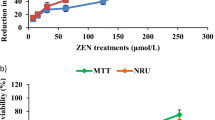
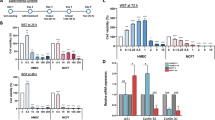
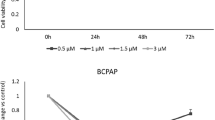
Abstract
Benzophenone-2 (BP2) is used as a UV screen in many cosmetics of daily use, which regularly come in contact with the human skin. It is well known that BP2 is an endocrine-disrupting chemical (EDC) that disrupts the thyroid hormone axis via the inhibition of thyroid peroxidase. However, the molecular mechanism of BP2-induced cellular toxicity remains unclear. We undertook a genome-wide analysis of transcriptional responses of human thyroid follicular carcinoma FTC-238 cells to non-toxic doses of BP2 (1 and 50 μM). Whole human genome microarrays detected a total of 4,495 genes that exhibited altered expression after exposure to both non-toxic doses of BP2 compared with a vehicle control, DMSO. We found that 534 and 4,142 genes showed different expression levels following treatment with the low and high nontoxic doses of BP2, respectively. Thus, low doses of BP2 can induce various changes in gene expression. The BP2 responsive genes were involved in glycolysis, translation, apoptosis, cell proliferation, generation of precursor metabolites and energy, and microtubulebased processes. Commonly expressed genes by 1 μM and 50 μM of BP2 treatments were associated with glycolysis components, including GAPDH, ENO1, PGK1, TROVE2, LDHA, ALDOA, and PKM2. The downregulated GAPDH, and ENO1 are typical glycolytic transcripts in glycolysis. The disruption of glycolysis including nutrients-replete conditions influences on cell cycle arrest or apoptosis. The possible roles of the responsive genes and their related pathways in BP2-induced cellular responses are discussed. This study helps to understand how cells respond to BP2 exposure via transcriptional regulation.
Similar content being viewed by others
References
Jarry, H., Christoffel, J., Rimoldi, G., Koch, L. & Wuttke, W. Multi-organic endocrine disrupting activity of the UV screen benzophenone 2 (BP2) in ovariectomized adult rats after 5 days treatment. Toxicology 205:87–93 (2004).
Jeon, H. K., Chung, Y. & Ryu, J. C. Simultaneous determination of benzophenone-type UV filters in water and soil by gas chromatography-mass spectrometry. J Chromatogr A 1131:192–202 (2006).
Schlecht, C., Klammer, H., Frauendorf, H., Wuttke, W. & Jarry, H. Pharmacokinetics and metabolism of benzophenone 2 in the rat. Toxicology 245:11–17 (2008).
Poiger, T., Buser, H. R., Balmer, M. E., Bergqvist, P. A. & Müller, M. D. Occurrence of UV filter compounds from sunscreens in surface waters: regional mass balance in two Swiss lakes. Chemosphere 55:951–963 (2004).
Schmutzler, C. et al. The ultraviolet filter benzophenone 2 interferes with the thyroid hormone axis in rats and is a potent in vitro inhibitor of human recombinant thyroid peroxidase. Endocrinology 148:2835–2844 (2007).
Schlecht, C., Klammer, H., Jarry, H. & Wuttke, W. Effects of estradiol, benzophenone-2 and benzophenone-3 on the expression pattern of the estrogen receptors (ER) alpha and beta, the estrogen receptor-related receptor 1 (ERR1) and the aryl hydrocarbon receptor (AhR) in adult ovariectomized rats. Toxicology 205: 123–130 (2004).
Seidlová-Wuttke, D., Jarry, H. & Wuttke, W. Pure estrogenic effect of benzophenone-2 (BP2) but not of bisphenol A (BPA) and dibutylphtalate (DBP) in uterus, vagina and bone. Toxicology 205:103–112 (2004).
Miller, D., Wheals, B. B., Beresford, N. & Sumpter, J. P. Estrogenic activity of phenolic additives determined by an in vitro yeast bioassay. Environ Health Persp 109:133–138 (2001).
Yamasaki, K., Takeyoshi, M., Yakabe, Y., Sawaki, M. & Takatsuki, M. Comparison of the reporter gene assay for ER-alpha antagonists with the immature rat uterotrophic assay of 10 chemicals. Toxicol Lett 142: 119–131 (2003).
Köhrle, J. Selenium and the control of thyroid hormone metabolism. Thyroid 15:841–853 (2005).
Boas, M., Feldt-Rasmussen, U., Skakkebaek, N. E. & Main, K. M. Environmental chemicals and thyroid function. Eur J Endocrinol 154:599–611 (2006).
Jahnke, G. D., Choksi, N. Y., Moore, J. A. & Shelby, M. D. Thyroid toxicants: assessing reproductive health effects. Environ Health Persp 112:363–368 (2004).
Zhou, T., Taylor, M. M., De Vito, M. J. & Crofton, K. M. Developmental exposure to brominated diphenyl ethers results in thyroid hormone disruption. Toxicol Sci 66:105–116 (2002).
Jannini, E. A., Ulisse, S. & D’Armiento, M. Thyroid hormone and male gonadal function. Endocr Rev 16: 443–459 (1995).
Song, M. K., Kim, Y. J., Song, M. & Ryu, J. C. Gene expression analysis identifies potential biomarkers of phenanthrene in human hepatocytes (HepG2). Toxicol Environ Health Sci 3:30–38 (2011).
Sarma, N. S., Song, M., Kim, Y. J. & Ryu, J. C. Genome-wide identification of ethylbenzene and trichloroethylene-regulated genes in human promyelocytic leukemia HL-60 cells. BioChip J 5:19–26 (2011).
Yeo, M. K. & Kim, H. E. Gene expression in zebrafish embryos following exposure to TiO2 nanoparticles. Mol Cell Toxicol 6:97–104 (2010).
Matsumoto, H., Adachi, S. & Suzuki, Y. Estrogenic activity of ultraviolet absorbers and the related compounds. Yakugaku Zasshi 125:643–652 (2005).
Cettour-Rose, P. et al. Hypothyroidism in rats decreases peripheral glucose utilisation, a defect partially corrected by central leptin infusion. Diabetologia 48:624–633 (2005).
Fanelli, A., Grollman, E. F., Wang, D. & Philp, N. J. MCT1 and its accessory protein CD147 are differentially regulated by TSH in rat thyroid cells. Am J Physiol Endoc M 285:E1223–E1229 (2003).
Moeller, L. C., Cao, X., Dumitrescu, A. M., Seo, H. & Refetoff, S. Thyroid hormone mediated changes in gene expression can be initiated by cytosolic action of the thyroid hormone receptor beta through the phosphatidylinositol 3-kinase pathway. Nuclear Receptor Signaling 4:e020 (2006).
Ness, G. C. & Pendleton, L. C. Thyroid hormone increases glyceraldehyde 3-phosphate dehydrogenase gene expression in rat liver. FEBS Lett 288:21–22 (1991).
Moreno-Sánchez, R., Rodríguez-Enríquez, S., Saavedra, E., Marín-Hernández, A. & Gallardo-Pérez, J. C. The bioenergetics of cancer: is glycolysis the main ATP supplier in all tumor cells? Biofactors 35:209–225 (2009).
Mason, E. F. et al. Aerobic glycolysis suppresses p53 activity to provide selective protection from apoptosis upon loss of growth signals or inhibition of BCR-Abl. Cancer Res 70:8066–8076 (2010).
Bohnsack, B. L. & Hirschi, K. K. Nutrient regulation of cell cycle progression. Annu Rev Nutr 24:433–453 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, M., Kim, YJ. & Ryu, JC. Identification of genes induced by benzophenone-2 in human thyroid follicular FTC-238 cells. Mol. Cell. Toxicol. 7, 103–111 (2011). https://doi.org/10.1007/s13273-011-0014-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-011-0014-0