Abstract
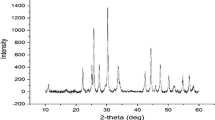
Synthesizing nanomixed metal oxides via green routes and adopting them as adsorbents in water remediation methods is an important aspect of chemical research. In the present investigation, nanoparticles of mixed oxides of Mn-Al–Fe of size 6.19 nm are successfully synthesized adopting ‘aloe vera gel’ as cap** agent and homogenous precipitation method for generating the precipitant (OH−) by urea hydrolysis, in a viscous mother liquor of composition: ‘glycerol:water (20:80)’. These nanoparticles are immobilized in Ca-alginate beads to prevent agglomeration. The mixed nanoparticles and beads are characterized and investigated for their sorption nature towards toxic chromate and phosphate ions. The adsorption mechanism is investigated by thermodynamic studies, isothermal and kinetic models. It is observed that chromate and phosphate can be simultaneously removed from waste water with the nanomixed metal oxides and beads at pH 7 and 10 respectively. The sorption capacities are 71.8 mg/g for phosphate and 87.3 mg/g for chromate with nanomixed metal oxides and 94.3 mg/g for phosphate and 109.8 mg/g for chromate with beads. The bead structure enhances sorption capacities besides permitting easy filtration. Thermodynamic studies confirm the endothermic and spontaneous nature of the adsorption. Pseudo 2nd-order kinetic models and Langmuir isotherms explain well the sorption mechanism. The adsorbents can be recycled for six cycles of re-generations and re-use. The sorbents developed are successfully applied for simultaneous remediation of chromate and phosphate from polluted water.
Graphical abstract












Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Ai T, Jiang X, Liu Q (2018) Chromium removal from industrial wastewater using Phyllostachys pubescens biomass loaded Cu-S nanospheres. Open Chem 16:842–852
Alam JB, Dikshit AK, Bandyopadhayay M (2005) Evaluation of thermodynamic properties of sorption of 2, 4-D and atrazine by tire rubber granules. Sep Purif Technol 42(1):85–90. https://doi.org/10.1016/j.seppur.2004.06.006
Apha APHA (1998) Standard methods for the examination of water and waste water. APHA, Washington, DC
Arshadi M, Eskandarloo H, Azizi M, Abbaspourrad A, Abdolmaleki MK, Eskandarloo H, Azizi M, Abbaspourrad A (2018) Synthesis of highly monodispersed, stable, and spherical nZVI of 20–30 nm on filter paper for the removal of phosphate from wastewater: batch and column study. ACS Sustain Chem Eng 6:11662–11676. https://doi.org/10.1021/acssuschemeng.8b01885
ASTM D4607–94 (2006) Standard test method for determination of iodine number of activated carbon.
Badessa T, Shaposhnik V (2016) The electrodialysis of electrolyte solutions of multi-charged cations. J Membr Sci 498:86–93. https://doi.org/10.1016/j.memsci.2015.09.017
Badessa TS, Wakuma E, Yimer AM (2020) Bio-sorption for effective removal of chromium (VI) from wastewater using Moringa stenopetala seed powder (MSSP) and banana peel powder (BPP). BMC Chemistry 14(1):1–2. https://doi.org/10.1186/s13065-020-00724-z
Biftu WK, Kunta R (2021c) Iron-alginate beads doped with green synthesised ‘nano-CeO2-ZrO2’as an effective adsorbent for removal of highly toxic arsenic-ions from polluted waterInt J Environ Anal Chem 1–19https://doi.org/10.1080/03067319.2021.1875452
Biftu WK, Mekala S, Ravindhranath K (2020) De-fluoridation of polluted water using aluminium alginate beads doped with green synthesized ‘nano SiO2+ nano CeO2-ZrO2’, as an effective adsorbent. Chemistry Select 5(47):15061–15074. https://doi.org/10.1002/slct.202003744
Biftu WK, Ravindhranath K (2020) Synthesis of nanoZrO2 via simple new green routes and its effective application as adsorbent in phosphate remediation of water with or without immobilization in Al-alginate beads. Water Sci Technol 81(12):2617–2633. https://doi.org/10.2166/wst.2020.318
Biftu WK, Ravindhranath K (2021) Calcium alginate beads doped with nano-ZrO2 and activated carbon of Annona reticulata plant as an effective adsorbent for water remediation of chromium(VI). Asian J Chem 33(2):281–290. https://doi.org/10.14233/ajchem.2021.22953
Biftu WK, Ravindhranath K, Ramamoorty M (2020) New research trends in the processing and applications of iron-based nanoparticles as adsorbents in water remediation methods. Nanotechnol Environ Eng 5:1–12. https://doi.org/10.1007/s41204-020-00076-y
Biftu WK, Suneetha M, Ravindhranath K (2021) Sequential adsorptive removal of phosphate, nitrate and chromate from polluted water using active carbon derived from stems of Carissa carandas plant. Water Pract Technol, IWA Publishing 16(1):117–134. https://doi.org/10.2166/wpt.2020.102
Biftu WK, Suneetha M, Ravindhranath K (2021b) Zirconium-alginate beads doped with H2SO4-activated carbon derived from leaves of Magnoliaceae plant as an effective adsorbent for the removal of chromate. Biomass Convers Biorefin 1-16https://doi.org/10.1007/s13399-021-01568-w
BIS (Bureau of Indian Standards) (1989) Activated carbon, powdered and granular-methods of sampling and its tests BIS New Delhi 877.
Biswas K, Gupta K, Goswami A (2010) Ghosh UC Fluoride removal efficiency from aqueous solution by synthetic iron(III)–aluminum(III)–chromium(III) ternary mixed oxide. Desalination 255(1–3):44–51. https://doi.org/10.1016/j.desal.2010.01.019
Bouamra F, Drouiche N, Abdi N, Grib H, Mameri N, Lounici H (2018) Removal of phosphate from wastewater by adsorption on marble waste: effect of process parameters and kinetic modeling. Int J Environ Res 12:13–27. https://doi.org/10.1007/s41742-018-0065-3
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60(2):309–319. https://doi.org/10.1021/ja01269a023
Bunce JT, Ndam E, Ofiteru ID, Moore A, Graham DW (2018) A review of phosphorus removal technologies and their applicability to small-scale domestic wastewater treatment systems. Front Environ Sci 6:8. https://doi.org/10.3389/fenvs.2018.00008
Bureau of Indian Standards (1991) IS: 10500.
Cai W, Fu F, Zhu L, Tang B (2019) Simultaneous removal of chromium(VI) and phosphate from water using easily separable magnetite/pyrite nanocomposite. J Alloy Compd 803:118–125. https://doi.org/10.1016/j.jallcom.2019.06.285
Chen D, **ao X, Yang K (2016) Removal of phosphate and hexavalent chromium from aqueous solutions by engineered waste eggshell. RSC Adv. https://doi.org/10.1039/C6RA05034D
Corbett JF (1972) Pseudo first-order kinetics. J Chem Educ 49:663. https://doi.org/10.1016/j.jece.2018.01.014
Dorofeev GA, Streletskii AN, Povstugar IV, Protasov AV, Elsukov EP (2012) Determination of nanoparticle sizes by X-ray diffraction. Colloid J 74(6):675–685. https://doi.org/10.1134/S1061933X12060051
Duan H, Zhang L, Wang Y, Liu Y, Wang Y (2021) Phosphate removal from aqueous solution by Fe-La binary (hydr)oxides: a study of batch experiments and mechanisms. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-15127-3
Dubinin MM, Radushkevich LV (1947) The equation of the characteristic curve of the activated charcoal. Proc Acad Sci Phys Chem Sect USSR 55:331–333
El-Hendawy ANA, Samra SE, Girgis BS (2001) Adsorption characteristics of activated carbons obtained from corncobs. Colloids Surf, A 180(3):209–221. https://doi.org/10.1016/S0927-7757(00)00682-8
Eljamal O, Khalil AME, Matsunaga N (2017) Experimental and modeling column study of phosphorus removal by permeable reactive materials. Int J Environ Agric Res 3(1):62–70
Eljamal O, Okawauchi J, Hiramatsu K (2014) Product rich in phosphorus produced from phosphorus-contaminated water. Adv Mater Res 894:261–265
Eljamal R, Eljamal O, Maamoun I, Yilmaz G, Sugihara Y (2020) Enhancing the characteristics and reactivity of nZVI: polymer’s effect and mechanisms. J Mol Liq 315:113714. https://doi.org/10.1016/j.molliq.2020.113714
Fan C, Zhang Y (2018) Adsorption isotherms, kinetics and thermodynamics of nitrate and phosphate in binary systems on a novel adsorbent derived from corn stalks. J Geochem Explor 188:95–100. https://doi.org/10.1016/j.gexplo.2018.01.020
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57(385471):1100–1107
George WK, Eren Y, Ayd S, Emik S, Tuba A, Osra F, Wasswa J (2019) A facile polymerisation of magnetic coal to enhanced phosphate removal from solution. J Environ Manag 247:356–362. https://doi.org/10.1016/j.jenvman.2019.06.088
Hall KR, Eagleton LC, Acrivos A, Vermeulen T (1966) Pore-and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions. Ind Eng Chem Fundam 5(2):212–223. https://doi.org/10.1021/i160018a011
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465. https://doi.org/10.1007/s11356-019-05050-z
Huang Z, Wang X, Yang D (2015) Adsorption of Cr (VI) in wastewater using magnetic multi-wall carbon nanotube. Water Sci Eng 8(3):226–232. https://doi.org/10.1016/j.wse.2015.01.009
Jiang J, Yuan M, Xu R, Bish DL (2015) Mobilization of phosphate in variable-charge soils amended with biochars derived from crop straws. Soil Tillage Res 146:139–147. https://doi.org/10.1016/j.still.2014.10.009
** T, Yuan W, Xue Y, Wei H, Zhang C, Li K (2017) Co-modified MCM-41 as an effective adsorbent for levofloxacin removal from aqueous solution: optimization of process parameters, isotherm, and thermodynamic studies. Environ Sci Pollut Res 24(6):5238–5248. https://doi.org/10.1007/s11356-016-8262-0
Kahani SA, Jafari M (2009) A new method for preparation of magnetite from iron oxyhydroxide or iron oxide and ferrous salt in aqueous solution. J Magn Magn Mater 321(13):1951–1954. https://doi.org/10.1016/j.jmmm.2008.12.026
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinu. J Am Chem Soc 40(9):1361–1403. https://doi.org/10.1021/ja02242a004
Lu J, Liu D, Hao J, Zhang G, Lu B (2005) Phosphate removal from aqueous solutions by a nano-structured Fe–Ti bimetal oxide sorbent. Chem Eng Res Des 93:652–661. https://doi.org/10.1016/j.cherd.2014.05.001
Lu J, Liu H, Liu R, Zhao X (2013) Adsorptive removal of phosphate by a nanostructured Fe–Al–Mn tri metal oxide adsorbent. Powder Technol 233:146–154. https://doi.org/10.1016/j.powtec.2012.08.024
Lv X, Jiang XXue G, Wu D, Sheng T, Zhou H, Xu X (2014) Journal of colloid and interface science 417–51.
Lv X, Xue G, Jiang J, Tang X Xu (2012) Journal of colloid and interface science 369–460.
Maamoun I, Eljamal O, Eljamal R, Falyouna O, Sugihara Y (2020c) Promoting aqueous and transport characteristics of highly reactive nanoscale zero valent iron via different layered hydroxide coatings. Appl Surf Sci 145018https://doi.org/10.1016/j.apsusc.2019.145018
Maamoun I, Eljamal O, Falyouna O, Eljamal R, Sugihara Y (2020) multi-objective optimization of permeable reactive barrier design for Cr (VI) removal from groundwater. Ecotoxicol Environ Saf 200:110773. https://doi.org/10.1016/j.ecoenv.2020.110773
Maamoun I, Eljamal O, Falyouna O, Eljamal R, Sugihara Y (2020) Stimulating effect of magnesium hydroxide on aqueous characteristics of iron nanocomposites. Water Sci Technol. https://doi.org/10.2166/wst.2020.027
Maamoun I, Falyouna O, Eljamal R, Bensaida K, Eljamal O (2021) Optimization modeling of nFe0/Cu-PRB design for Cr(VI) removal from groundwater. International J Environ Sci Dev 12:(5). https://doi.org/10.18178/ijesd.2021.12.5.1330
Mahdavi S, Akhzari D (2016) The removal of phosphate from aqueous solutions using two nano-structures: copper oxide and carbon tubes. Clean Technol Environ Policy 18(3):817–827. https://doi.org/10.1007/s10098-015-1058-y
Mekonnen DT, Alemayehu E, Lennartz B (2021) Adsorptive removal of phosphate from aqueous solutions using low-cost volcanic rocks: kinetics and equilibrium approaches. Materials 14:1312. https://doi.org/10.3390/ma14051312
Mohammad A, Inamuddin Hussain S (2014) Poly (3,4-ethylenedioxythiophene): polystyrene sulfonate (PEDOT: PSS) Zr(IV) phosphate composite cation exchanger: sol-gel synthesis and physicochemical characterization. Ionics 21:1063–1071. https://doi.org/10.1007/s11581-014-1247-4
Mohan GK, Babu AN, Kalpana K, Ravindhranath K (2019) Removal of chromate (VI) from water using adsorbent derived from spent coffee grounds. Int J Environ Sci Technol 16(1):101–112. https://doi.org/10.1007/s13762-017-1593-7
Muhammad A, **ng X, Baoyu G, Qinyan Y, Shang Y, Rizwan K, Muhamma AI (2020) Adsorptive removal of phosphate by the bimetallic hydroxide nanocomposites embedded in pomegranate peel. J Environ Sci 91:189–198. https://doi.org/10.1016/j.jes.2020.02.005
Onyango MS, Kojima Y, Aoyi O, Bernardo EC, Matsuda H (2004) Adsorption equilibrium modeling and solution chemistry dependence of fluoride removal from water by trivalent-cation-exchanged zeolite F-9. J Colloid Interface Sci 279(2):341–350. https://doi.org/10.1016/j.jcis.2004.06.038
Owlad M, Aroua MK, Wan Daud WA, Baroutian S (2008) Removal of hexavalent chromium-contaminated water and wastewater: a review. Water Air Soil Pollut. https://doi.org/10.1007/s11270-008-9893-7
Panda H, Tiadi N, Mohanty M, Mohanty CR (2017) Studies on adsorption behavior of an industrial waste for removal of chromium from aqueous solution. S Afr J Chem Eng 23:132–138. https://doi.org/10.1016/j.sajce.2017.05.002
Qin Q, Wang Q, Fu D, Ma J (2011) An efficient approach for Pb(II) and Cd(II) removal using manganese dioxide formed in situ. Chem Eng J 172(1):68–74. https://doi.org/10.1016/j.cej.2011.05.066
Qiu H, Liang C, Yu J, Zhang Q, Song M, Chen F (2017) Preferable phosphate sequestration by nano-La (III) (Hydr) oxides modified wheat straw with excellent properties in regeneration. Chem Eng J 315:345–354. https://doi.org/10.1016/j.cej.2017.01.043
Qurie M, Khamis M, Manassra A, Ibrahim A, Shlomo N, Scrano L, Bufo SA, Rafik K (2013) Removal of Cr(VI) from aqueous environments using micelle-clay adsorption. Scientific World Journal. https://doi.org/10.1155/2013/942703
Ravindhranath K, Ramamoorty M (2017) Nickel based nano particles as adsorbents in water purification methods-a review. Oriental J Chem 33(4):1603. https://doi.org/10.13005/ojc/330403
Rezak N, Bahmani A, Bettahar N (2021) Adsorptive removal of P(V) and Cr (VI) by calcined Zn-Al-Fe ternary LDHs. Water Sci Technol 83(10):2504–2517. https://doi.org/10.2166/wst.2021.123
Ruzhitskaya O, Gogina E (2017) Methods for removing of phosphates from wastewater. MATEC Web of Conferences 106:07006. https://doi.org/10.1051/matecconf/201710607006
Salah Abde WM, Abdel Monem AS, Mohamed A, Mohamed MA (2013) Adsorption Studies on the removal of hexavalent chromium contaminated wastewater using activated carbon and bentonite. Asian J Chem 25(15):8245–8252
Sereshti H, ZamiriAfsharian E, EsmaeiliBidhendi M, RashidiNodeh H, AfzalKamboh M, Yilmaz M (2020) Removal of phosphate and nitrate ions aqueous using strontium magnetic graphene oxide nanocomposite: isotherms, kinetics, and thermodynamics studies. Environ Prog Sustainable Energy 39(2):13332. https://doi.org/10.1002/ep.13332
Shokrollahi M et al (2019) Application of ZnO nanostructures in ceramic and polymeric membranes for water and wastewater technologies. Chem Eng J 391:123475. https://doi.org/10.1016/j.cej.2019.123475
Shokrollahi M, Rezakazemi M, Younas M (2020) Producing water from saline streams using membrane distillation: modeling and optimization using CFD and design expert. Int J Energy Res 44(11):8841–8853. https://doi.org/10.1002/er.5578
Sneha Latha P, Biftu WK, Suneetha M, Ravindhranath K (2021) De-fluoridation studies: using Lanthanum-alginate-beads impregnated with green synthesized nSiO2 and active carbon of Terminalia ivorensis plant as an effective adsorbent. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-021-03575-w
Stella C, Soundararajan N, Ramachandran K (2014) Structural, optical, dielectric and magnetic properties of Mn1-xCoxO2 nanowires. Superlattices Microstruct 71:203–210. https://doi.org/10.1016/j.spmi.2014.03.044
Suneetha M, Ravindhranath K (2015) Adsorption of nitrite ions onto the bio-sorbents derived from Azadirachta indica plant. Asian J Wate Environ Pollut 14(2):71–79
Suneetha M, Ravindhranath K (2018) Removal of nitrites from waste waters using adsorbents derived from Phyllanthus neruri plant. Indian J Chem Technol 25(4):345–352
Suneetha M, Syama S, Rabindranath K (2015) De-fluoridation of waters using low-cost HNO3 activated carbon derived from stems of Senna occidentalis plant. Int J Environ Technol Manage 18(5/6):420–447. https://doi.org/10.1504/IJETM.2015.073079
Temkin MJ, Pyzhev V (1940) Recent modifications to Langmuir isotherms. Acta Physiochim USSR 12:217–222
Trinh VT, Nguyen TMP, Hoang HT, Van LP, Nguyen TV, Ha LT, Xuan HV, Pham TT, Nguyen TN, Quang NV, Nguyen (2020) Phosphate adsorption by silver nanoparticles-loaded activated carbon derived from tea residue. Sci Rep 10(1). https://doi.org/10.1038/s41598-020-60542-0
Trivedy R K (1995) Pollution management in industries, Environmental Publications. 2nd Ed Karad India.
US Department of Health and Human Services Profile for Chromate (1991) Public Health Service Agency for toxic substances and diseases. Washington DC.
Wang H, Xu X, Ren Z, Gao B (2016) Removal of phosphate and chromium (VI) from liquids by an amine-crosslinked nano-Fe3O4biosorbent derived from corn straw. RSC Adv 6:47237–47248. https://doi.org/10.1039/C6RA06801D
Wang J, Liang Y, ** Q, Hou J, Liu B, Li X, Chen X, Hayat W, Ahmed T, Wang A (2017) Simultaneous removal of graphene oxide and chromium(VI) on the rare earth doped titanium dioxide coated carbon sphere composites. ACS Sustainable Chem Eng 56:5550–5561. https://doi.org/10.1021/acssuschemeng.7b00957
Wang Y, **e X, Chen X, Huang C, Yang S (2020) Biochar-loaded Ce3+-enriched ultra-ne ceria nanoparticles for phosphate adsorption. J Hazard Mater 396:122626
Wang Z, Fang W, **ng M, Wu D (2017) A bench-scale study on the removal and recovery of phosphate by hydrous zirconia coated magnetite nanoparticles. J Magn Mater 424:213–220. https://doi.org/10.1016/j.jmmm.2016.10.067
Wu FC, Tseng RL, Juang RS (2009) Characteristics of Elovich equation used for the analysis of adsorption kinetics in dye-chitosan systems. Chem Eng J 150(2–3):366–373. https://doi.org/10.1016/j.cej.2009.01.014
Yadesa W, Taddesse AM, Kibret K, Dechassa N (2018) Synthesis and characterization of fe-al-mn nanocomposite sorbent for phosphate sorption-desorption study. Bull Chem Soc Ethiop 32(3):421–436. https://doi.org/10.4314/bcse.v32i3.2
Yao DZR, Sun C, Zheng Y, Cheng Liu C (2021) Effluents by recoverable La(OH)3/foamed nickel adsorbent. ACS Omega 6:5399–5407. https://doi.org/10.1021/acsomega.0c05489
Yu** T, Jonas W, Harald K, Martin W, Peter S, Kramer RC (2015) Optically probing Al—O and O—H vibrations to characterize water adsorption and surface reconstruction on α-alumina: an experimental and theoretical study. The J of Chemical Physics 142(054704):1–12. https://doi.org/10.1063/1.4906346
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pala, S.L., Biftu, W.K., Mekala, S. et al. Adsorptive removal of toxic chromate and phosphate ions from polluted water using green-synthesized nanometal (Mn-Al–Fe) oxide. Biomass Conv. Bioref. 13, 16835–16853 (2023). https://doi.org/10.1007/s13399-021-02293-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-02293-0