Abstract
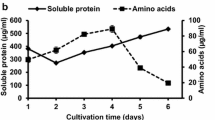
Feather is a good source of nitrogen but the highly rigid structure limits its utilization and leads to environmental pollution and resource wastage. Traditional feather degradation processes such as alkali hydrolysis and steam pressure cooking not only destroy the amino acids but also consume large amounts of energy. Therefore, searching for feather degrading bacteria to transform feather waste to nitrogen fertilizer to improve soil and plant growth is needed. And this also will solve the soil nitrogen deficiency problem in China. In this study, a bacteria that could degrade feather and promote plant growth was isolated from soil sample using dilution plate method. And the isolated bacteria was identified as Bacillus pumilus JYL according to its morphology, biochemical characteristics and 16S rRNA sequence. And the maximum keratinase activity (57.14 U/mL) was obtained under optimal conditions of 0.036% FeSO4, 0.025% MgSO4, 0.073% K2HPO4, 0.5% feather with 2% inoculant at pH 6.5 fermented for 36 h at 35 ℃ after optimization using (response surface methodology) RSM. The effect of hydrolyzate on wheat seed germination and growth was studied. The results showed that germination engergy and germination index with 3% feather hydrolyzate addition group was 94.22% and 87.52%, significantly higher than that of the control group (p < 0.05), respectively. The plant height, root length, fresh weight and dry weight in the T2 treatment (added 5 mL feather hydrolysate in 100 g soil) at 60th day were 1.10, 1.16, 1.77 and 1.73 times than that in the CK treatment (without addition of hydrolysate), respectively. And the soil enzyme activities in all treatment groups (T1, T2, T3) significantly increased compared with control group (CK) (p < 0.05). This result will provide a potential utilization method for feather valoration.
Graphic Abstract






Similar content being viewed by others
References
**, E., Reddy, N., Zhu, Z.Y.: Graft polymerization of native chicken feathers for thermoplastic applications. J. Agric. Food Chem. 59, 1729–1738 (2011)
Dou, Y., Xue, H., Buning, Z., Ming, H., Guoqiang, Y., Yingde, C.: Preparation and characterization of dialdehyde starch crosslinked feather keratin film for food packaging application. Rsc Adv. 5, 27168–27174 (2015)
Gousterova, A., Braikova, D., Goshev, I., Christov, P., Tishinov, K., Vasileva-Tonkova, E., Haertlé, T., Nedkov, P.: Degradation of keratin and collagen containing wastes by newly isolated thermoactinomycetes or by alkaline hydrolysis. Lett. Appl. Microbiol. 40, 335–340 (2005)
Onifade, A.A., Al-Sane, N.A., Al-Musallam, A.A., Al-Zarban, S.: A review: potentials for biotechnological applications of keratin-degrading microorganisms and their enzymes for nutritional improvement of feathers and other keratins as livestock feed resources. Bioresour. Technol. 66, 1–11 (1998)
Pintubala, K., Subhra, S.R., Kumar, S.S., Thangjam, S.S., Meraj, A.A., Narendra, P., Ngachan, S.V.: Transforming chicken feather waste into feather protein hydrolysate using a newly Iisolated multifaceted keratinolytic bacterium Chryseobacterium sediminis RCM-SSR-7. Waste Biomass Valor. 10, 1–11 (2019)
Williams, C.M., Lee, C.G., Garlich, J.D., Shih, J.C.H.: Evaluation of a bacterial feather fermentation product, feather-lysate, as a feed protein. Poultry Sci. 70, 85–94 (1991)
Yamamura, S., Morita, Y., Hasan, Q., Yokoyama, K., Tamiya, E.: Keratin degradation: a cooperative action of two enzymes from Stenotrophomonas sp. Biochem. Biophys. Res. Commun. 294, 1138–1143 (2002)
Jain, R., Jain, A., Rawat, N., Nair, M., Gumashta, R.: Feather hydrolysate from Streptomyces sampsonii GS 1322: a potential low cost soil amendment. J. Biosci. Bioeng. 121, 672–677 (2016)
**ha, J., Omi, L., Youngdong, J., Jeongdo, K., Nari, L., Chungyeol, L., Hongjoo, S.: Production of keratinolytic enzyme by a newly isolated feather-degrading Stenotrophomonas maltophilia that produces plant growth-promoting activity. Process Biochem. 45, 1738–1745 (2010)
Kumar, A.G., Swarnalatha, S., Gayathri, S., Nagesh, N., Sekaran, G.: Characterization of an alkaline active-thiol forming extracellular serine keratinase by the newly isolated Bacillus pumilus. J. Appl. Microbiol. 104, 411–419 (2008)
Okoroma, E.A., Hemda, G., Abiola, O.O., Diane, P.: Identification and characterisation of a Bacillus licheniformis strain with profound keratinase activity for degradation of melanised feather. Int. Biodeter. Biodegr. 74, 54–60 (2012)
Pillai, P., Mandge, S., Archana, G.: Statistical optimization of production and tannery applications of a keratinolytic serine protease from Bacillus subtilis P13. Process Biochem. 46, 1110–1117 (2011)
Brandelli, A., Daroit, D.J., Riffel, A.: Biochemical features of microbial keratinases and their production and applications. Appl. Microbiol. Biotechnol. 85, 1735–1750 (2010)
Choi, J.M., Nelson, P.V.: Develo** a slow-release nitrogen fertilizer from organic sources: using poultry feathers. J. Am. Soc. Hortic. Sci. 121, 634–638 (1996)
Vesela, M., Friedrich, J.: Amino acid and soluble protein cocktail from waste keratin hydrolysed by a fungal keratinase of Paecilomyces marquandii. Biotechnol. Bioprocess. Eng. 14, 84–90 (2009)
Paul, T., Halder, S.K., Das, A., Bera, S., Maity, C.: Exploitation of chicken feather waste as a plant growth promoting agent using keratinase producing novel isolate Paenibacillus woosongensis. Biocatal. Agric. Biotechnol. 2, 50–57 (2013)
Jeong, J.H., Lee, O.M., Jeon, Y.D., Kim, J.D., Lee, N.R.: Production of keratinolytic enzyme by a newly isolated feather-degrading Stenotrophomonas maltophilia that produces plant growth promoting activity. Process Biochem. 45, 1738–1745 (2010)
Lateef, A., Oloke, J.K., Gueguim Kana, E.B., Sobowale, B.O., Ajao, S.O., Bello, B.Y.: Keratinolytic activities of a new feather-degrading isolate of Bacillus cereus LAU 08 isolated from Nigerian soil. Int. Biodeter. Biodegr. 64, 162–165 (2010)
Glickman, E., Dessaux, Y.: A critical examination of the specificity of the Salkowski reagent for indolic compounds produced by phytopathogenic bacteria. Appl. Environ. Microbiol. 61, 793–796 (1995)
Schwyn, B., Neilands, J.B.: Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 160, 47–52 (1987)
Penrose, D.M., Glick, B.R.: Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol. Plant. 118, 10–15 (2003)
Kumar, V., Narula, N.: Solubilization of inorganic phosphates and growth emergence of wheat as affected by Azotobacter chroococcum mutants. Biol. Fertil. Soils. 28, 301–305 (1999)
Singh, A.V., Agarwal, A., Goel, R.: Comparative phosphates solubilization efficiency of two bacterial isolates and their effect on Cicer arietinum seeds in indigenous and alternative soil system. Environ. Ecol. 28, 1979–1983 (2010)
Cappuccino, J.G., Sherman, N.: Microbiology a Laboratory Manual. Benjamin Cummings Science Publishing, California (1996)
Madigan, M.T., Imhoff, J.F.: Phylum BXIII. Firmicutes. In: Boone, D.R., Castenholz, R.W., Garrity, G.M. (eds.) Bergey’s Manual® of Systematic Bacteriology. Springer, New York (2001)
Hosek, J., Svastova, P., Moravkova, M., Pavlik, I., Bartos, M.: Methods of mycobacterial DNA isolation from different biological material: a review. Vet Med. 51, 180–192 (2006)
Rahman, R.N., Mahamad, S., Salleh, A.B., Basri, M.: A new organic solvent tolerant protease from Bacillus pumilus 115b. J. Ind. Microbiol. Biot. 34, 509–517 (2007)
Tamura, K., Stecher, G., Peterson, D., Filipski, A., Kumar, S.: MEGA6: molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30, 2725–2729 (2013)
Gradisar, H., Kern, S., Friedrich, J.: Keratinase of Doratomyces microsporus. Appl. Microbiol. Biotechnol. 53, 196–200 (2000)
Plackett, R.L., Burman, J.P.: The design of optimum multifactorial experiments. Biometrika 33, 305–325 (1946)
Nustorova, M., Braikova, D., Gousterova, A., Vasileva-Tonkova, E., Nedkov, P.: Chemical, microbiological and plant analysis of soil fertilized with alkaline hydrolysate of sheep’s wool waste. World J. Microb. Biot. 22, 383–390 (2006)
Wang, Y.F., Ma, Z.T., Wang, X.W., Sun, Q.R., Dong, H.Q., Wang, G.S., Chen, X.S., Yin, C.M., Han, Z.H., Mao, Z.Q.: Effects of biochar on the growth of apple seedlings, soil enzyme activities and fungal communities in replant disease soil. Sci. Hortic-Amsterdam. (2019). https://doi.org/10.1016/j.scienta.2019.108641
Zhang, Z., Chen, Q., Yin, C., Shen, X., Chen, X., Sun, H., Gao, A.N., Mao, Z.: The effects of organic matter on the physiological features of Malus hupehensis seedlings and soil properties under replant conditions. Sci. Hortic. 146, 52–58 (2012)
Richardson, A.E., Barea, J.M., McNeill, A.M., Prigent-Combaret, C.: Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil 321, 305–339 (2009)
Mendes, R., Kruijt, M., de Bruijn, I., Dekkers, E., van der Voort, M., Schneider, J.H., Piceno, Y.M., DeSantis, T.Z., Andersen, G.L., Bakker, P.A., Raaijmakers, J.M.: Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332, 1097–1100 (2011)
Kumari, P., Meena, M., Upadhyay, R.S.: Characterization of plant growth promoting rhizobacteria (PGPR) isolated from the rhizosphere of Vigna radiata (mung bean). Biocatal. Agric. Biotechnol. 16, 155–162 (2018)
Bushra, T., Anwar, K., Muhammad, T., Memoona, R., Muhammad, S.I.K., Naila, S., Khadija, A.: Bottlenecks in commercialisation and future prospects of PGPR. Appl. Soil Ecol. 121, 102–117 (2017)
Dong, X., Cai, M.: Handbook for Systematic Identification of Common Bacteria. China Science Press, Bei**g (2001)
Gupta, R., Ramnani, P.: Microbial keratinases and their prospective applications: an overview. Appl. Microbiol. Biotechnol. 70, 21–33 (2006)
Din, B.U., Sarfraz, S., **a, Y., Kamran, M.A., Javed, M.T., Sultan, T., Munis, M.F.H., Chaudhary, H.J.: Mechanistic elucidation of germination potential and growth of wheat inoculated with exopolysaccharide and ACC- deaminase producing Bacillus strains under induced salinity stress. Ecotoxicol. Environ. Saf. 183, 109466 (2019)
Sobucki, L., Ramos, R.F., Gubiani, E., Brunetto, G., Kaiser, D.R., Daroit, D.J.: Feather hydrolysate as a promising nitrogen-rich fertilizer for greenhouse lettuce cultivation. Int. J. Recycl. Org. Waste Agric. 8, 493–499 (2019). https://doi.org/10.1007/s40093-019-0281-7
Joardar, J.C., Rahman, M.M.: Poultry feather waste management and effects on plant growth. Int. J. Recycl. Org. Waste Agric. 7, 183–188 (2018)
Gurav, R.G., Jadhav, J.P.: A novel source of biofertilizer from feather biomass for banana cultivation. Environ. Sci. Pollut. Res. 20, 4532–4539 (2013). https://doi.org/10.1007/s11356-012-1405-z
Hadas, A., Kautsky, L.: Feather meal, a semi-slow release nitrogen fertilizer for organic farming. Fert. Res. 38, 165–170 (1994)
Gupta, R., Rajput, R., Sharma, R., Gupta, N.: Biotechnological applications and prospective market of microbial keratinases. Appl. Microbiol. Biotechnol. 97, 9931–9940 (2013)
Rai, S.K., Mukherjee, A.K.: Optimization for production of liquid nitrogen fertilizer from the degradation of chicken feather by iron-oxide (Fe3O4) magnetic nanoparticles coupled β-keratinase. Biocatal. Agric. Biotechnol. 4, 632–644 (2015)
Gianfreda, L., Antonietta, R.M., Piotrowska, A., Palumbo, G., Colombo, C.: Soil enzyme activities as affected by anthropogenic alterations: intensive agricultural practices and organic pollution. Sci. Total Environ. 341, 265–279 (2005)
Acosta-Martínez, V., Cruz, L., Sotomayor-Ramírez, D., Pérez-Alegría, L.: Enzyme activities as affected by soil properties and land use in a tropical watershed. Appl. Soil Ecol. 35, 35–45 (2007)
Wei, T., Zhang, P., Wang, K., Ding, R., Yang, B., Nie, J., Jia, Z., Han, Q.: Effects of wheat straw incorporation on the availability of soil nutrients and enzyme activities in semiarid areas. PLoS ONE 10, e0120994 (2015)
Acknowledgements
This research was sponsored by Shandong Modern Agricultural Industry Technology System (SDAIT-05-05), Shandong “Double Tops” Program (SYL2017XTTD15), Shandong Province Agricultural Major Application Technology Innovation Project (2018), Shandong Province Major Science and Technology Innovation Project (2017CXGC0311), and Agricultural Science and Technology Innovation Project funded by Shandong Academy of Agricultural Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, Z., Li, X., Liu, K. et al. Optimization for Production of a Plant Growth Promoting Agent from the Degradation of Chicken Feather Using Keratinase Producing Novel Isolate Bacillus pumilus JYL. Waste Biomass Valor 12, 1943–1954 (2021). https://doi.org/10.1007/s12649-020-01138-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-020-01138-7