Abstract
Introduction
The ability of B cells to negatively regulate cellular immune responses and inflammation has been described. The regulatory B (Breg) cells with the unique CD1dhiCD5+CD19+ phenotype and the capacity to produce IL-10 are potent negative regulators of inflammation and autoimmunity in several in vivo mouse models of autoimmune disease.
Aim
To investigate whether Breg cell deficiency participates in autoimmune thyroiditis (AIT) in an animal model.
Materials and methods
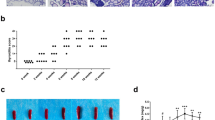
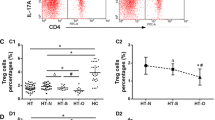
Non-obese diabetic (NOD).H-2h4 mice at 4 weeks of age were randomly divided into control and iodine-treated groups; the iodine-treated group received sterile water containing 0.005 % NaI for 10 or 20 weeks. The percentage of CD1dhiCD5+CD19+ Bregs, CD4+CD25+FoxP3+ regulatory T cells (Treg) and CD4+IL17+ T helper 17 cells (Th17) in splenic mononuclear cells was detected by multicolor flow cytometry. The expression of IL-10 mRNA and TGF-β mRNA in splenocytes was measured by real-time RT-PCR.
Results
NOD.H-2h4 mice spontaneously develop anti-thyroglobulin autoantibodies and intrathyroidal lymphocyte infiltration when supplied with iodine in drinking water. Mice with AIT had a decreased CD1dhiCD5+CD19+ Breg subset and reduced IL-10 mRNA expression in splenocytes compared with controls (p < 0.05) and maintained relatively low levels during the development of thyroiditis. The proportion of Breg cells was negatively correlated with the proportion of Th17 cells, but positively correlated with CD4+CD25+FoxP3+ Treg cells in splenocytes (All p < 0.05).
Conclusions
The defective expression of Breg cells combined with impaired Treg cells and enhanced Th17 cells might play an important role in the development of iodine-induced AIT in NOD.H-2h4 mice.






Similar content being viewed by others
Abbreviations
- AIT:
-
Autoimmune thyroiditis
- NOD.H-2h4 :
-
Non-obese diabetic H-2h4
- Treg:
-
Regulatory T cells
- Breg:
-
Regulatory B cells
- SAT:
-
Spontaneously developed autoimmune thyroiditis
- Th1:
-
T helper type 1
- Th2:
-
T helper type 2
- EAE:
-
Experimental autoimmune encephalitis
- RA:
-
Rheumatoid arthritis
- CIA:
-
Collagen-induced arthritis
- TgAb:
-
Anti-thyroglobulin antibody
- MTg:
-
Mouse thyroglobulin
References
Braley-Mullen H, Sharp GC, Medling B, Tang H (1999) Spontaneous autoimmune thyroiditis in NOD.H-2h4 mice. J Autoimmun 12:157–165
Hutchings PR, Verma S, Phillips JM, Harach SZ, Howlett S, Cooke A (1999) Both CD4 and CD8T cells are required for iodine accelerated thyroiditis in NOD mice. Cell Immunol 192:113–121
Braley-Mullen H, Yu S (2000) Early requirement for B cells for development of spontaneous autoimmune thyroiditis in NOD.H-2h4 mice. J Immunol 165:7262–7269
Yu S, Dunn R, Kehry MR, Braley-Mullen H (2008) B cell depletion inhibits spontaneous autoimmune thyroiditis in NOD.H-2h4 mice. J Immunol 180:7706–7713
Park H, Li Z, Yang XO et al (2005) A distinct lineage of CD4 T cells regulates tissue inflammation by producing intereleukin-17. Nat Immunol 6:1133–1141
Afzali B, Lombardi G, Lechler RI, Lord GM (2007) The role of T helper 17 (Th17) and regulatory T cells (Treg) in human organ transplantation and autoimmune disease. Clin Exp Immunol 148:32–46
Komiyama Y, Nakae S, Matsuki T et al (2006) IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 177:566–573
Le Bien TW, Tedder TF (2008) B-lymphocytes: how they develop and function. Blood 112:1570–1579
Bouaziz JD, Yanaba K, Tedder TF (2008) Regulatory B cells as inhibitors of immune responses and inflammation. Immunol Rev 224:201–214
DiLillo DJ, Matsushita T, Tedder TF (2010) B10 cells and regulatory B cells balance immune responses during inflammation, autoimmunity, and cancer. Ann N Y Acad Sci 1183:38–57
Mizoguchi A, Mizoguchi E, Takedatsu H, Blumberg RS, Bhan AK (2002) Chronic intestinal inflammatory condition generates IL-10-producing regulatory B cell subset characterized by CD1d upregulation. Immunity 16:219–230
Mauri C, Gray D, Mushtaq N, Londei M (2003) Prevention of arthritis by interleukin 10-producing B cells. J Exp Med 197:489–501
Hussain S, Delovitch TL (2007) Intravenous transfusion of BCR-activated B cells protects NOD mice from type 1 diabetes in an IL-10-dependent manner. J Immunol 179:7225–7232
Matsushita T, Yanaba K, Bouaziz JD, Fujimoto M, Tedder TF (2008) Regulatory B cells inhibit EAE initiation in mice while other B cells promote disease progression. J Clin Investig 118:3420–3430
Allen EM, Appel MC, Braverman LE (1986) The effects of iodide ingestion on the development of spontaneous lymphocytic thyroiditis in the diabetes-prone BB/W rats. Endocrinol 118:1977–1981
Xue H, Wang W, Shan Z et al (2011) Dynamic changes of CD4+ CD25+ regulatory T cells in NOD.H-2h4 mice with iodine-induced autoimmune thyroiditis. Biol Trace Elem Res 143:292–301
Lemoine S, Morva A, Youinou P, Jamin C (2009) Regulatory B cells in autoimmune diseases: how do they work? Ann N Y Acad Sci 1173:260–267
Tian J, Zekzer D, Hanssen L, Lu Y, Olcott A, Kaufman DL (2001) Lipopolysaccharide-activated B cells downregulate Th1 immunity and prevent autoimmune diabetes in nonobese diabetic mice. J Immunol 167:1081–1089
Rieger A, Bar-Or A (2008) B-cell-derived interleukin-10 in autoimmune disease: regulating the regulators. Nat Rev Immunol 8:486–487
Dal Canto MC, Melvold RW, Kim BS, Miller SD (1995) Two models of multiple sclerosis: experimental allergic encephalomyelitis (EAE) and Theiler’s murine encephalomyelitis virus (TMEV) infection. A pathological and immunological comparison. Microsc Res Tech 32:215–229
Kong YC, Morris GP, Brown NK, Yan Y, Flynn JC, David CS (2009) Autoimmune thyroiditis: a model uniquely suited to probe regulatory T cell function. J Autoimmun 33:239–246
Horie I, Abiru N, Nagayama Y et al (2009) T helper type 17 immune response plays an indispensable role for development of iodine-induced autoimmune thyroiditis in nonobese diabetic-H2h4 mice. Endocrinology 150:5135–5142
Wei B, Velazquez P, Turovskaya O et al (2005) Proc Natl Acad Sci USA 102:2010–2015
Mann MK, Maresz K, Shriver LP, Tan Y, Dittel BN (2007) B cell regulation of CD4+ CD25+ T regulatory cells and IL-10 via B7 is essential for recovery from experimental autoimmune encephalomyelitis. J Immunol 178:3447–3456
Reichardt P, Dornbach B, Rong S et al (2007) Naive B-cells generate regulatory T-cells in the presence of a mature immunological synapse. Blood 5:1519–1529
Acknowledgments
We thank Ms. Hongmei Zhang and Ms. Hong Wang for their excellent technical assistance. This project was supported by a research fund from the National Nature Science Foundation of China (81170731).
Conflict of interest
The authors Lei Shi, Mei Bi, Rong Yang, ** Zhou, Shujie Zhao, Chenling Fan, Zhongyan Shan, Yushu Li, and Wei** Teng declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Y. Li and W. Teng have contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Shi, L., Bi, M., Yang, R. et al. Defective expression of regulatory B cells in iodine-induced autoimmune thyroiditis in non-obese diabetic H-2h4 mice. J Endocrinol Invest 37, 43–50 (2014). https://doi.org/10.1007/s40618-013-0013-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-013-0013-1