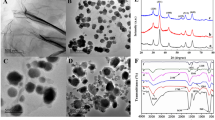
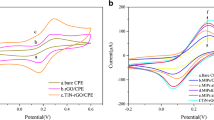
Abstract
In this research, the electrochemical sensor was fabricated based on glassy carbon electrode decorated by the reduced gold nanoparticles and molecularly imprinted polymer (MIP) on the electrode surface with methyl parathion (MP) as the template molecule. Four parameters, such as amount of gold nanoparticles, deposition time of gold nanoparticles, pH of acetate buffer solution, and extraction time of ethanol acidic solution, have been successfully determined by achieving the optimal sensor preparation. The MP in aqueous matrix can be quantitatively determined in the range of 0.05 to 15 μM with two linear equations at low and high ranges. In addition, the selectivity of the sensor as prepared toward the MP measurement is superior by comparing with similar structured compounds, such as imidacloprid and fenitrothion. This study also demonstrates that the deviation of current response from the MP in different matrix samples is negligible using MIP/Au/glassy carbon electrode sensor with recoveries ranged from 87.7 to 124.8%.

ᅟ
Graphical Abstract





Similar content being viewed by others
References
I. Bakas, A. Hayat, S. Piletsky, E. Piletska, M.M. Chehimi, T. Noguer, R. Rouillon, Talanta 130, 294 (2014)
H. Li, Z. Wang, B. Wu, X. Liu, Z. Xue, X. Lu, Electrochim. Acta 62, 319 (2012)
X. Tan, Q. Hu, J. Wu, X. Li, P. Li, H. Yu, X. Li, F. Lei, Sensor Actuat. B-Chem 220, 216 (2015)
G. Liu, W. Guo, Z. Yin, Biosens. Bioelectron. 53, 440 (2014)
M.Y. Wang, J.R. Huang, M. Wang, D.E. Zhang, J. Chen, Food Chem. 151, 191 (2014)
X. Xue, Q. Wei, D. Wu, H. Li, Y. Zhang, R. Feng, B. Du, Electrochim. Acta 116, 366 (2014)
J.G. Pacheco, M. Castro, S. Machado, M.F. Barroso, H.P.A. Nouws, C. Delerue-Matos, Sensors Actuat. B-Chem 215, 107 (2015)
J. Bai, X. Zhang, Y. Peng, X. Hong, Y. Liu, S. Jiang, Z. Gao, Sensor Actuat. B-Chem 238, 420 (2016)
B. Wang, O.K. Okoth, K. Yan, J. Zhang, Sensor Actuat. B-Chem 236, 294 (2016)
Y. Li, J. Liu, M. Liu, F. Yu, L. Zhang, H. Tang, B.C. Ye, L. Lai, Biosens. Bioelectron. 78, 308 (2016)
Y. Li, J. Liu, M. Liu, F. Yu, L. Zhang, H. Tang, B.C. Ye, L. Lai, Biosens. Bioelectron. 49, 199 (2013)
B. Zhang, J. Liu, X. Ma, P. Zuo, B.C. Ye, Y. Li, Biosens. Bioelectron. 80, 491 (2016)
K.K. Aswini, A.M. Vinu Mohan, V.M. Biju, Mat. Sci. Eng. C 65, 116 (2016)
Y. Li, J. Liu, M. Liu, F. Yu, L. Zhang, H. Tang, B.C. Ye, L. Lai, Electrochem. Commun. 64, 42 (2016)
B. Deiminiat, G.H. Rounaghi, M.H. Arbab-Zavar, Sensor Actuat. B-Chem 238, 651 (2017)
F. Tan, L. Cong, X. Li, Q. Zhao, H. Zhao, X. Quan, J. Chen, Sensor Actuat. B-Chem 233, 599 (2016)
Y. Yun, M. Pan, G. Fang, Y. Yang, T. Guo, J. Deng, B. Liu, S. Wang, Sensor Actuat. B-Chem. 238, 32 (2017)
G. Yang, F. Zhao, Electrochim. Acta 174, 33 (2015)
L. Zhao, B. Zeng, F. Zhao, Electrochim. Acta 146, 611 (2014)
G. Yang, F. Zhao, Biosens. Bioelectron. 64, 416 (2015)
C. Li, Z. Wang, G. Zhan, Colloid Surface B 82, 40 (2011)
L. Yang, F. Zhao, B. Zeng, Electrochim. Acta 210, 293 (2016)
H. Jiang, D. Jiang, J. Shao, X. Sun, Biosens. Bioelectron. 75, 411 (2016)
B. Liu, B. **ao, L. Cui, M. Wang, Mat. Sci. Eng. C 55, 457 (2015)
Y. Li, H. Song, L. Zhang, P. Zuo, B.C. Ye, J. Yao, W. Chen, Biosens. Bioelectron. 78, 308 (2016)
Acknowledgements
The authors wish to thank for the financial support by the Ministry of Science and Technology (MOST) in Taiwan under the contract number of 105-2221-E-035-002-MY3. The support in providing the fabrication and measurement facilities from the Precision Instrument Support Center of Feng Chia University is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, FR., Lee, GJ., Haridharan, N. et al. Electrochemical Sensor Using Molecular Imprinting Polymerization Modified Electrodes to Detect Methyl Parathion in Environmental Media. Electrocatalysis 9, 1–9 (2018). https://doi.org/10.1007/s12678-017-0411-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-017-0411-9