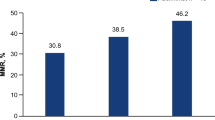
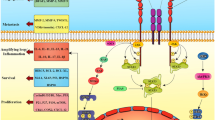
Abstract
Dysregulation of Bruton's tyrosine kinase (BTK) signalling has been linked to various B cell malignancies and autoimmune diseases. Orelabrutinib (宜诺凯®) is an orally administered, potent, irreversible and highly selective BTK-inhibitor being developed by InnoCare Pharma for the treatment of B cell malignancies and autoimmune diseases. In December 2020, orelabrutinib received its first approval in China for the treatment of patients with mantle cell lymphoma (MCL) or chronic lymphocytic leukaemia (CLL)/small lymphocytic lymphoma (SLL), who have received at least one treatment in the past. Clinical development of orelabrutinib for various indications is underway in the USA and China. This article summarizes the milestones in the development of orelabrutinib leading to this first approval.
Similar content being viewed by others
References
Vargas L, Hamasy A, Nore BF, et al. Inhibitors of BTK and ITK: state of the new drugs for cancer, autoimmunity and inflammatory diseases. Scand J Immunol. 2013;78(2):130–9.
Lorenzo-Vizcaya A, Fasano S, Isenberg DA. Bruton’s tyrosine kinase inhibitors: a new therapeutic target for the treatment of SLE? Immunotargets Ther. 2020;9:105–10.
Pal Singh S, Dammeijer F, Hendriks RW. Role of Bruton’s tyrosine kinase in B cells and malignancies. Mol Cancer. 2018;17(1):57.
Zarrin AA, Bao K, Lupardus P, et al. Kinase inhibition in autoimmunity and inflammation. Nat Rev Drug Discov. 2021;20(1):39–63.
Shaw ML. Second-generation BTK inhibitors hit the treatment bullseye with fewer off-target effects. Am J Manag Care. 2020;26(7 Spec No.):SP226–7.
NMPA China. Orelabrutinib tablets. 2021. http://app1.nmpa.gov.cn/data_nmpa/face3/dir.html. Accessed 27 Jan 2021.
InnoCare Pharma Ltd. Orelabrutinib: Chinese prescribing information. Bei**g: Innocare Pharma Ltd.; 2021.
Zhang B, Zhao R, Liang R, et al. Orelabrutinib, a potent and selective Bruton's tyrosine kinase inhibitor with superior safety profile and excellent PK/PD properties [abstract no. CT132]. In: Proceedings American Association for Cancer Research Annual Meeting. 2020.
Song Y, Liu L, Zhang M, et al. Safety and efficacy of orelabrutinib monotherapy in Chinese patients with relapsed or refractory mantle cell lymphoma: a multicenter, open-label, phase II study [abstract no. 755]. Blood 2019;134(Suppl. 1).
Song Y, Song Y, Liu L, et al. Long-term safety and efficacy of orelabrutinib monotherapy in Chinese patients with relapsed or refractory mantle cell lymphoma: a multicenter, open-label, phase II study [abstract]. Blood. 2020;136(Suppl. 1):1.
Xu W, Song Y, Li Z, et al. Safety, tolerability and efficacy of orelabrutinib, once a day, to treat Chinese patients with relapsed or refractory chronic lymphocytic leukemia/small cell leukemia [abstract]. Blood. 2019;134(Suppl. 1):4319.
Xu W, Song Y, Wang T, et al. Updated results from the phase II study of orelabrutinib monotherapy in Chinese patients with relapsed or refractory chronic lymphocytic leukemia/ small cell leukemia [abstract]. Blood. 2020;136(Suppl. 1):26–7.
Song Y, Xu W, Liu L, et al. Pooled analysis of safety data from clinical trials of orelabrutinib monotherapy in hematologic malignancies [abstract]. Blood. 2020;136(Suppl. 1):43.
Funding
The preparation of this review was not supported by any external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Authorship and Conflict of interest
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Sohita Dhillon is a contracted employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Ethics approval, consent to participate, consent to publish, availability of data and material, code availability
Not applicable.
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Rights and permissions
About this article
Cite this article
Dhillon, S. Orelabrutinib: First Approval. Drugs 81, 503–507 (2021). https://doi.org/10.1007/s40265-021-01482-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-021-01482-5