Abstract
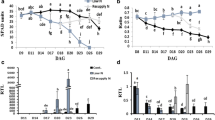
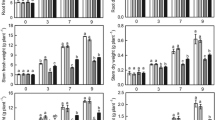
Nitrogen (N) remobilization is an important N metabolic process in plants. The objective of this study was to increase our understanding of the relationship between low N tolerance and N remobilization in cucumber. Two cucumber cultivars, namely D0328 and D0422, were included in the analyses, the former of which displays a stronger capacity for low N tolerance. Source leaf transcriptomes were analyzed, which revealed differentially expressed genes in a number of interesting pathways, including amino acid biosynthesis, tyrosine and tryptophan biosynthesis, plant hormone signal transduction, and E3 ligase activity, implying that these pathways are involved in low N tolerance in cucumber. In agreement with transcriptome data, old D0328 leaves had significantly higher tryptophan and indole-3-acetic acid contents than old D0422 leaves under low N conditions. N remobilization efficiency in old leaves was increased by naphthaleneacetic acid treatment, whereas it was repressed by antiauxins α-(p-chlorophenoxy) isobutyric acid treatment. Taken together, the results of this study demonstrate that auxin promotes N remobilization in cucumber leaves during the reproductive stage of plant development and this positive regulation is associated with low N tolerance in cucumber.





Similar content being viewed by others
References
Baxter R, Grantley M, Ashenden TW, Farrar J (1994) Effects of elevated carbon dioxide on three grass species from montane pasture. II Nutrient allocation and efficiency of nutrient use. J Exp Bot 45:1267–1278
Bi YM, Wang RL, Zhu T, Rothstein SJ (2007) Global transcription profiling reveals differential response to chronic nitrogen stress and putative regulatory components in Arabidopsis. BMC Genom 8:281–298
Brian GF (2002) Local and long-range signaling pathways regulating plant responses to nitrate. Annu Rev Plant Biol 53:203–224
Chen R, Tian M, Wu X, Huang Y (2011) Differential global gene expression changes in response to low nitrogen stress in two maize inbred lines with contrasting low nitrogen tolerance. Genes Genomics 33:491–497
Coruzzi GM, Zhou L (2001) Carbon and nitrogen sensing and signaling in plants: emerging ‘matrix effects’. Curr Opin Plant Biol 4:247–253
Diaz C, Lemaître T, Christ A, Azzopardi M, Kato Y, Sato F, Morot-Gaudry JF, Le Dily F, Masclaux-Daubresse C (2008) Nitrogen recycling and remobilization are differentially controlled by leaf senescence and development stage in Arabidopsis under low nitrogen nutrition. Plant Physiol 147:1437–1449
Fan SC, Lin CS, Hsu PK, Lin SH, Tsay YF (2009) The Arabidopsis nitrate transporter NRT1.7, expressed in phloem, is responsible for source-to-sink remobilization of nitrate. Plant Cell 21:2750–2761
Fujiwara T, Hirai MY, Chino M, Komeda Y, Naito S (1992) Effects of sulfur nutrition on expression of the soybean seed storage protein genes in transgenic petunia. Plant Physiol 99:263–268
Gray WM, Estelle I (2000) Function of the ubiquitin–proteasome pathway in auxin response. Trends Biochem Sci 25:133–138
Hannah MA, Caldana C, Steinhauser D, Balbo I, Fernie AR, Willmitzer L (2010) Combined transcript and metabolite profiling of Arabidopsis grown under widely variant growth conditions facilitates the identification of novel metabolite-mediated regulation of gene expression. Plant Physiol 152:2120–2129
Kant S, Bi YM, Rothstein SJ (2011) Understanding plant response to nitrogen limitation for the improvement of crop nitrogen use efficiency. J Exp Bot 62:1499–1509
Kiba T, Kudo T, Kojima M, Sakakibara H (2011) Hormonal control of nitrogen acquisition: roles of auxin, abscisic acid, and cytokinin. J Exp Bot 62:1399–1409
Lezhneva L, Kiba T, Feria-Bourrellier AB, Lafouge F, Boutet-Mercey S, Zoufan P, Sakakibara H, Daniel-Vedele F, Krapp A (2014) The Arabidopsis nitrate transporter NRT2.5 plays a role in nitrate acquisition and remobilization in nitrogen-starved plants. Plant J 80:230–241
Liu J, Wu YH, Yan JJ, Liu YD, Shen FF (2008) Protein degradation and nitrogen remobilization during leaf senescence. J Plant Biol 51:11–19
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T) (−Delta Delta C) method. Methods 25:402–408
Ma W, Li J, Qu B, He X, Zhao X, Li B, Fu X, Tong Y (2014) Auxin biosynthetic gene TAR2 is involved in low nitrogen-mediated reprogramming of root architecture in Arabidopsis. Plant J 78:70–79
Martin A, Belastegui-Macadam X, Quillere I, Floriot M, Valadier MH, Pommel B, Andrieu B, Donnison I, Hirel B (2005) Nitrogen management and senescence in two maize hybrids differing in the persistence of leaf greenness: agronomic, physiological and molecular aspects. New Phytol 167:483–492
Masclaux-Daubresse C, Reisdorf-Cren M, Pageau K (2006) Glutamine synthetase-glutamate synthase pathway and glutamate dehydrogenase play distinct roles in the sink-source nitrogen cycle in tobacco (Nicotiana tabacum L.). Plant Physiol 140:444–456
Masclaux-Daubresse C, Reisdorf-Cren M, Orsel M (2008) Leaf nitrogen remobilisation for plant development and grain filling. Plant Biol 10:23–36
Miller AJ, Fan X, Shen Q, Smith SJ (2008) Amino acids and nitrate as signals for the regulation of nitrogen acquisition. J Exp Bot 59:111–119
Peoples MB, Freney JR, Mosier AR (1995) Minimizing gaseous losses of nitrogen. In: Bacon PE (eds) Nitrogen fertilization in the environment. Marcel Dekker Inc, New York, pp 565–602
Pettersson R, MacDonald JS, Stadenburg I (1993) Response of small birch plants (Betula pendula Roth.) to elevated CO2 and nitrogen supply. Plant, Cell Environ 16:1115–1121
Sanders A, Collier R, Trethewy A, Gould G, Sieker R, Tegeder M (2009) AAP1 regulates import of amino acids into develo** Arabidopsis embryos. Plant J 59:540–552
Schulze W, Schulze ED, Stadler J (1994) Growth and reproduction of Arabidopsis in relation to storage of starch and nitrate in the wild-type and in starch-deficient and nitrate uptake-deficient mutants. Plant, Cell Environ 17:795–809
Stone SL, Hauksdóttir H, Troy A, Herschleb J, Kraft E, Callis J (2005) Functional analysis of the RING-type ubiquitin ligase family of Arabidopsis. Plant Physiol 137:13–30
Tabuchi M, Abiko T, Yamaya T (2007) Assimilation of ammonium ions and reutilization of nitrogen in rice (Oryza sativa L.). J Exp Bot 58:2319–2327
Tian Q, Chen F, Liu J, Zhang F, Mi G (2008) Inhibition of maize root growth by high nitrate supply is correlated with reduced IAA levels in roots. J Plant Physiol 165:942–951
Wang B, Chu J, Yu T, Xu Q, Sun X, Yuan J, **ong G, Wang G, Wang Y, Li J (2015) Tryptophan-independent auxin biosynthesis contributes to early embryogenesis in Arabidopsis. Proc Natl Acad Sci USA 112:4821–4826
Wu T, Cao J, Zhang Y (2008) Comparison of antioxidant activities and endogenous hormone levels between bush and vine-type tropical pumpkin (Cucurbita moschata Duchesne). Sci Hortic 116:27–33
Wu T, Qin Z, Zhou X, Feng Z, Du Y (2010) Transcriptome profile analysis of floral sex determination in cucumber. J Plant Physiol 167:905–913
**e Q, Guo HS, Dallman G, Fang S, Weissman AM, Chua NH (2002) SINAT5 promotes ubiquitin-related degradation of NAC1 to attenuate auxin signals. Nature 419:167–170
Yu ML, Qin ZW, Xu JJ, Zhou XY (2011) Screening of cucumber genotypes with low-nitrogen tolerance and its genetic analysis. China Veg 1:46–51 (in Chinese)
Zhao W, Yang X, Yu H, Jiang W, Sun N, Liu X, Liu X, Zhang X, Wang Y, Gu X (2015) RNA-Seq-Based transcriptome profiling of early nitrogen deficiency response in cucumber seedlings provides new insight into the putative nitrogen regulatory network. Plant Cell Physiol 56:455–467
Acknowledgements
This work was supported by National Natural Science Foundation of China (No. 31101545); Program for New Century Excellent Talents in Heilongjiang Provincial University (No. 1452G03); University Nursing Program for Young Scholars with Creative Talents in Heilongjiang Province (No. UNPYSCT-2015001); ‘Academic backbone’ Project of Northeast Agricultural University (No. 16XG05); ‘Young Talents’ Project of Northeast Agricultural University (No. 14QC07); Key Laboratory of Biology and Genetic Improvement of Horticultural Crops (Northeast Region), Ministry of Agriculture/Northeast Agricultural University (No. neauhc201601).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Du, Y., Fan, L., Tian, C. et al. Auxin positively regulates nitrogen remobilization in cucumber leaves. Hortic. Environ. Biotechnol. 59, 189–198 (2018). https://doi.org/10.1007/s13580-018-0020-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-018-0020-4