Abstract
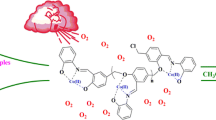
A novel oxo–peroxo tungsten(VI) Schiff base complex, [WO(O2)L(CH3OH)], as a homogeneous catalyst for sulfoxidation was synthesized by treating WO3 in H2O2 with an ONO–tridentate Schiff base ligand. The complex was characterized by FT-IR, UV–Vis, 1H NMR and 13C{1H} NMR spectroscopy. Moreover, the structure of the crystalline tungsten(VI) complex was further investigated by SC-XRD. The crystal structure analysis revealed that the coordination environment of the W complex is seven-coordinate, occupied by ONO sets of the coordinated ligand in a distorted pentagonal–bipyramidal. Also, the immobilized W(VI) Schiff base on chloromethylated polystyrene, showed high efficiency in the oxidation of the sulfide under heterogeneous conditions. The heterogeneous catalyst was systematically characterized by FT-IR, diffuse reflectance UV–Vis, XRD, FE-SEM, EDX, and ICP–AES techniques. Comparatively, the immobilized heterogeneous catalyst showed superior activity over its homogeneous counterpart and was reused several times.











Similar content being viewed by others
References
T. Katsuki, Coord. Chem. Rev. 140, 189 (1995)
G. Licini, V. Conte, A. Coletti, M. Mba, C. Zonta, Coord. Chem. Rev. 255, 2345 (2011)
C. Bolm, Coord. Chem. Rev. 237, 245 (2003)
M. Amini, M.M. Haghdoost, M. Bagherzadeh, Coord. Chem. Rev. 268, 83 (2014)
M. Bagherzadeh, M. Amini, A. Ellern, L.K. Woo, Inorg. Chem. Commun. 15, 52 (2012)
M. Bagherzadeh, M. Zare, V. Amani, A. Ellern, L.K. Woo, Polyhedron 53, 223 (2013)
S.K. Maiti, K.M.A. Malik, S. Gupta, S. Chakraborty, A.K. Ganguli, A.K. Mukherjee, R. Bhattacharyya, Inorg. Chem. 45, 9843 (2006)
R. Curci, J.O. Edwards, in Catalytic Oxidations with Hydrogen Peroxide as Oxidant, vol. 97 ed. by G. Strukul (Kluwer, Dordrecht, 1992)
J.A.L. da Silva, J.J.R.F. da Silva, A.J.L. Pombeiro, Coord. Chem. Rev. 255, 2232 (2011)
M.L. Ramos, L.L.G. Justino, H.D. Burrows, Dalton Trans. 40, 4374 (2011)
N. Mizuno, K. Yamaguchi, K. Kamata, Y. Nakagawa, in Activation of Hydrogen Peroxide by Polyoxometalates (Mechanism in Homogeneous and Heterogeneous Catalytic Epoxidation) ed by T. Oyama (Elsevier, Amsterdam, 155, 2008)
B.S. Lane, K. Burgess, Chem. Rev. 103, 2457 (2003)
N.Y. **, J. Coord. Chem. 65, 4013 (2012)
M. Amini, M. Bagherzadeh, Z. Moradi-Shoeili, D.M. Boghaei, A. Ellern, L.K. Woo, J. Coord. Chem. 66, 464 (2013)
M.A. Fascione, S.J. Adshead, P.K. Mandal, C.A. Kilner, A.G. Leach, W.B. Turnbull, Chem. Eur. J. 18, 2987 (2012)
N. Gharah, S. Chakraborty, A.K. Mukherjee, R. Bhattacharyya, Inorg. Chim. Acta 362, 1089 (2009)
S.K. Maiti, S. Dinda, S. Banerjee, A.K. Mukherjee, R. Bhattacharyya, Eur. J. Inorg. Chem. 2008, 2038 (2008)
M. Bagherzadeh, P. Esmailpour, A. Abbasi, A. Akbari, M. Amini, J. Coord. Chem. 71, 3405 (2018)
L.E. Bevers, P.L. Hagedoorn, W.R. Hagen, Coord. Chem. Rev. 253, 269 (2009)
S.M. Islam, A.S. Roy, P. Mondal, K. Tuhina, M. Mobarak, J. Mondal, Tetrahedron Lett. 53, 127 (2012)
J.P. Colman, L.S. Hegedus, J.R. Norton, R.G. Finkle, Principles and Applications of Organotransition Metal Chemistry, University Science Books, Mill Valley, USA, 1987. D. Cole, J. Hamiltone, Science 299, 1702 (2003)
S.J. Shuttleworth, S.M. Allin, R.D. Wilson, D. Nasturica, Synthesis 1035 (2000)
S.V. Ley, I.R. Baxendale, R.N. Bream, P.S. Jackson, A.G. Leach, D.A. Longhottom, M. Nesi, J.S. Scott, R. Storer, S.J. Taylor, J. Chem. Soc. Perkin Trans. 1, 3815 (2000)
D.C. Sherrington, Catal. Today 57, 81 (2000)
H. Deleuze, X. Schultze, D.C. Sherrington, J. Mol. Catal. A: Chem. 159, 257 (2000)
N.E. Leadbeater, M. Marco, Chem. Rev. 102, 3217 (2002)
H.U. Blaser, B. Pugin, F. Spinder, J. Mol. Catal. A: Chem. 231, 1 (2005)
C. Ren, X. Zhu, N. Zhao, S. Fang, Z. Li, Polymer 204, 122797 (2020)
J. Lu, P.H. Toy, Chem. Rev. 109, 815 (2009)
S. Shanmugam, S. Xu, N.N.M. Adnan, C. Boyer, Macromolecules 51, 779 (2018)
C.J. Whiteoak, A.H. Henseler, C. Ayats, A.W. Kleij, M.A. Pericas, Green Chem. 16, 1552 (2014)
G.R. Krishnan, K. Sreekumar, Eur. J. Org. Chem. 28, 4763 (2008)
V. Mirkhani, S. Tangestaninejad, M. Moghadam, I. Mohammadpoor-Baltork, H. Kargar, J. Mol. Catal. A: Chem. 242, 251 (2005)
M. Hatefi, M. Moghadam, I. Sheikhshoaei, V. Mirkhani, S. Tangestaninejad, I. Mohammadpoor-Baltork, H. Kargar, Appl. Catal. A: Gen. 370, 66 (2009)
M. Moghadam, V. Mirkhani, S. Tangestaninejad, I. Mohammadpoor-Baltork, H. Kargar, I. Sheikhshoaei, M. Hatefi, J. Iran. Chem. Soc. 8, 1019 (2011)
H. Kargar, Inorg. Chem. Commun. 14, 863 (2011)
H. Kargar, Transit. Met. Chem. 39, 811 (2014)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Transit. Met. Chem. 46, 437 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Polyhedron 208, 115428 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Inorg. Chim. Acta 526, 120535 (2021)
H. Kargar, A. Kaka-Naeini, M. Fallah-Mehrjardi, R. Behjatmanesh Ardakani, H. AmiriRudbari, K.S. Munawar, J. Coord. Chem. 74, 1563 (2021)
H. Kargar, M. Fallah-Mehrjardi, J. Iran. Chem. Soc. 18, 3443 (2021)
H. Kargar, J. Iran. Chem. Soc. (2021). https://doi.org/10.1007/s13738-021-02355-0
H. Kargar, A. Moghimi, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, H.A. Rudbari, K.S. Munawar, J. Sulfur Chem. (2021). https://doi.org/10.1080/17415993.2021.1941020
H. Kargar, M. Bazrafshan, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, H.A. Rudbari, K.S. Munawar, M. Ashfaq, M.N. Tahir, Polyhedron 202, 115194 (2021)
H. Kargar, P. Forootan, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, H.A. Rudbari, K.S. Munawar, M. Ashfaq, M.N. Tahir, Inorg. Chim. Acta 523, 120414 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, M. Ashfaq, M.N. Tahir, Inorg. Chim. Acta 527, 120568 (2021)
H. Kargar, M. Fallah-Mehrjardi, R. Behjatmanesh-Ardakani, K.S. Munawar, J. Mol. Struct. 1245, 131259 (2021)
M. Fallah-Mehrjardi, H. Kargar, Inorg. Chem. Commun. 134, 109016 (2021)
H. Kargar, M. Nateghi-Jahromi, M. Fallah-Mehrjardi, R. Behjatmanesh Ardakani, K.S. Munawar, S. Ali, M. Ashfaq, M.N. Tahir, J. Mol. Struct. 1249, 131645 (2022)
X–AREA, version 1.30, program for the acquisition and analysis of data, Stoe & Cie GmbH, Darmstadt, Germany, (2005)
X–RED, version 1.28b, program for data reduction and absorption correction, Stoe & Cie GmbH, Darmstadt, Germany, (2005)
X–SHAPE, version 2.05, program for crystal optimization for numerical absorption correction, Stoe & Cie GmbH, Darmstadt, Germany, (2004)
M.C. Burla, R. Caliandro, M. Camalli, B. Carrozzini, G.L. Cascarano, L. De Caro, C. Giacovazzo, G. Polidori, R. Spagna, J. Appl. Crystallogr. 38, 381 (2005)
G.M. Sheldrick, Acta Crystallogr. A 64, 112 (2008)
A.D. Westland, F. Haque, J.M. Bouchard, Inorg. Chem. 19, 2255 (1980)
H. Oku, N. Ueyama, A. Nakamura, Inorg. Chem. 34, 3667 (1995)
Y. Ishii, H. Tanaka, Y. Nisiyama, Chem. Lett. 1 (1994)
Z. Stec, J. Zawadiak, A. Skibinski, G. Pastuch, Polish J. Chem. 70, 1121 (1996)
R. Neumann, D. Juwiler, Tetrahedron 52, 8781 (1996)
F.M. Collins, A.R. Lucy, C. Sharp, J. Mol. Catal. A: Chem. 117, 397 (1997)
N. Noshiranzadeh, M. Mayeli, R. Bikas, K. S ́lepokura, T. Lis, Transit. Met. Chem. 39, 33 (2014)
Acknowledgements
The support of this work by Ardakan University, Isfahan University, and Payame Noor University is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kargar, H., Moghadam, M., Shariati, L. et al. Novel oxo–peroxo W(VI) Schiff base complex: synthesis, SC-XRD, spectral characterization, supporting on chloromethylated polystyrene, and catalytic oxidation of sulfides. J IRAN CHEM SOC 19, 3067–3077 (2022). https://doi.org/10.1007/s13738-022-02517-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02517-8