Abstract
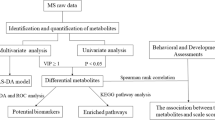
Autism spectrum disorders (ASD) are increasingly common neurodevelopmental disorders accompanied by dysregulation of amino acid (AA) metabolism, and for which there are currently no reliable early diagnostic biomarkers. This study evaluated whether specific AAs can serve as biomarkers for screening ASD patients by analyzing the abundance 21 plasma AAs in 70 ASD patients and 70 control subjects by liquid chromatography–tandem mass spectrometry. We found significant differences between the two groups for eight of the AAs—namely, arginine, cysteine, homocysteine, histidine, methionine, serine, tyrosine, and valine. However, only homocysteine level was positively correlated with ASD symptom severity. Arginine, cysteine, histidine, and methionine were used to generate a predictive model in the Fisher discriminant analysis; cross-validation of this model showed that 88.6% of individuals were correctly segregated into ASD and healthy subject groups with a sensitivity of 85.5% and specificity of 92.2%. The area under the receiver operating characteristic curve was 0.959 (0.927–0.991). Thus, detection of a combination of AAs is an effective method for distinguishing ASD patients from healthy subjects, which may be useful for the early diagnosis of ASD.


Similar content being viewed by others
References
Agrawal A, Ilango K, Singh PK, Karmakar D, Singh GP et al (2015) Age dependent levels of plasma homocysteine and cognitive performance. Behav Brain Res 283:139–144. https://doi.org/10.1016/j.bbr.2015.01.016
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association, Washington, DC. 58–62
Anwar A, Abruzzo PM, Pasha S, Rajpoot K, Bolotta A, Ghezzo A, Marini M, Posar A, Visconti P, Thornalley PJ, Rabbani N (2018) Advanced glycation endproducts, dityrosine and arginine transporter dysfunction in autism - a source of biomarkers for clinical diagnosis. Mol Autism 9:3. https://doi.org/10.1186/s13229-017-0183-3
Azzini E, Ruggeri S, Polito A (2020) Homocysteine: its possible emerging role in at-risk population groups. Int J Mol Sci 21(4). https://doi.org/10.3390/ijms21041421
Baio J, Wiggins L, Christensen DL, Maenner MJ, Daniels J, et al (2018) Prevalence of autism spectrum disorder among children aged 8 years - Autism and Developmental Disabilities Monitoring Network, 11 sites, United States, 2014. MMWR Surveill Summ 67(6): 1-23. https://doi.org/10.15585/mmwr.ss6706a1
Bitar T, Mavel S, Emond P, Nadal-Desbarats L, Lefevre A et al (2018) Identification of metabolic pathway disturbances using multimodal metabolomics in autistic disorders in a Middle Eastern population. J Pharm Biomed Anal 152:57–65. https://doi.org/10.1016/j.jpba.2018.01.007
Campbell NG, Shekar A, Aguilar JI, Peng D, Navratna V, Yang D, Morley AN, Duran AM, Galli G, O’Grady B, Ramachandran R, Sutcliffe JS, Sitte HH, Erreger K, Meiler J, Stockner T, Bellan LM, Matthies HJG, Gouaux E, Mchaourab HS, Galli A (2019) Structural, functional, and behavioral insights of dopamine dysfunction revealed by a deletion in SLC6A3. Proc Natl Acad Sci U S A 116(9):3853–3862. https://doi.org/10.1073/pnas.1816247116
Desai A, Sequeira JM, Quadros EV (2016) The metabolic basis for developmental disorders due to defective folate transport. Biochimie 126:31–42. https://doi.org/10.1016/j.biochi.2016.02.012
di Dieme B, Mavel S, Blasco H, Tripi G, Bonnet-Brilhault F et al (2015) Metabolomics study of urine in autism spectrum disorders using a multiplatform analytical methodology. J Proteome Res 14(12):5273–5282. https://doi.org/10.1021/acs.jproteome.5b00699
Diémé B, Mavel S, Hln B, Tripi G, Fdr B-B et al (2015) Metabolomics study of urine in autism spectrum disorders using a multiplatform analytical methodology. J Proteome Res 14(12):5273–5282
Evans C, Dunstan RH, Rothkirch T, Roberts TK, Reichelt KL et al (2008) Altered amino acid excretion in children with autism. Nutr Neurosci 11(1):9–17. https://doi.org/10.1179/147683008x301360
Fuentes-Albero M, Cauli O (2018) Homocysteine levels in autism spectrum disorder: a clinical update. Endocr Metab Immune Disord Drug Targets 18(4):289–296. https://doi.org/10.2174/1871530318666180213110815
Gevi F, Zolla L, Gabriele S, Persico AM (2016) Urinary metabolomics of young Italian autistic children supports abnormal tryptophan and purine metabolism. Mol Autism 7:47. https://doi.org/10.1186/s13229-016-0109-5
Gotham K, Pickles A, Lord C (2009) Standardizing ADOS scores for a measure of severity in autism spectrum disorders. J Autism Dev Disord 39(5):693–705. https://doi.org/10.1007/s10803-008-0674-3
Huang F, Chen X, Jiang X, Niu J, Cui C, Chen Z, Sun J (2019) Betaine ameliorates prenatal valproic-acid-induced autism-like behavioral abnormalities in mice by promoting homocysteine metabolism. Psychiatry Clin Neurosci 73:317–322. https://doi.org/10.1111/pcn.12833
Hus V, Gotham K, Lord C (2014) Standardizing ADOS domain scores: separating severity of social affect and restricted and repetitive behaviors. J Autism Dev Disord 44(10):2400–2412. https://doi.org/10.1007/s10803-012-1719-1
Kelly RS, Boulin A, Laranjo N, Lee-Sarwar K, Chu SH, Yadama A, Carey V, Litonjua A, Lasky-Su J, Weiss S (2019) Metabolomics and communication skills development in children; evidence from the ages and stages questionnaire. Metabolites 9(3):42. https://doi.org/10.3390/metabo9030042
Kuwabara H, Yamasue H, Koike S, Inoue H, Kawakubo Y, Kuroda M, Takano Y, Iwashiro N, Natsubori T, Aoki Y, Kano Y, Kasai K (2013) Altered metabolites in the plasma of autism spectrum disorder: a capillary electrophoresis time-of-flight mass spectroscopy study. PLoS One 8(9):e73814. https://doi.org/10.1371/journal.pone.0073814
Liu A, Zhou W, Qu L, He F, Wang H, Wang Y, Cai C, Li X, Zhou W, Wang M (2019) Altered urinary amino acids in children with autism spectrum disorders. Front Cell Neurosci 13:7. https://doi.org/10.3389/fncel.2019.00007
Lussu M, Noto A, Masili A, Rinaldi AC, Dessi A et al (2017) The urinary (1) H-NMR metabolomics profile of an Italian autistic children population and their unaffected siblings. Autism Res 10(6):1058–1066. https://doi.org/10.1002/aur.1748
Maenner MJ, Shaw KA, Baio J, Washington A, Patrick M et al (2020) Prevalence of autism spectrum disorder among children aged 8 years - Autism and Developmental Disabilities Monitoring Network, 11 sites, United States, 2016. MMWR Surveill Summ 69(4):1–12. https://doi.org/10.15585/mmwr.ss6904a1
Maynard TM, Manzini MC (2017) Balancing act: maintaining amino acid levels in the autistic brain. Neuron 93(3):476–479. https://doi.org/10.1016/j.neuron.2017.01.015
Pasca SP, Nemes B, Vlase L, Gagyi CE, Dronca E et al (2006) High levels of homocysteine and low serum paraoxonase 1 arylesterase activity in children with autism. Life Sci 78(19):2244–2248. https://doi.org/10.1016/j.lfs.2005.09.040
Proenza AM, Crespi C, Roca P, Palou A (2001) Gender related differences in the effect of aging on blood amino acid compartmentation*. J Nutr Biochem 12(7):431–440
Puig-Alcaraz C, Fuentes-Albero M, Calderon J, Garrote D, Cauli O (2015) Increased homocysteine levels correlate with the communication deficit in children with autism spectrum disorder. Psychiatry Res 229(3):1031–1037. https://doi.org/10.1016/j.psychres.2015.05.021
Ranjan S, Nasser JA (2015) Nutritional status of individuals with autism spectrum disorders: do we know enough? Adv Nutr 6(4):397–407. https://doi.org/10.3945/an.114.007914
Rosina E, Battan B, Siracusano M, Di Criscio L, Hollis F et al (2019) Disruption of mTOR and MAPK pathways correlates with severity in idiopathic autism. Transl Psychiatry 9(1):50. https://doi.org/10.1038/s41398-018-0335-z
Schaevitz LR, Berger-Sweeney JE (2012) Gene-environment interactions and epigenetic pathways in autism: the importance of one-carbon metabolism. ILAR J 53(3–4):322–340. https://doi.org/10.1093/ilar.53.3-4.322
Smith AM, King JJ, West PR, Ludwig MA, Donley ELR, et al (2018) Amino acid dysregulation metabotypes: potential biomarkers for diagnosis and individualized treatment for subtypes of autism spectrum disorder. Biol Psychiatry. https://doi.org/10.1016/j.biopsych.2018.08.016
Smith AM, King JJ, West PR, Ludwig MA, Donley ELR, Burrier RE, Amaral DG (2019) Amino acid dysregulation metabotypes: potential biomarkers for diagnosis and individualized treatment for subtypes of autism spectrum disorder. Biol Psychiatry 85(4):345–354. https://doi.org/10.1016/j.biopsych.2018.08.016
Sperringer JE, Addington A, Hutson SM (2017) Branched-chain amino acids and brain metabolism. Neurochem Res 42(6):1697–1709. https://doi.org/10.1007/s11064-017-2261-5
Sun X, Allison C, Wei L, Matthews FE, Auyeung B, Wu YY, Griffiths S, Zhang J, Baron-Cohen S, Brayne C (2019) Autism prevalence in China is comparable to Western prevalence. Mol Autism 10:7. https://doi.org/10.1186/s13229-018-0246-0
Sweeten TL, Posey DJ, Shankar S, McDougle CJ (2004) High nitric oxide production in autistic disorder: a possible role for interferon-gamma. Biol Psychiatry 55(4):434–437. https://doi.org/10.1016/j.biopsych.2003.09.001
Tarlungeanu DC, Deliu E, Dotter CP, Kara M, Janiesch PC et al (2016) Impaired amino acid transport at the blood brain barrier is a cause of autism spectrum disorder. Cell 167(6):1481–1494.e1418. https://doi.org/10.1016/j.cell.2016.11.013
Tirouvanziam R, Obukhanych TV, Laval J, Aronov PA, Libove R, Banerjee AG, Parker KJ, O’Hara R, Herzenberg LA, Herzenberg LA, Hardan AY (2012) Distinct plasma profile of polar neutral amino acids, leucine, and glutamate in children with autism spectrum disorders. J Autism Dev Disord 42(5):827–836. https://doi.org/10.1007/s10803-011-1314-x
Tu WJ, Chen H, He J (2012) Application of LC-MS/MS analysis of plasma amino acids profiles in children with autism. J Clin Biochem Nutr 51(3):248–249. https://doi.org/10.3164/jcbn.12-45
Uehara EU, Shida Bde S and de Brito CA (2015) Role of nitric oxide in immune responses against viruses: beyond microbicidal activity. 64(11): 845-852. https://doi.org/10.1007/s00011-015-0857-2
van Sadelhoff JHJ, Perez Pardo P, Wu J, Garssen J, van Bergenhenegouwen J, Hogenkamp A, Hartog A, Kraneveld AD (2019) The gut-immune-brain axis in autism spectrum disorders; a focus on amino acids. Front Endocrinol (Lausanne) 10:247. https://doi.org/10.3389/fendo.2019.00247
Vargason T, Kruger U, McGuinness DL, Adams JB, Geis E et al (2018) Investigating plasma amino acids for differentiating individuals with autism spectrum disorder and typically develo** peers. Res Autism Spectr Disord 50:60–72. https://doi.org/10.1016/j.rasd.2018.03.004
Weiser MJ, Butt CM, Mohajeri MH (2016) Docosahexaenoic acid and cognition throughout the lifespan. Nutrients 8(2):99. https://doi.org/10.3390/nu8020099
Wu J, de Theije CGM, da Silva SL, Abbring S, van der Horst H, Broersen LM, Willemsen L, Kas M, Garssen J, Kraneveld AD (2017) Dietary interventions that reduce mTOR activity rescue autistic-like behavioral deficits in mice. Brain Behav Immun 59:273–287. https://doi.org/10.1016/j.bbi.2016.09.016
Zhang S, Wang R, Wang G (2019) Impact of dopamine oxidation on dopaminergic neurodegeneration. ACS Chem Neurosci 10(2):945–953. https://doi.org/10.1021/acschemneuro.8b00454
Zou M, Sun C, Wang J, Kang J, Xu Z, Ma Y, Chen L, Zhang X, **a W, Wu L (2018) Factors influencing the severity of behavioral phenotype in autism spectrum disorders: implications for research. Psychiatry Res 261:290–297. https://doi.org/10.1016/j.psychres.2017.12.084
Acknowledgments
We thank all patients and their parents for their support and participation.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81773458) and Natural Science Foundation of Heilongjiang Province (No. LH2019H007).
Author information
Authors and Affiliations
Contributions
SX and LL did the literature review; DL and LW wrote the manuscript. YL and WX performed statistical analysis. CS and MZ assisted with the writing of the manuscript. All authors read, corrected, and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
The study protocol was approved by the Institutional Review Board of Harbin Medical University for Medical Sciences. All experiments on human subjects were conducted in accordance with the Declaration of Helsinki. All procedures were carried out with the adequate understanding and written informed consent from the guardian of each participant prior to the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zou, M., Li, D., Wang, L. et al. Identification of Amino Acid Dysregulation as a Potential Biomarker for Autism Spectrum Disorder in China. Neurotox Res 38, 992–1000 (2020). https://doi.org/10.1007/s12640-020-00242-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00242-9