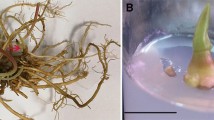
Abstract
In the present study, an efficient in vitro propagation protocol has been developed from clove explants of Allium sativum L., one of the oldest vegetable and medicinal plant used worldwide. Garlic is propagated vegetatively as cross-fertilization is strictly precluded due to sterile flowers. Due to a low rate of multiplication, limited genetic improvement possibility and increased germplasm degradation, plant tissue culture becomes an efficient and preferred tool for quality and rapid propagation of garlic. Here, the clove explants were cultured on Murashige and Skoog basal medium amended with different concentrations of Plant Growth Regulators (PGRs) namely 2,4-dichlorophenoxy acetic acid (2,4-D), 6-benzyl amino purine (BAP), and 1-naphthalene acetic acid (NAA). Within 2 weeks of inoculation, white compact callus was formed, maximum callus induction frequency (85.99%) was on 1.5 mg l−1 2, 4-D added MS medium. Induced callus transformed into an embryogenic callus on 2, 4-D and BAP amended MS medium with highest embryogenic frequency (77.7%) was noted on 0.25 mg l−1 2, 4-D and 1.0 mg l−1 BAP added medium. Embryogenic callus differentiated into progressive stages of somatic embryos starting from globular, scutellar, and finally to coleoptilar stage of the embryo. Histological and scanning electron microscopic study of embryogenic callus was conducted, showing different stages of embryos, their origin and development, re-confirming somatic embryogenesis incidence in A. sativum. Green and mature somatic embryos were germinated and converted into plantlets on 0.5 mg l−1 BAP amended MS medium. The in vitro regenerated plants were cultured separately in IBA and NAA supplemented media for root induction. The MS medium amended with 1.0 mg l−1 IBA proved to be the best PGR treatment in inducing roots. The rooted plants were acclimatized and transferred ex vitro with about 87% survival rate. Cytological and flow cytometric analyses were performed to assess the genetic stability of in vitro regenerated plants. Cytological studies of in vitro regenerated plants showed 2n = 16 chromosome number and did not reveal any numerical variation in chromosomes. Flow cytometry was employed to measure the 2C DNA content of somatic embryo regenerated A. sativum plants and compared with in vivo grown garlic. The histogram peaks of relative 2C DNA content of in vitro regenerated plantlets were similar to the corresponding 2C DNA peak of in vivo grown plants. Flow cytometric 2C DNA content of embryo regenerated and field-grown A. sativum plants were the same, i.e., 33.45 pg and 33.56 pg, respectively, confirming genetic similarity. In conclusion, the present cytological and flow cytometric study suggest that the in vitro culture conditions are quite safe, did not encourage genetic alterations, and regenerants were “true to type.”





Similar content being viewed by others
References
Alan AR, Zeng H, Assani A, Shi WL, McRae HE, Murch SJ, Saxena PK (2007) Assessment of genetic stability of the germplasm lines of medicinal plant Scutellaria baicalensis Georgi (Huang-qin) in long-term, in vitro maintained cultures. Plant Cell Rep 26:1345–1355
Ali M, Mujib A, Tonk D, Zafar N (2016) Plant regeneration through somatic embryogenesis and genome size analysis of Coriandrum sativum L. Protoplasma 254:343–352
Alizadeh M, Krishna H, Eftekhari M, Modareskia M, Modareskia M (2015) Assessment of clonal fidelity in micropropagated horticultural plants. J Chem Pharm Res 7(12):977–990
Altamura MM, Della RF, Fattorini L et al (2016) Recent advances on genetic and physiological bases of in vitro somatic embryo formation. In: Germana MA, Lambardi M (eds) In vitro embryogenesis in higher plants. Springer, New York, pp 47–85. https://doi.org/10.1007/978-1-4939-3061-6_3
Bairu MW, Fennell CW, Van Staden J (2006) The effect of plant growth regulators on somaclonal variation in Cavendish banana (Musa AAA cv. ‘Zelig’). Sci Hortic 108:347–351
Bairu MW, Aremu AO, Van Staden J (2011) Somaclonal variation in plants: causes and detection methods. Plant Growth Regul 63:147–173
Bennett MD, Leitch IJ (2011) Nuclear DNA amounts in angiosperms: targets, trends and tomorrow. Ann Bot 107(3):467–590
Block E (2010) Garlic and other alliums. The Lore and the Science. RSC Publishing, UK, pp 1–434
Borlinghaus J, Albrecht F, Gruhlke MCH, Nwachukwu ID, Slusarenko AJ (2014) Allicin: chemistry and biological properties. Molecules 19:12591–12618
Bradley JM, Organ CL, Lefer DJ (2016) Garlic-derived organic polysulfides and myocardial protection. J Nutr 146:403–409
Bystrická J, Kovarovič J, Lenková M, Horváthová J, Končeková L, Halmová D, Lidiková A (2019) The content of polyphenols, antioxidant activity and macroelements in selected garlic varieties. J Microbiol Biotechnol Food Sci 8:738–740
Camargo AB, Manucha W (2017) Potential protective role of nitric oxide and Hsp70 linked to functional foods in the atherosclerosis. Clin Investig Arterioscler 29:36–45
Cavagnaro PF, Camargo A, Galmarini CR, Simon PW (2007) Effect of cooking on garlic (Allium sativum L), antiplatelet activity and thiosulfinates content. J Agri Food Chem 55:1280–1288
Clarindo WR, Carvalho CR, Arauojo FS, Abreu IS, Otoni WC (2008) Recovering polyploid papaya in vitro regenerants as screened by flow cytometry. Plant Cell Tissue Organ Cult 92:207–214
Das A, Kesari V, Rangan L (2013) Micropropagation and cytogenetic assessment of Zingiber species of Northeast India. Biotechnol 3:471–479
Delporte F, Pretova A, Jardin P, Watillon B (2014) Morpho-histology and genotype dependence of in vitro morphogenesis in mature embryo cultures of wheat. Protoplasma 251:1455–1470
Dipti S, Fatima S, Mujib A (2014) Morphological anomalies in somatic embryo structure in Catharanthus roseus: improving embryo germination by amending plant growth regulators, activated charcoal and sucrose level. British Biotechnol J 4(1):10–20
Elhiti M, Stasolla C, Wang AM (2013) Molecular regulation of plant somatic embryogenesis. In Vitro Cell Dev Biol - Plant 49:631–642
Fan B, He R, Shang Y, Xu L, Wang N, Gao H, Liu X, Wang Z (2017) System construction of virus-free and rapid-propagation technology of Baodi garlic (Allium sativum L). Scientia Horticulturae 225:498–504
Fehér A (2015) Somatic embryogenesis: stress-induced remodeling of plant cell fate. Biochim Biophys Acta 1849:385–402
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228
Galbraith DW (2009) Simultaneous flow cytometric quantification of plant nuclear DNA contents over the full range of described angiosperm 2C values. Cytom Part A 75:692–698
Gimenez C, de Garcı’a E, de Enrech NX, Blanca I (2001) Somaclonal variation in banana: cytogenetic and molecular characterization of the somaclonal variant CIEN BTA-03. In Vitro Cell Dev Biol - Plant 37:217–222
Gimenez MD, Yañezsantos AM, Paz RC, Quiroga MP, Marfil CF, Conci VC, Garcíalampasona SC (2016) Assessment of genetic and epigenetic changes in virus free garlic (Allium sativum L.) plants obtained by meristem culture followed by in vitro propagation. Plant Cell Rep 35:129
Giorgetti L, Ruffini-Castiglione M, Turrini A, Martini G, NutiRonchi V, Geri C (2011) Cytogenetic and histological approach for early detection of “mantled” somaclonal variants of oil palm regenerated by somatic embryogenesis: first results on the characterization of regeneration system. Caryologia 64:223–234
Gliwicka M, Nowak K, Balazadeh S, Mueller-Roeber B, Gaj MD (2013) Extensive modulation of the transcription factor transcriptome during somatic embryogenesis in Arabidopsis thaliana. PLoS One 8(7):e69261
Guercio V, Galeone C, Turati F, La Vecchia C (2014) Gastric cancer and allium vegetable intake: a critical review of the experimental and epidemiologic evidence. Nutr Cancer 66:757–773
Gulzar B, Mujib A, Rajam MV, Frukh A, Zafar N (2019) Identification of somatic embryogenesis (SE) related proteins through label-free shotgun proteomic method and cellular role in Catharanthus roseus (L.) G. Don. Plant Cell Tissue Organ Cult 137(2):225–237
Hofmann N (2014) Getting to the root of regeneration: adventitious rooting and callus formation. Plant Cell 26:845
Horstman A, Bemer M, Boutilier K (2017) A transcriptional view on somatic embryogenesis. Regeneration (Oxf) 4:201–216
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451
Ikeuchi M, Favero DS, Sakamoto Y, Iwase A, Coleman D, Rymen B, Sugimoto K (2019) Molecular mechanisms of plant regeneration. Annu Rev Plant Biol 70:377–406
Johansen DA (1940) Botanical Microtechnique, vol 523. McCraw-Hill Book, New York
Kim YJ, Lee OR, Kim KT, Yang DC (2012) High frequency of plant regeneration through cyclic secondary somatic embryogenesis in Panax ginseng. J Ginseng Res 36(4):442–448
Krishna H, Alizadeh M, Singh D, Singh U, Chauhan N, Eftekhari M, Sadh RK (2016) Somaclonal variations and their applications in horticultural crops improvement. 3. Biotech 6:54
Kumar V, Van Staden J (2017) New insights into plant somatic embryogenesis: an epigenetic view. Acta Physiol Plant 39:194
Loureiro J, Capelo A, Brito G, Rodriguez E, Silva S, Pinto G, Santos C (2007) Micropropagation of Juniperus phoenicea from adult plant explants and analysis of ploidy stability using flow cytometry. Biol Plant 51:7–14
Luciani G, Mary A, Pellegrini C, Curvetto N (2006) Effects of explants and growth regulators in garlic callus formation and plant regeneration. Plant Cell Tissue Organ Cult 87:139–143
Makowczynska J, Andrzejewska-Golec E, Sliwinska E (2008) Nuclear DNA content in different plant material of Plantago asiatica L. cultured in vitro. Plant Cell Tissue Organ Cult 94:65–71
Mallón R, Rodríguez-Oubiña J, González ML (2010) In vitro propagation of the endangered plant Centaurea ultreiae: assessment of genetic stability by cytological studies, flow cytometry and RAPD analysis. Plant Cell Tissue Organ Cult 101:31–39
Meriga B, Mopuri R, Muralikrishna T (2012) Insecticidal, antimicrobial and antioxidant activities of bulb extracts of Allium sativum. Asian Pac J Trop Med 5:391–395
Mujib A, Banerjee S, Ghosh PD (2007) Callus induction, somatic embryogenesis and chromosomal instability in tissue culture raised Hippeastrum (Hippeastrum hybridum cv. United Nations). Prop Ornamental Plants 7:169–174
Mujib A, Banerjee S, Ghosh PD (2013a) Tissue culture induced variability in some horticultural important ornamentals: chromosomal and molecular basis – a review. Biotechnology 12(6):213–224
Mujib A, Banerjee S, Maqsood M, Ghosh PD (2013b) Somatic embryogenesis of some member ornamental genera of Amaryllidaceae and allied families: the similarities and differences. Open Hortic J 6:9–18
Mujib A, Ali M, Tonk D, Zafar N (2017) Nuclear 2C DNA and genome size analysis in somatic embryo regenerated gladiolus plants using flow cytometry. Adv Hortic Sci 31(3):165–174
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497
Nasim SA, Mujib A, Kapoor R, Fatima S, Aslam J (2010) Somatic embryogenesis in Allium sativum L. (cv. Yamuna Safed 3): improving embryo maturation and germination with PGRs and carbohydrates. An Biol 32:1–9
Nicastro HL, Ross SA, Milner JA (2015) Garlic and onions: their cancer prevention properties. Cancer Prev Res 8:181–189
Nic-Can GI, Loyola-Vargas VM (2016) The role of the auxins during somatic embryogenesis. Fundamental Aspects and Applications, Somatic Embryogenesis, pp 171–181. https://doi.org/10.1007/978-3-319-33705-0_10
Pinto G, Loureiro J, Lopes T, Santos C (2004) Analysis of the genetic stability of Eucalyptus globules Labill. Somatic embryos by flow cytometry. Theor Appl Genet 109:580–587
Pulianmackal AJ, Kareem AVK, Durgaprasad K, Trivedi ZB, Prasad K (2014) Competence and regulatory interactions during regeneration in plants. Front Plant Sci 5:142. https://doi.org/10.3389/fpls.2014.00142
Raji MR, Lotfi M, Tohidfar M, Zahedi B, Carra A, Abbate L, Carimi F (2017) Somatic embryogenesis of muskmelon (Cucumis melo L.) and genetic stability assessment of regenerants using flow cytometry and ISSR markers. Protoplasma 255:873–883
Ramakrishnan M, Ceasar SA, Duraipandiyan V (2013) Efficacious somatic embryogenesis and fertile plant recovery from shoot apex explants of onion (Allium cepa. L.). In Vitro Cell Dev Biol- Plant 49:285
Santos-Mendoza M, Dubreucq B, Baud S, Parcy F, Caboche M, Lepiniec L (2008) Deciphering gene regulatory networks that control seed development and maturation in Arabidopsis. Plant J 54:608–620
Shemesh ME, Winiarczyk K, Błaszczyk L, Kosmala A, Rabinowitch HD, Kamenetsky R (2013) Male gametogenesis and sterility in garlic (Allium sativum L.): barriers on the way to fertilization and seed production. Planta 237:103–120
Sikand G, Kris-Etherton P, Boulos NM (2015) Impact of functional foods on prevention of cardiovascular disease and diabetes. Curr Cardiol Rep 17:39
Yang JL, Niu YD, Yang CP, Liu GF, Li CH (2011) Induction of somatic embryogenesis from female flower buds of elite Schisandra chinensis. Plant Cell Tissue Organ Cult 106:391–399
Zafar N, Mujib A, Ali M, Tonk D, Gulzar B, Malik M, Syeed R, Mamgain J (2019) Genome size analysis of field grown and tissue culture regenerated Rauvolfia serpentina (L) by flow cytometry: histology and scanning electron microscopic study for in vitro morphogenesis. Indust Crops Products 128:545–555
Acknowledgments
We are thankful to the Department of Botany, Jamia Hamdard for providing us the lab facilities and to BD-JH FACS Academy Jamia Hamdard, New Delhi for flow cytometry facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by: Barbara Naganowska
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 309 kb)
Rights and permissions
About this article
Cite this article
Malik, M.Q., Mujib, A., Gulzar, B. et al. Genome size analysis of field grown and somatic embryo regenerated plants in Allium sativum L.. J Appl Genetics 61, 25–35 (2020). https://doi.org/10.1007/s13353-019-00536-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-019-00536-5