (ZDSYS201603311013489), the Natural Science Foundation of Shenzhen (JCYJ20150630145302231 and JCYJ20150331101823677), and the Undergraduate Training Program for Innovation and Entrepreneurship of Guangdong (2016S10).
Author information
Authors and Affiliations
Corresponding author
Additional information
Chaoqun Shang received his bachelor’s degree from Qingdao University of Science and Technology in 2009, master’s degree from Qingdao University of Science and Technology in 2012, and PhD degree from Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences in 2015. He is currently a Postdoc in the Department of Materials Science & Engineering, Southern University of Science and Technology, Shenzhen, China. His current research interests mainly focus on designing high-performance electrode materials for energy storage systems.
Mingyang Yang received his bachelor’s degree from Henan Normal University in 2012, master’s degree from the Central South University in 2015. He is currently a PhD candidate in the Department of Materials Science & Engineering, Southern University of Science and Technology, Shenzhen, China. His current research interests mainly focus on water splitting and lithium sulfur batteries.
Zhouguang Lu is now an associate professor in the Department of Materials Science and Engineering, South University of Science and Technology of China. He obtained his bachelor’s from the Central South University in 2001 and got his master’s degree under the joint master program between Tsinghua University and Central South University in 2004, and PhD from City University of Hong Kong in 2009. He is the recipient of Fulbright Scholarship of USA Government in 2008–2009 and the Overseas High-Caliber Personnel (Level B) of Shenzhen Government in 2013. His research mainly covers the design and synthesis of nanostructures and their application in energy storage and conversion with focus on lithium/sodium ion batteries, and lithium-air batteries. He has authored for more than 100 peer-review papers with total citations of more than 2600 and H-index of 30.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Shang, C., Yang, M., Wang, Z. et al. Encapsulated MnO in N-do** carbon nanofibers as efficient ORR electrocatalysts. Sci. China Mater. 60, 937–946 (2017). https://doi.org/10.1007/s40843-017-9103-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-017-9103-1