Abstract
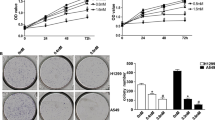
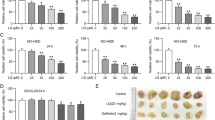
Small cell lung cancer (SCLC) accounts for 12 to 16 % of lung neoplasms and has a high rate of metastasis. The present study demonstrates the antiproliferative effect of retinoic acid amide in vitro and in vivo against human lung cancer cells. The results from MTT assay showed a significant growth inhibition of six tested lung cancer cell lines and inhibition of clonogenic growth at 30 μM. Retinoic acid amide also leads to G2/M-phase cell cycle arrest and apoptosis of lung cancer cells. It caused inhibition of JAK2, STAT3, and STAT5, increased the level of p21WAF1, and decreased cyclin A, cyclin B1, and Bcl-XL expression. Retinoic acid amide exhibited a synergistic effect on antiproliferative effects of methotrexate in lung cancer cells. In lung tumor xenografts, the tumor volume was decreased by 82.4 % compared to controls. The retinoic acid amide-treated tumors showed inhibition of JAK2/STAT3 activation and Bcl-XL expression. There was also increase in expression of caspase-3 and caspase-9 in tumors on treatment with retinoic acid amide. Thus, retinoic acid amide exhibits promising antiproliferative effects against human lung cancer cells in vitro and in vivo and enhances the antiproliferative effect of methotrexate.






Similar content being viewed by others
References
Jackman DM, Johnson BE. Small-cell lung cancer. Lancet. 2005;366:1385–96.
Datto MB, Li Y, Panus JF, Howe DJ, **ong Y, Wang XF. Transforming growth factor h induces the cyclin-dependent kinase inhibitor p21 through a p53-independent mechanism. Proc Natl Acad Sci U S A. 1995;92:5545–9.
Alpan RS, Pardee AB. p21WAF1/CIP1/SDI1 is elevated through a p53-independent pathway by mimosine. Cell Growth Differ. 1996;7:893–901.
Zeng YX, El-Deiry WS. Regulation of p21WAF1/CIP1 expression by p53-independent pathways. Oncogene. 1996;12:1557–64.
Steinman RA, Hoffman B, Iro A, Guillouf C, Liebermann DA, El-Houseini ME. Induction of p21 (WAF-1/CIP1) during differentiation. Oncogene. 1994;9:3389–96.
Ferraldeschi R, Baka S, Jyoti B, et al. Modern management of small-cell lung cancer. Drugs. 2007;67:2135–52.
Simon GR, Turrisi A, American College of Chest Physicians. Management of small cell lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest. 2007;132:324S–39S.
Planchard D, Le Péchoux C. Small cell lung cancer: new clinical recommendations and current status of biomarker assessment. Eur J Cancer. 2011;47:S272–83.
Darnell Jr JE. STATs and gene regulation. Science. 1997;277:1630–5.
Reich NC, Liu L. Tracking STAT nuclear traffic. Nat Rev Immunol. 2006;6:602–12.
Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C, et al. Stat3 as an oncogene. Cell. 1999;98:295–303.
Niu G, Wright KL, Huang M, Song L, Haura E, Turkson J, et al. Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene. 2002;21:2000–8.
Deng J, Grande F, Neamati N. Small molecule inhibitors of Stat3 signaling pathway. Curr Cancer Drug Targets. 2007;7:91–107.
Zhang XF, Wang JF, Matczak E, Proper JA, Groopman JE. Janus kinase 2 is involved in stromal cell-derived factor-1 alpha-induced tyrosine phosphorylation of focal adhesion proteins and migration of hematopoietic progenitor cells. Blood. 2001;97:3342–8.
Soriano SF, Hernanz-Falcon P, Rodriguez-Frade JM, De Ana AM, Garzon R, Carvalho-Pinto C, et al. Functional inactivation of CXC chemokine receptor 4-mediated responses through SOCS3 up-regulation. J Exp Med. 2002;196:311–21.
Orfanos CE, Ehlert R, Gollnick H. The retinoids. A review of their clinical pharmacology and therapeutic use. Drugs. 1987;34:459–503.
Kligman AM. The growing importance of topical retinoids in clinical dermatology: a retrospective and prospective analysis. J Am Acad Dermatol. 1998;39:S2–7.
Chandraratna RA. Current research and future developments in retinoids: oral and topical agents. Cutis. 1998;61:40–5.
Zheng Y, Kramer PM, Lubet RA, Steele VE, Kelloff GJ, Pereira MA. Effect of retinoids on AOM-induced colon cancer in rats: modulation of cell proliferation, apoptosis and aberrant crypt foci. Carcinogenesis. 1999;20:255–60.
Liang JY, Fontana JA, Rao JN, Ordonez JV, Dawson MI, Shroot B, et al. Synthetic retinoid CD437 induces S-phase arrest and apoptosis in human prostate cancer cells LNCaP and PC-3. Prostate. 1999;38:228–36.
Weber E, Ravi RK, Knudsen ES, Williams JR, Dillehay LE, Nelkin BD, et al. Retinoic acid-mediated growth inhibition of small cell lung cancer cells is associated with reduced myc and increased p27Kip1 expression. Int J Cancer. 1999;80:935–43.
Mologni L, Ponzanelli I, Bresciani F, Sardiello G, Bergamaschi D, Gianni M, et al. The novel synthetic retinoid 6-[3-adamantyl-4-hydroxyphenyl]-2-naphtalene carboxylic acid (CD437) causes apoptosis in acute promyelocytic leukemia cells through rapid activation of caspases. Blood. 1999;93:1045–61.
Irving H, Lovat PE, Hewson QC, Malcolm AJ, Pearson AD, Redfern CP. Retinoid induced differentiation of neuroblastoma: comparison between LG69, an RXR-selective analogue and 9-cis retinoic acid. Eur J Cancer. 1998;34:111–7.
Dirks PB, Patel K, Hubbard SL, Ackerley C, Hamel PA, Rutka JT. Retinoic acid and the cyclin dependent kinase inhibitors synergistically alter proliferation and morphology of U343 astrocytoma cells. Oncogene. 1997;15:2037–48.
Yung WK, Kyritsis AP, Gleason MJ, Levin VA. Treatment of recurrent malignant gliomas with high-dose 13-cis-retinoic acid. Clin Cancer Res. 1996;2:1931–5.
Defer GL, Adle-Biassette H, Ricolfi F, Martin L, Authier FJ, Chomienne C, et al. All-trans retinoic acid in relapsing malignant gliomas: clinical and radiological stabilization associated with the appearance of intratumoral calcifications. J Neurooncol. 1997;34:169–77.
Kaba SE, Kyritsis AP, Conrad C, Gleason MJ, Newman R, Levin VA, et al. The treatment of recurrent cerebral gliomas with all-trans-retinoic acid (tretinoin). J Neurooncol. 1997;34:145–51.
Bouterfa H, Picht T, Keb D, Herbold C, Noll E, Black PM, et al. Retinoids inhibit human glioma cell proliferation and migration in primary cell cultures but not in established cell lines. Neurosurgery. 2000;46:419–30.
Rotan R. Retinoids as modulators of tumor cell invasion and metastasis. Semin Cancer Biol. 1991;2:197–208.
Li WX. Canonical and non-canonical JAK-STAT signaling. Trends Cell Biol. 2008;18:545–51.
Toyonaga T, Nakano K, Nagano M, et al. Blockade of constitutively activated Janus kinase/signal transducer and activator of transcription-3 pathway inhibits growth of human pancreatic cancer. Cancer Lett. 2003;201:107–16.
Sahu RP, Srivastava SK. The role of STAT-3 in the induction of apoptosis in pancreatic cancer cells by benzyl isothiocyanate. J Natl Cancer Inst. 2009;101:176–93.
Tan SH, Nevalainen MT. Signal transducer and activator of transcription 5A/B in prostate and breast cancers. Endocrinol Relat Cancer. 2008;15:367–90.
Lewis RS, Ward AC. Stat5 as a diagnostic marker for leukemia. Expert Rev Mol Diagn. 2008;8:73–82.
Adnane J, Bizouarn FA, Qian Y, Hamilton AD, Sebti SM. p21(WAF1/CIP1) is upregulated by the geranylgeranyltransferase I inhibitor GGTI-298 through a transforming growth factor h- and Sp1-responsive element: involvement of the small GTPaseRhoA. Mol Cell Biol. 1998;18:6962–70.
Yamaguchi Y, Watanabe H, Yrdiran S, et al. Detection of mutations of p53 tumor suppressor gene in pancreatic juice and its application to diagnosis of patients with pancreatic cancer: comparison with K-ras mutation. Clin Cancer Res. 1999;5:1147–53.
Bai J, Sui J, Demirjian A, Vollmer Jr CM, Marasco W, Callery MP. Predominant Bcl-XL knockdown disables antiapoptotic mechanisms: tumor necrosis factor-related apoptosis-inducing ligand-based triple chemotherapy overcomes chemoresistance in pancreatic cancer cells in vitro. Cancer Res. 2005;65:2344–52.
Ghaneh P, Kawesha A, Evans JD, Neoptolemos JP. Molecular prognostic markers in pancreatic cancer. J Hepatobiliary Pancreat Surg. 2002;9:1–11.
Conflicts of interest
None
Author contributions
Hong-**ng Li and Wei Zhao designed the study. Yan Shi, Ya-Na Li, Lian-Shuang Zhang, and Hong-Qin Zhang wrote the manuscript. Dong Wang and Hong-**ng Li performed the immunohistochemistry and the related statistical analysis. All authors approved the final version of the manuscript.
Funding support
The project was financially supported by Shandong Province Natural Science Foundation (ZR2013HM047and 2011HL063) and Shandong Province Higher Education Technology Plan (J10LF57).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, HX., Zhao, W., Shi, Y. et al. Retinoic acid amide inhibits JAK/STAT pathway in lung cancer which leads to apoptosis. Tumor Biol. 36, 8671–8678 (2015). https://doi.org/10.1007/s13277-015-3534-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3534-8