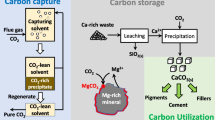
Abstract
Aqueous mineral carbonation is the most selected ‘ex-situ’ mineral carbonation route under research scrutiny and is among the earliest routes studied on a pilot scale. This review summarises the recent advancements in aqueous mineral carbonation and discusses the current frontiers, research gaps, and future perspectives of the process. It also provides useful insights into the current practices and conveys the required actions to overcome the present challenges in the field. The key factors hindering the successful deployment of the technology on a large scale are the high cost of operation, energy intensity, slow reaction, and low carbon dioxide fixation efficiency ratio. Once the current challenges are circumvented, this aqueous route can stand as a potentially viable carbon sequestration technology for application in small-to-medium scale carbon dioxide emitters. The exponential increase in the number of studies and noticeable pilot-scale initiatives in recent years indicate that the aqueous mineral carbonation research is progressing in the right direction towards develo** the technology as a promising, economically viable, and sustainable industrial carbon dioxide sequestration method.

Similar content being viewed by others
References
Arce FGLA, Okamoto S, Dos Santos JC et al (2018) CO2 sequestration by pH-swing mineral carbonation based on HCl/NH4OH system using iron-rich lizardite 1T. J CO2 Util 24:164–173. https://doi.org/10.1016/j.jcou.2018.01.001
Azdarpour A, Asadullah M, Mohammadian E et al (2015) A review on carbon dioxide mineral carbonation through pH-swing process. Chem Eng J 279:615–773. https://doi.org/10.1016/j.cej.2015.05.064
Baciocchi R, Costa G, Di Gianfilippo M et al (2015) Thin-film versus slurry-phase carbonation of steel slag: CO2 uptake and effects on mineralogy. J Hazard Mater 283:302–313. https://doi.org/10.1016/j.jhazmat.2014.09.016
Bao W, Li H, Yi Z (2010) Selective leaching of steelmaking slag for indirect CO2 mineral sequestration. Ind Eng Chem Res 49:2055–2063. https://doi.org/10.1021/ie801850s
Benhelal E, Rashid MI, Rayson MS et al (2018) Study on mineral carbonation of heat activated lizardite at pilot and laboratory scale. J CO2 Util 26:230–238. https://doi.org/10.1016/j.jcou.2018.05.015
Benhelal E, Rashid MI, Rayson MS et al (2019) Direct aqueous carbonation of heat activated serpentine: discovery of undesirable side reactions reducing process efficiency. Appl Energy 2425:1369–1382. https://doi.org/10.1016/j.apenergy.2019.03.170
Bobicki ER, Liu Q, Xu Z, Zeng H (2012) Carbon capture and storage using alkaline industrial wastes. Prog Energy Combust Sci 38:302–320. https://doi.org/10.1016/j.mineng.2014.08.009
Bobicki ER, Liu Q, Xu Z (2015) Mineral carbon storage in pre-treated ultramafic ores. Miner Eng 70:43–54. https://doi.org/10.1016/j.mineng.2014.08.009
Bodénan F, Bourgeois F, Petiot C et al (2014) Ex situ mineral carbonation for CO2 mitigation: evaluation of mining waste resources, aqueous carbonation processability and life cycle assessment (Carmex project). Miner Eng 59:52–63. https://doi.org/10.1016/j.mineng.2014.01.011
Bodor M, Santos RM, Van Gerven T, Vlad M (2013) Recent developments and perspectives on the treatment of industrial wastes by mineral carbonation - A review. Cent Eur J Eng 3(4):566–584. https://doi.org/10.2478/s13531-013-0115-8
Boot-Handford ME, Abanades JC, Anthony EJ et al (2014) Carbon capture and storage update. Energy Environ Sci 7:130–189. https://doi.org/10.1039/C3EE42350F
Chang EE, Pan SY, Chen YH et al (2012) Accelerated carbonation of steelmaking slags in a high-gravity rotating packed bed. J Hazard Mater 227:97–106. https://doi.org/10.1016/j.jhazmat.2012.05.021
Chang EE, Chen TL, Pan SY et al (2013a) Kinetic modeling on CO2 capture using basic oxygen furnace slag coupled with cold-rolling wastewater in a rotating packed bed. J Hazard Mater 260:937–946. https://doi.org/10.1016/j.jhazmat.2013.06.052
Chang EE, Chiu AC, Pan SY et al (2013b) Carbonation of basic oxygen furnace slag with metalworking wastewater in a slurry reactor. Int J Greenh Gas Control 12:382–389. https://doi.org/10.1016/j.ijggc.2012.11.026
Chen ZY, O’Connor WK, Gerdemann SJ (2006) Chemistry of aqueous mineral carbonation for carbon sequestration and explanation of experimental results. Environ Prog 25:161–166. https://doi.org/10.1002/ep.10127
Chen Z, Song HS, Portillo M et al (2009) Long-term calcination/carbonation cycling and thermal pretreatment for CO2 capture by limestone and dolomite. Energy Fuels 23(3):1437–1444. https://doi.org/10.1021/ef800779k
Chiang P-C, Pan S-Y (2017a) Carbon dioxide mineralization and utilization. Springer, Singapore
Chiang P-C, Pan S-Y (2017b) System optimization. In: Carbon dioxide mineralization and utilization. Springer, Singapore, pp 403–439. https://doi.org/10.1007/978-981-10-3268-4_19
Dean CC, Blamey J, Florin NH et al (2011) The calcium loo** cycle for CO2 capture from power generation, cement manufacture and hydrogen production. Chem Eng Res Des 89(6):836–855. https://doi.org/10.1016/j.cherd.2010.10.013
Dindi A, Quang DV, Vega LF et al (2019) Applications of fly ash for CO2 capture, utilization, and storage. J CO2 Util 29:82–102. https://doi.org/10.1016/j.jcou.2018.11.011
Ding W, Yang H, Ouyang J, Long H (2016) Modified wollastonite sequestrating CO2 and exploratory application of the carbonation products. RSC Adv 6(81):78090–78099. https://doi.org/10.1039/c6ra13908f
Dri M, Sanna A, Maroto-Valer MM (2014) Mineral carbonation from metal wastes: effect of solid to liquid ratio on the efficiency and characterization of carbonated products. Appl Energy 113:515–523. https://doi.org/10.1016/j.apenergy.2013.07.064
Eikeland E, Blichfeld AB, Tyrsted C et al (2015) Optimized carbonation of magnesium silicate mineral for CO2 storage. ACS Appl Mater Interfaces 7(9):5258–5264. https://doi.org/10.1021/am508432w
Eloneva S, Teir S, Salminen J et al (2008) Steel converter slag as a raw material for precipitation of pure calcium carbonate. Ind Eng Chem Res 47:7104–7111. https://doi.org/10.1021/ie8004034
Fagerlund J, Nduagu E, Romaoãc I, Zevenhoven R (2012) CO2 fixation using magnesium silicate minerals Part 1: process description and performance. Energy 41(1):184–191. https://doi.org/10.1016/j.energy.2011.08.032
Gadikota G, Park AHA (2014) Accelerated carbonation of Ca- and Mg-bearing minerals and industrial wastes using CO2. In: Carbon dioxide utilisation: closing the carbon cycle: First Edition. Elsevier Inc, pp 115–137. https://doi.org/10.1016/B978-0-444-62746-9.00008-6
Gadikota G, Matter J, Kelemen P, Park AHA (2014) Chemical and morphological changes during olivine carbonation for CO2 storage in the presence of NaCl and NaHCO3. Phys Chem Chem Phys 16:4679–4693. https://doi.org/10.1039/c3cp54903h
Galina NR, Arce GLAF, Ávila I (2019) Evolution of carbon capture and storage by mineral carbonation: data analysis and relevance of the theme. Miner Eng 142:105879. https://doi.org/10.1016/j.mineng.2019.105879
Gerdemann SJ, O’Connor WK, Dahlin DC et al (2007) Ex situ aqueous mineral carbonation. Environ Sci Technol 41(7):2587–2593. https://doi.org/10.1021/es0619253
Ghacham AB, Pasquier LC, Cecchi E et al (2016) CO2 sequestration by mineral carbonation of steel slags under ambient temperature: parameters influence, and optimization. Environ Sci Pollut Res 23:17635–17646. https://doi.org/10.1007/s11356-016-6926-4
Guo Y, Tan C, Wang P et al (2019) Magnesium-based basic mixtures derived from earth-abundant natural minerals for CO2 capture in simulated flue gas. Fuel 243:298–305. https://doi.org/10.1016/j.fuel.2019.01.108
Han SJ, Im HJ, Wee JH (2015) Leaching and indirect mineral carbonation performance of coal fly ash-water solution system. Appl Energy 142:274–282. https://doi.org/10.1016/j.apenergy.2014.12.074
Hemmati A, Shayegan J, Bu J et al (2014a) Process optimization for mineral carbonation in aqueous phase. Int J Miner Process 130:20–27. https://doi.org/10.1016/j.minpro.2014.05.007
Hemmati A, Shayegan J, Sharratt P et al (2014b) Solid products characterization in a multi-step mineralization process. Chem Eng J 252:210–219. https://doi.org/10.1016/j.cej.2014.04.112
Hitch M, Dipple GM (2012) Economic feasibility and sensitivity analysis of integrating industrial-scale mineral carbonation into mining operations. Miner Eng 39:268–275. https://doi.org/10.1016/j.mineng.2012.07.007
Hitch M, Ballantyne SM, Hindle SR (2010) Revaluing mine waste rock for carbon capture and storage. Int J Mining, Reclam Environ 24:64–79. https://doi.org/10.1080/17480930902843102
Huijgen WJJ, Witkamp GJ, Comans RNJ (2005) Mineral CO2 sequestration by steel slag carbonation. Environ Sci Technol 39(24):9676–9682. https://doi.org/10.1021/es050795f
Huijgen WJJ, Ruijg GJ, Comans RNJ, Witkamp GJ (2006) Energy consumption and net CO2 sequestration of aqueous mineral carbonation. Ind Eng Chem Res 45(26):9184–9194. https://doi.org/10.1021/ie060636k
Huijgen WJJ, Comans RNJ, Witkamp GJ (2007) Cost evaluation of CO2 sequestration by aqueous mineral carbonation. Energy Convers Manag 48(7):1923–1935. https://doi.org/10.1016/j.enconman.2007.01.035
Ibrahim MH, El-Naas M, Benamor A et al (2019a) Carbon mineralization by reaction with steel-making waste: a review. Processes 7(2):115. https://doi.org/10.3390/pr7020115
Ibrahim MH, El-Naas MH, Zevenhoven R, Al-Sobhi SA (2019b) Enhanced CO2 capture through reaction with steel-making dust in high salinity water. Int J Greenh Gas Control 91:102819. https://doi.org/10.1016/j.ijggc.2019.102819
Iizuka A, Honma M, Hayakawa Y et al (2012) Aqueous mineral carbonation process via concrete sludge. Kagaku Kogaku Ronbunshu 38:129–134. https://doi.org/10.1252/kakoronbunshu.38.129
Iizuka A, Sasaki T, Honma M et al (2017) Pilot-scale operation of a concrete sludge recycling plant and simultaneous production of calcium carbonate. Chem Eng Commun 204(1):79–85. https://doi.org/10.1080/00986445.2016.1235564
IPCC (2005) IPCC special report on carbon dioxide capture and storage. https://www.ipcc.ch/report/carbon-dioxide-capture-and-storage/
IPCC (2014) Climate change 2014: Synthesis report. Contribution of working groups I, II and III to the fifth assessment report of the Intergovernmental Panel on Climate Change. https://www.ipcc.ch/site/assets/uploads/2018/05/SYR_AR5_FINAL_full_wcover.pdf
IPCC (2019) Global warming of 1.5 °C: An IPCC special report on the impacts of global warming of 1.5 °C above pre-industrial levels and related global greenhouse gas emission pathways, in the context of strengthening the global response to the threat of climate change. Intergovernmental Panel on Climate Change. https://www.ipcc.ch/sr15/download/#chapter
Jacobs AD, Hitch M (2011) Experimental mineral carbonation: approaches to accelerate CO2 sequestration in mine waste materials. Int J Mining Reclam Environ 25:321–331. https://doi.org/10.1080/17480930.2011.608883
Jo H, Park SH, Jang YN et al (2014) Metal extraction and indirect mineral carbonation of waste cement material using ammonium salt solutions. Chem Eng J 254:313–323. https://doi.org/10.1016/j.cej.2014.05.129
Julcour C, Bourgeois F, Bonfils B et al (2015) Development of an attrition-leaching hybrid process for direct aqueous mineral carbonation. Chem Eng J 262:716–726. https://doi.org/10.1016/j.cej.2014.10.031
Jung S, Dodbiba G, Fujita T (2014) Mineral carbonation by blowing incineration gas containing CO2 into the solution of fly ash and ammonia for ex situ carbon capture and storage. Geosystem Eng 17(2):125–135. https://doi.org/10.1080/12269328.2014.930358
Kandji EHB, Plante B, Bussière B et al (2017) Geochemical behavior of ultramafic waste rocks with carbon sequestration potential: a case study of the Dumont Nickel Project, Amos, Québec. Environ Sci Pollut Res 24:11734–11751. https://doi.org/10.1007/s11356-017-8735-9
Kelly KE, Silcox GD, Sarofim AF, Pershing DW (2011) An evaluation of ex situ, industrial-scale, aqueous CO2 mineralization. Int J Greenh Gas Control 5:1587–1595. https://doi.org/10.1016/j.ijggc.2011.09.005
Kemache N, Pasquier LC, Cecchi E et al (2017) Aqueous mineral carbonation for CO2 sequestration: from laboratory to pilot scale. Fuel Process Technol 166:209–216. https://doi.org/10.1016/j.fuproc.2017.06.005
Kim MJ, Pak SY, Kim D, Jung S (2017) Optimum conditions for extracting Ca from CKD to store CO2 through indirect mineral carbonation. KSCE J Civ Eng 21(3):629–635. https://doi.org/10.1007/s12205-016-0913-7
Kodama S, Nishimoto T, Yamamoto N et al (2008) Development of a new pH-swing CO2 mineralization process with a recyclable reaction solution. Energy 33(5):776–784. https://doi.org/10.1016/j.energy.2008.01.005
Lackner KS (2002) Carbonate chemistry for sequestering fossil carbon. Annu Rev energy Environ 27:193–232. https://doi.org/10.1146/annurev.energy.27.122001.083433
Lackner KS, Wendt CH, Butt DP et al (1995) Carbon dioxide disposal in carbonate minerals. Energy 20:1153–1170. https://doi.org/10.1016/0360-5442(95)00071-N
Li J, Hitch M (2015a) Ultra-fine grinding and mechanical activation of mine waste rock using a high-speed stirred mill for mineral carbonation. Int J Miner Metall Mater 22:1005–1016. https://doi.org/10.1007/s12613-015-1162-3
Li J, Hitch M (2015b) Carbon dioxide sorption isotherm study on pristine and acid-treated olivine and its application in the vacuum swing adsorption process. Minerals 5:259–275. https://doi.org/10.3390/min5020259
Li J, Hitch M (2016a) Mechanical activation of ultramafic mine waste rock in dry condition for enhanced mineral carbonation. Miner Eng 95:1–4. https://doi.org/10.1016/j.mineng.2016.05.020
Li J, Hitch M (2016b) Characterization of the microstructure of mechanically-activated olivine using X-ray diffraction pattern analysis. Miner Eng 86:24–33. https://doi.org/10.1016/j.mineng.2015.11.010
Li J, Hitch M (2017a) A review on integrated mineral carbonation process in ultramafic mine deposit. Geo-Resour Environ Eng 2:148–154. https://doi.org/10.15273/gree.2017.02.027
Li J, Hitch M (2017b) Ultra-fine grinding and mechanical activation of mine waste rock using a planetary mill for mineral carbonation. Int J Miner Process 158:18–26. https://doi.org/10.1016/j.minpro.2016.11.016
Li J, Hitch M, Power I, Pan Y (2018) Integrated mineral carbonation of ultramafic mine deposits—a review. Minerals 8:147. https://doi.org/10.3390/min8040147
Li J, Jacobs AD, Hitch M (2019) Direct aqueous carbonation on olivine at a CO2 partial pressure of 6.5 MPa. Energy 173:902–910. https://doi.org/10.1016/j.energy.2019.02.125
Liu Q, Liu W, Hu J et al (2018) Energy-efficient mineral carbonation of blast furnace slag with high value-added products. J Clean Prod 197:242–252. https://doi.org/10.1016/j.jclepro.2018.06.150
Mercier G, Blais J-F, Cecchi E, et al (2016) Carbon dioxide chemical sequestration from industrial emissions by carbonation. U.S. Patent 9: 440–189, issued September 13, 2016
Mercier G, Blais J-F, Cecchi E, et al (2018) Carbon dioxide chemical sequestration from industrial emissions by carbonation. U.S. Patent Application 10/150,079, filed December 11, 2018
Naraharisetti PK, Yeo TY, Bu J (2017) Factors influencing CO2 and energy penalties of CO2 mineralization processes. ChemPhysChem 18(22):3189–3202. https://doi.org/10.1002/cphc.201700565
Naraharisetti PK, Yeo TY, Bu J (2019) New classification of CO2 mineralization processes and economic evaluation. Renew Sustain Energy Rev 99:220–233. https://doi.org/10.1016/j.rser.2018.10.008
Ncongwane MS, Broadhurst JL, Petersen J (2018) Assessment of the potential carbon footprint of engineered processes for the mineral carbonation of PGM tailings. Int J Greenh Gas Control 77:70–81. https://doi.org/10.1016/j.ijggc.2018.07.019
O’Connor WK, Dahlin DC, Nilsen DN, et al (2000) Carbon dioxide sequestration by direct mineral carbonation with carbonic acid. No. DOE/ARC-2000-008. Albany Research Center (ARC), Albany, OR. Available online https://www.osti.gov/servlets/purl/896218
O’Connor WK, Dahlin DC, Nilsen DN, et al (2001a) Carbon dioxide sequestration by direct mineral carbonation: results from recent studies and current status. Albany Research Center, OR (US). DOE/ARC-2002-001. Available online https://www.osti.gov/biblio/897885
O’Connor WK, Dahlin DC, Nilsen DN, et al (2001b) Research status on the sequestration of carbon dioxide by direct aqueous mineral carbonation. Albany Research Center, OR (US). DOE/ARC-2001-029. Available online https://www.osti.gov/biblio/897125
O’Connor WK, Dahlin DC, Nilsen DN, et al (2002) Continuing studies on direct aqueous mineral carbonation of CO2 sequestration. Albany Research Center, OR (US). DOE/ARC-2002-003. Available online https://www.osti.gov/biblio/821898
O’Connor WK, Dahlin DC, Rush GE, et al (2005) Aqueous mineral carbonation: Mineral availability, pretreatment, reaction parametrics, and process studies. NETL Energy Technol Lab US DOE Available online http//www webcitation org/query 3051–3075
Olajire AA (2013) A review of mineral carbonation technology in sequestration of CO2. J Pet Sci Eng 109:364–392. https://doi.org/10.1016/j.petrol.2013.03.013
Pan SY, Chiang PC, Chen YH et al (2013) Ex situ CO2 capture by carbonation of steelmaking slag coupled with metalworking wastewater in a rotating packed bed. Environ Sci Technol 47(7):3308–3315. https://doi.org/10.1021/es304975y
Pan SY, Chen YH, Da Chen C et al (2015) High-gravity carbonation process for enhancing CO2 fixation and utilization exemplified by the steelmaking industry. Environ Sci Technol 49(20):12380–12387. https://doi.org/10.1021/acs.est.5b02210
Pan SY, Liu HL, Chang EE et al (2016) Multiple model approach to evaluation of accelerated carbonation for steelmaking slag in a slurry reactor. Chemosphere 154:63–71. https://doi.org/10.1016/j.chemosphere.2016.03.093
Park AHA, Fan LS (2004) CO2 mineral sequestration: physically activated dissolution of serpentine and pH swing process. Chem Eng Sci 59(22–23):5241–5247. https://doi.org/10.1016/j.ces.2004.09.008
Pasquier LC, Mercier G, Blais JF et al (2014a) Parameters optimization for direct flue gas CO2 capture and sequestration by aqueous mineral carbonation using activated serpentinite based mining residue. Appl Geochem. https://doi.org/10.1016/j.apgeochem.2014.08.008
Pasquier LC, Mercier G, Blais JF et al (2014b) Reaction mechanism for the aqueous-phase mineral carbonation of heat-activated serpentine at low temperatures and pressures in flue gas conditions. Environ Sci Technol 48:5163–5170. https://doi.org/10.1021/es405449v
Pasquier LC, Mercier G, Blais JF et al (2016) Technical & economic evaluation of a mineral carbonation process using southern Québec mining wastes for CO2 sequestration of raw flue gas with by-product recovery. Int J Greenh Gas Control. https://doi.org/10.1016/j.ijggc.2016.04.030
Polettini A, Pomi R, Stramazzo A (2016) CO2 sequestration through aqueous accelerated carbonation of BOF slag: a factorial study of parameters effects. J Environ Manage 167:185–195. https://doi.org/10.1016/j.jenvman.2015.11.042
Rashid MI, Benhelal E, Farhang F et al (2019) Development of concurrent grinding for application in aqueous mineral carbonation. J Clean Prod 212:151–161. https://doi.org/10.1016/j.jclepro.2018.11.189
Revathy TDR, Palanivelu K, Ramachandran A (2016) Direct mineral carbonation of steelmaking slag for CO2 sequestration at room temperature. Environ Sci Pollut Res 23:7349–7359. https://doi.org/10.1007/s11356-015-5893-5
Romanov V, Soong Y, Carney C et al (2015) Mineralization of carbon dioxide: a Literature review. ChemBioEng Rev 2:231–256. https://doi.org/10.1002/cben.201500002
Said A, Eloneva S, Fogelholm CJ, et al (2011) Integrated carbon capture and storage for an oxyfuel combustion process by using carbonation of Mg(OH)2 produced from serpentinite rock. In: Energy Procedia. Elsevier Ltd, 2839–2846
Said A, Laukkanen T, Järvinen M (2016) Pilot-scale experimental work on carbon dioxide sequestration using steelmaking slag. Appl Energy 177:602–611. https://doi.org/10.1016/j.apenergy.2016.05.136
Sanna A, Maroto-valer M (2017) CO2 sequestration by ex situ mineral carbonation. World Scientific publishing, Singapore. https://doi.org/10.1142/q0046
Sanna A, Dri M, Maroto-Valer M (2013) Carbon dioxide capture and storage by pH swing aqueous mineralisation using a mixture of ammonium salts and antigorite source. Fuel 114:153–161. https://doi.org/10.1016/j.fuel.2012.08.014
Sanna A, Lacinska A, Styles M, Maroto-Valer MM (2014a) Silicate rock dissolution by ammonium bisulphate for pH swing mineral CO2 sequestration. Fuel Process Technol 120:128–135. https://doi.org/10.1016/j.fuproc.2013.12.012
Sanna A, Uibu M, Caramanna G et al (2014b) A review of mineral carbonation technologies to sequester CO2. Chem Soc Rev 43:8049–8080. https://doi.org/10.1039/C4CS00035H
Saran RK, Arora V, Yadav S (2018) CO2 sequestration by mineral carbonation: a review. Glob Nest J 20:497–503. https://doi.org/10.30955/gnj.002597
Seifritz W (1990) CO2 disposal by means of silicates. Nature 345:486. https://doi.org/10.1038/345486b0
Sipilä J, Teir S, Zevenhoven R (2008) Carbon dioxide sequestration by mineral carbonation Literature review update 2005-2007. Report Vt1: 2008. http://innovationconcepts.nl/res/literatuurGPV/mineralcarbonationliteraturereview0507.pdf
Siriruang C, Toochinda P, Julnipitawong P, Tangtermsirikul S (2016) CO2 capture using fly ash from coal fired power plant and applications of CO2-captured fly ash as a mineral admixture for concrete. J Environ Manage 170:70–78. https://doi.org/10.1016/j.jenvman.2016.01.010
Stocker T (2014) Climate change 2013: the physical science basis: working group I contribution to the Fifth assessment report of the Intergovernmental panel on climate change. Cambridge University Press, Cambridge
Teir S, Revitzer H, Eloneva S et al (2007) Dissolution of natural serpentinite in mineral and organic acids. Int J Miner Process 83:36–46. https://doi.org/10.1016/j.minpro.2007.04.001
Teir S, Eloneva S, Revitzer H et al (2013) Method of producing calcium carbonate from waste and by-products. Energy. 32:528–539. https://doi.org/10.1016/j.energy.2006.06.023
Thom JGM, Dipple GM, Power IM, Harrison AL (2013) Chrysotile dissolution rates: implications for carbon sequestration. Appl Geochem 35:244–254. https://doi.org/10.1016/j.apgeochem.2013.04.016
Torróntegui DM, Mazzotti D-MAVT M, Werner D-MAVT M (2010) Assessing the mineral carbonation science and technology. Master thesis, Swiss Federal Institute of Process Engineering Separation Processes Laboratory, Zürich, Switzerland. Available online https://www.greensand.nl/media/2010%20torrontigue%20assessing%20the%20mineral%20carbonation%20science%20and%20technology.pdf
Uibu M, Velts O, Kuusik R (2010) Developments in CO2 mineral carbonation of oil shale ash. J Hazard Mater 174:209–214. https://doi.org/10.1016/j.jhazmat.2009.09.038
Uibu M, Kuusik R, Andreas L, Kirsimäe K (2011) The CO2-binding by Ca-Mg-silicates in direct aqueous carbonation of oil shale ash and steel slag. Energy Procedia 4:925–932. https://doi.org/10.1016/j.egypro.2011.01.138
UNCC N (2015) Historic Paris agreement on climate change-195 nations set path to keep temperature rise well below 2 °C. UNFCCC Newsroom. Available online http://newsroom.unfccc.int/unfccc-newsroom/finale-cop21
Veetil SP, Mercier G, Blais JF et al (2014) CO2 sequestration by direct dry gas-solid contact of serpentinite mining residues: a solution for industrial CO2 emission. Int J Environ Pollut Remediat 2:52–58. https://doi.org/10.1016/j.minpro.2015.04.024
Veetil SP, Mercier G, Blais JF et al (2015a) Magnetic separation of serpentinite mining residue as a precursor to mineral carbonation. Int J Miner Process 2:52–58. https://doi.org/10.11159/ijepr.2014.008
Veetil SP, Pasquier LC, Blais JF et al (2015b) Direct gas–solid carbonation of serpentinite residues in the absence and presence of water vapor: a feasibility study for carbon dioxide sequestration. Environ Sci Pollut Res 22(17):13486–13495. https://doi.org/10.1007/s11356-015-4580-x
Vicedo-Cabrera AM, Guo Y, Sera F et al (2018) Temperature-related mortality impacts under and beyond Paris agreement climate change scenarios. Clim Change 150:391–402. https://doi.org/10.1007/s10584-018-2274-3
Wang X, Maroto-Valer MM (2011) Integration of CO2 capture and mineral carbonation by using recyclable ammonium salts. Chem Susain Chem 4(9):1291–1300. https://doi.org/10.1002/cssc.201000441
Wang F, Dreisinger DB, Jarvis M, Hitchins T (2018) The technology of CO2 sequestration by mineral carbonation: current status and future prospects. Can Metall Q 57:46–58. https://doi.org/10.1080/00084433.2017.1375221
Werner M, Hariharan S, Mazzotti M (2014) Flue gas CO2 mineralization using thermally activated serpentine: from single- to double-step carbonation. Phys Chem Chem Phys 16:24978–24993. https://doi.org/10.1039/C4CP02786H
Yadav S, Mehra A (2017) Experimental study of dissolution of minerals and CO2 sequestration in steel slag. Waste Manag 64:348–357. https://doi.org/10.1016/j.wasman.2017.03.032
Yan H, Zhang J, Zhao Y et al (2015) CO2 Sequestration by direct aqueous mineral carbonation under low-medium pressure conditions. J Chem Eng Jpn 48(11):937–948. https://doi.org/10.1252/jcej.14we381
Acknowledgements
The authors wish to thank all who assisted in conducting this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: M.Abbaspour.
Rights and permissions
About this article
Cite this article
Veetil, S.P., Hitch, M. Recent developments and challenges of aqueous mineral carbonation: a review. Int. J. Environ. Sci. Technol. 17, 4359–4380 (2020). https://doi.org/10.1007/s13762-020-02776-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02776-z