Abstract
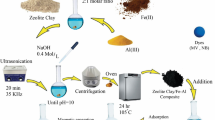
Porous clay adsorption is one of the most economical ways to treat organic dye wastewater. However, it takes a long time and cannot separate mixed organic dyes. In this context, we used three low-concentration polyoxometalates of tungstophosphoric acid, silicotungstic acid and phosphomolybdic acid to synthesize acid-activated sepiolite (SEP) using a simple hydrothermal method. Polyoxometalates with excellent dye adsorption ability and separation ability can acidify sepiolite. From the characterization of the sample, it was found that the sepiolite after acid activation had no great influence on its crystal structure and chemical composition. It is worth noting that the morphology of the sample changed. The adsorption test experiment shows that the adsorption capacity of SEP after POMs activation is better than that of raw SEP or SEP treated with NaCl and HCl. The 0.10 mol/L H3PMo12O40-treated SEP can quickly and efficiently capture cationic dyes from mixed dye solutions, and it also has effective separation ability in MB/MO and MO/RhB mixed solutions.











Similar content being viewed by others
References
A.M. Ahmat, T. Thiebault, R. Guégan, Appl. Clay Sci. 180, 105–188 (2019)
H. Albatrni, H. Qiblawey, F. Almomani, S. Adham, M. Khraisheh, Chemosphere 233, 809–817 (2019)
G. Annadurai, R.S. Juang, D.J. Lee, Hazard Mater 92, 263–274 (2002)
M. Attoui, E. Pouget, R. Oda, D. Talaga, L.G. Bourdon, T. Buffeteau, S. Nlate, Chem. A Eur. J 24, 11236–11236 (2018)
K.V. Avramidou, F. Zaccheria, S.A. Karakoulia, K.S. Triantafyllidis, N. Ravasio, Mol. Catal. 439, 60–71 (2017)
S. Bakhtiary, M. Shirvani, H. Shariatmadari, Micropor. Mesopor. Mat. 168, 30–36 (2013)
C.F. Barranco, A.E. Kozioł, K. Skrzypiec, M. Rawski, M. Drewniak, A.Y. Rodriguez, Appl. Clay Sci. 127–128, 129–133 (2016)
M. Bergaoui, A. Nakhli, Y. Benguerba, M. Khalfaoui, A. Erto, F.E. Soetaredjo, S. Ismadji, B. Ernst, J. Mol. Liq. 272, 697–707 (2018)
Brindley, Am Mineral 44, 495–500(1959).
B. Chen, Y. Jia, M. Zhang, X. Li, J. Yang, X. Zhang, Appl. Clay Sci. 174, 1–9 (2019)
C. Cheng, L. Ma, J. Ren, L. Li, G. Zhang, Q. Yang, C. Zhao, Chem. Eng. J. 171, 1132–1142 (2011)
M. Cobas, L. Ferreira, M.A. Sanromán, M. Pazos, Ecol Eng 70, 287–294 (2014)
S. Cotillas, C. Sáez, P. Cañizares, I. Cretescu, M.A. Rodrigo, Sep. Purif. Technol. 194, 19–25 (2018)
H. Dai, Y. Huang, H. Huang, Carbohyd. Polym. 185, 1–11 (2018)
L.Y. Dai, M. Gong, T. Yu, X. Chen, J. Alloy. Compd. 638, 435–444 (2015)
A. Demirbas, Hazard Mater. 167, 1–9 (2009)
C. Deng, J. Wen, Z. Li, N. Luo, M. Huang, R. Yang, Ecotoxicol. Environ. Saf. 164, 270–276 (2018)
E. Derakhshani, A. Naghizadeh, J. Mol. Liq. 259, 76–81 (2018)
D. Du, G. Tang, Zhang and X. Wu, Chinese J Catal 36, 2219–2228 (2015)
N. Frini-Srasra, E. Srasra, Desalination 250, 26–34 (2010)
H. Gan, G. Zhang, Y. Guo, Colloid. Interface Sci. 386, 373–380 (2012)
M. Genovese, Y.W. Foong, K. Lian, Electrochim. Acta 199, 261–269 (2016)
W. Grünert, R. Feldhaus, K. Anders, E.S. Shpiro, G.V. Antoshin, K.M. Minachev, J. Electron Spectrosc. Relat. Phenom. 40, 187–192 (1986)
S. Jana, J. Ray, B. Mondal, T. Tripathy, Appl. Clay Sci. 3, 46–64 (2019)
Z. Jiang, L. Yonghong, X. Lei, L. Zhiyuan, Ecotoxicol. Environ. Saf. 165, 115–125 (2018)
D. Karadag, M. Turan, E. Akgul, S. Tok, A. Faki, J. Chem. Eng. Data 52, 1615–1620 (2007)
S.D. Khattri, M.K. Singh, Water Air Soil Pollut. 120, 283–294 (2000)
S.D. Khattri and M.K. Singh, Adsorpt. Sci. Technol. 17, 26982 (1999).
S. Letaief, S. Grant and C. Detellier, Appl. Clay Sci. 53, 0–243 (2011).
F. Li, Y. Jiang, M. **a, M. Sun, B. Xue, X. Ren, J. Hazard. Mater. 165, 1219–1223 (2009)
J. Li, J. Cai, L. Zhong, H. Cheng, H. Wang, Q. Ma, Appl. Clay Sci. 16, 9–22 (2018)
Q. Li, L. Zhang, Y. Xu, Q. Li, H. Xue, H. Pang, ACS Sustain. Chem. Eng. 7, 5027–5033 (2019)
L. Liu, S. Wang, W. Chen, E. Wang, Mater. Lett. 78, 22–24 (2012)
A. Maleki, M. Mohammad, Z. Emdadi, N. Asim, M. Azizi and J. Safaei, Arab. J. Chem. (2018).
C. Mani, M. Ramalingam, S. Manickam, K.R. Srinivasalu, E. Deivanayagam, B.I. Mohammed, Chem. Select 31, 9934–9942 (2017)
A. Martínez-de la Cruz and S. Obrego´n Alfaro, Solid State Sci. 11, 829–835(2009).
S. Megala, S. Prabhu, S. Harish, M. Navaneethan, S. Sohila, R. Ramesh, Appl. Surf. Sci. 481, 385–393 (2019)
A. Miura, K. Nakazawa, T. Takei, N. Kumada, N. Kinomura and R. Ohki, Ceram. Int. 38, 0–0(2012).
M.A. Moreira, K.J. Ciuffi, V. Rives, M.A. Vicente, Appl. Clay. Sci. 135, 394–404 (2017)
P.P. Nadiminti, H. Sharma, S.R. Kada, F.M. Pfeffer, L.A. O’Dell, D.M. Cahill, ACS Sustain. Chem. Eng. 7, 10962–10970 (2019)
M.S. Nikolic, R. Petrovic, D. Veljovic, V. Cosovic, N. Stankovic, J. Djonlagic, Eur. Polymer J. 97, 198–209 (2017)
A.S. Özcan, B. Erdem, A. Özcan, Coll. Surf., A 266, 73–81 (2005)
A.S. Özcan, A. Özcan, Sep. Sci. Technol. 3, 301–320 (2005)
J. Pan, X. Zou, X. Wang, W. Guan, C. Li, Y. Yan, X. Wu, Chem. Eng. J. 166, 40–48 (2011)
D. Papoulis, S. Komarneni, D. Panagiotaras, A. Nikolopoulou, H. Li, S. Yin, S. Tsugio, H. Katsuki, Appl. Clay. Sci. 83–84, 191–197 (2013)
S. S. Patil, M. G. Mali, A. Roy, M. S. Tamboli, V. G. Deonikar and D. R. Patil, Energy Chem., 0, 1–9(2016).
C. R. Santos Sílvia and A. R. Boaventura Rui, Chem. Eng. J., 4, 1473–1483(2016).
Y.A. Sethi, R.P. Panmand, S.R. Kadam, A.K. Kulkarni, B.B. Kale, J. Colloid Interface Sci. 487, 504–512 (2017)
F. E. Tuler, R. Portela, P. ávila, J. P. Bortolozzi, E. E. Miró and V. G. Milt, Micropor. Mesopor. Mater. 230, 11–19(2016).
Y. Turhan, P. Turan, M. Dogˇan, M. Alkan, H. Namli, O. Demirbas, Ind. Eng. Che. Res. 47, 1883–1895 (2008)
M. Uğurlu, Micropor. Mesopor. Mater. 119, 276–283 (2009)
J.L. Valentín, M.A. López-Manchado, A. Rodríguez, P. Posadas, L. Ibarra, Appl. Clay. Sci. 36, 245–255 (2007)
K. Vignesh, R. Hariharan, M. Rajarajan, A. Suganthi, Solid State Sci. 21, 91–99 (2013)
V. Vimonses, S. Lei, B. **, C.W.K. Chow, C. Saint, Chem. Eng. J. 148, 354–364 (2009)
D. Wang, Y. Xu, F. Sun, Q. Zhang, P. Wang, X. Wang, Appl. Surf. Sci. 377, 221–227 (2016)
X.S. Wang, J. Liang, L. Li, Z.J. Lin, P.P. Bag, S.Y. Gao, Inorg. Chem. 55, 2641–2649 (2016)
X.S. Wang, L. Liang, H. Liu, X. **e, Q. Hao, Mater. Lett. 161, 336–339 (2015)
S. Yang, C. Ding, W. Cheng, Z. **, Y. Sun, J. Mol. Liq. 204, 170–175 (2015)
S. Yang, Y. Wang, S. Lin, J. Fan, C. Liu, X. Yan, Chemosphere 19, 117–122 (2018)
L.D. Youcef, L.S. Belaroui, A.L. Galindo, Appl. Clay Sci. 179, 105–145 (2019)
S. Yu, L. Zhai, S. Zhong, Y. Qiu, L. Cheng, X. Ren, J. Taiwan Inst. Chem. E. 59, 221–228 (2016)
Y. O. Zdemir, M. Dog˘an and M. Alkan, Micropor. Mesopor. Mater., 96, 419–427(2006).
G. Zhang, Q. **ong, W. Xu, S. Guo, Appl. Clay Sci. 102, 231–237 (2014)
M.G. Zhang, Chem. Eng. J. 288, 70–78 (2016)
F. Zhou, C. Yan, Y. Zhang, J. Tan, H. Wang, S. Zhou, S. Pu, Appl. Clay. Sci. 124–125, 119–126 (2016)
R. Hojiyev, G. Ersever, İE. Karaağaçlıoğlu, F. Karakaş, F. Boylu, Appl. Clay. Sci. 128, 105–110 (2016)
S. Zhu, Q. Li, F. Li, W. Cao, T.H. Li, J. Phys. Chem. Solids. 92, 11–18 (2016)
S. Vahidhabanu, A. Abideen Idowu, D. Karuppasamy, ACS Sustain. Chem. Eng., 5, 10361–10370 (2017).
Acknowledgements
The authors acknowledge with thanks the financial support of Hunan 2011 Collaborative Innovation Center of Chemical Engineering & Technology with Environmental Benignity and Effective Resource Utilization and the National Natural Science Foundation of China (51965009) and Science and Technology Plan Project of Guizhou Province:[2019]5616.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflict to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Song, B., Chi, X., Zhou, M. et al. Enhanced adsorption and dye separation ability of low-cost sepiolite acidified by polyoxometalate acid. J IRAN CHEM SOC 19, 1457–1465 (2022). https://doi.org/10.1007/s13738-021-02396-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02396-5