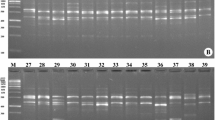
Abstract
North east India is considered as one of the major biodiversity hotspots worldwide and centre of origin of several plant species including Musa. Musa acuminata Colla is known to be one of the wild progenitors of cultivated bananas and plantains. Three single primer based DNA marker techniques viz., random amplified polymorphic DNA (RAPD), inter simple sequence repeat (ISSR) and directed amplification of minisatellites DNA (DAMD) were used for diversity diagnostics among 25 genotypes of wild M. acuminata collected from Meghalaya province of north east India. A total of 58 primers (26-RAPD, 21-ISSR, and11-DAMD) yielded 451 DNA fragments, of which 395 (87.58 %) were found to be polymorphic in nature. The polymorphic information content (PIC) values were almost identical for each marker system. The resolving power of the marker system was found to be highest in RAPD (3.96) whereas ISSR resolved highest marker index (16.39) in the study. Selected amplicon data obtained through single primer amplification reactions were utilized for determination of diversity within and among the populations of M. acuminata. Nei’s genetic differentiation (Gst) value (0.451) indicated higher proportion of the genetic variation within the populations which is supported by the AMOVA analysis (88 %). The study provides insight into the efficacy of RAPD, ISSR and DAMD to analyse the genetic variation existing in the wild Musa germplasm, which can further be exploited for quality trait improvement and domestication of such important horticultural crops. The genetic diversity based population structure may shed light on the genetic basis of speciation and evolution of various species within the genus Musa.


Similar content being viewed by others
References
Arnaud-Haond S, Teixeira S, Massa S, Billot C, Saenger P, Coupland G, Duarte CM, Serrão EA (2006) Genetic structure at range edge: low diversity and high inbreeding in Southeast Asian mangrove (Avicennia marina) populations. Mol Ecol 15:3515–3525
Bhat KV, Bhat SR, Chandel KPS (1992) Survey of isozyme polymorphism for clonal identification in Musa. I. Esterase, acid phosphatase and catalase. J Hort Sci 67:501–507
Bhattacharya E, Dandin SB, Ranade SA (2005) Single primer amplification methods reveal exotic and indigenous mulberry varieties are similarly diverse. J Biosci 30:669–677
Byars SG, Parsons Y, Hoffmann AA (2009) Effect of altitude on the genetic structure of an Alpine grass, Poa hiemata. Ann Bot 103:885–899
Creste S, Tulmann-Neto A, Silva SO, Figueira A (2003) Genetic characterization of banana cultivars (Musa spp.) from Brazil using microsatellite markers. Euphytica 132:259–268
Creste S, Tulmann-Neto A, Vencovsky R, Silva SO, Figueira A (2004) Genetic diversity of Musa diploid and triploid accessions from the Brazilian banana breeding program estimated by Microsatellite markers. Genet Res Crop Evol 51:723–733
Daniells JW, Jenny C, Karamura DA, Tomekpe K, Arnaud E, Sharrock S (2001) Musalogue: A catalogue of Musa germplasm diversity in the genus Musa. INIBAP, France
Dickinson A (2000). Recommended intakes of vitamins and essential minerals. In: CRN (Council for responsible Nutrition). The benefits of Nutritional Supplements
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Frison EA, Escalant JV, Sharrock S (2004) The global Musa genomic consortium: A boost for banana improvement. In: Jain SM, Swennen R (eds) Banana improvement: Cellular, molecular biology, and induced mutations. Science Publishers, Enfield, pp 341–349
Gawel NJ, Jarret RL (1991) A modified CTAB DNA extraction procedure for Musa and Ipomoea. Plant Mol Biol Rep 9:262–266
Ge XJ, Liu MH, Wang WK, Schaal BA, Chiang TY (2005) Population structure of wild bananas, Musa balbisiana, in China determined by SSR fingerprinting and cpDNA-RFLP. Mol Ecol 14:933–944
Hamrick JL, Godt MJW (1996) Effects of life history traits on genetic diversity in plant species. Philos Trans R Soc Lond B Biol Sci 351:1291–1298
Heath DD, Iwana GK, Delvin RH (1993) PCR primed with VNTR core sequences yields species specific patterns and hypervariable probes. Nucleic Acids Res 21:5782–5785
Hogbin PM, Peakall R (1999) Evaluation of the contribution of genetic research to the management of the endangered plant Zieria prostrata. Conserv Biol 13:514–522
INIBAP/CIRAD (1996) Descriptors for Banana (Musa spp.) INIBAB Montpellier. France. http://www.Musalit.org/pdf/IN100330_en.pdf.
Jarret RL, Gawel NJ, Wittermore A (1992) RFLP-based phylogeny of Musa species in Papua New Guinea. Theor Appl Genet 84:579–584
Jarret RL, Vulysteke D, Gawel NJ, Pimentel R, Dunbar L (1993) Detecting genetic diversity in diploid bananas using PCR and primers from a highly repetitive DNA sequence. Euphytica 68:69–76
Javed MA, Chai M, Othman RY (2004) Study of resistance of Musa acuminata to Fusarium oxysporum using RAPD markers. Biol Plant 48:93–99
Kimura M, Crow J (1964) The number of alleles that can be maintained in a finite population. Genetics 49:725–738
Kumar S, Kumaria S, Sharma SK, Rao SR, Tandon P (2011) Genetic diversity assessment of Jatropha curcas L. germplasm from Northeast India. Biomass Bioenerg 35:3063–3070
Kumar S, Kumaria S, Tandon P (2013) SPAR methods coupled with seed-oil content revealed intra-specific natural variation in Jatropha curcas L. from Northeast India. Biomass Bioenerg 54:100–106
Kumar A, Mishra P, Singh SC, Sundaresan V (2014) Efficiency of ISSR and RAPD markers in genetic divergence analysis and conservation management of Justicia adhatoda L., a medicinal plant. Plant Syst Evol 300:1409–1420
Lagoda PJL, Noyer JL, Dambier D, Baurens FC, Grapin A, Lanaud C (1998) Sequence tagged microsatellite site (STMS) markers in the Musaceae. Mol Ecol 7:657–666
Lewontin RC (1972) The apportionment of human diversity. Evol Biol 6:381–398
Liu YF, **ng M, Zhao W, Fan RJ, Luo S, Chen X (2012) Genetic diversity analysis of Rhododendron aureum Georgi (Ericaceae) located on Changbai Mountain using ISSR and RAPD markers. Plant Syst Evol 298:921–930
Lynch M, Milligan BG (1994) Analysis of population structure with RAPD markers. Mol Ecol 3:91–99
McDermott JM, McDonald BA (1993) Gene flow in plant pathosystems. Ann Rev Phytopathol 31:353–373
Meyer W, Mitchell TG, Freedman EZ, Vilgays R (1993) Hybridization probes for conventional DNA fingerprinting used as single primers in the polymerase chain reaction to distinguish strains of Cryptococcus neoformans. J Clin Microbiol 31:2274–2280
Miller MP (1998) AMOVA-PREP1.01. A Program for the Preparation of AMOVA Input Files From Dominant-Marker Raw Data. Northern Arizona University, Flagstaff, Arizona. USA.
Mukunthakumar S, Padmesh P, Vineesh PS, Skaria R, Kumar KH, Krishnan PN (2013) Genetic diversity and differentiation analysis among wild antecedents of banana (Musa acuminata Colla) using RAPD markers. Indian J Biotechnol 12:493–498
Myers N, Mittermeier RA, Mittermeier CG, Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci U S A 70:3321–3323
Ohsawa T, Ide Y (2008) Global patterns of genetic variation in plant species along vertical and horizontal gradients on mountains. Glob Ecol Biogeogr 17:152–163
Onguso JM, Kahangi EM, Ndiritu DW, Mizutani F (2004) Genetic characterization of cultivated bananas and plantains in Kenya by RAPD markers. Sci Hort 99:9–20
Padmesh P, Mukunthakumar S, Vineesh PS, Skaria R, Kumar KH, Krishnan PN (2012) Exploring wild genetic resources of Musa acuminata Colla distributed in the humid forests of southern Western Ghats of peninsular India using ISSR markers. Plant Cell Rep 31:1591–1601
Prevost A, Wilkinson MJ (1999) A new system of comparing PCR primers applied to ISSR fingerprinting of potato cultivars. Theor App Genet 98:107–112
Ranade SA, Rana TS, Narzary D (2009) SPAR profile and genetic diversity amongst pomegranate (Punica granatum L.) genotypes. Physiol Mol Biol Plants 15:61–70
Scariot V, de Keyser E, Handa T, de Riek J (2007) Comparative study of the discriminating capacity and effectiveness of AFLP, STMS and EST markers in assessing genetic relationships among evergreen azaleas. Plant Breed 126(2):207–212
Sehgal D, Rajpal VR, Raina SN, Sasanuma T, Sasakuma T (2009) Assaying polymorphism at DNA level for genetic diversity diagnostics of the safflower (Carthamus tinctorius L.) world germplasm resources. Genetica 135:457–470
Sharma SK, Rawat D, Kumar S, Kumar A, Kumaria S, Rao SR (2010) Single primer amplification reaction (SPAR) reveals intra-specific natural variation in Prosopis cineraria (L.) Druce. Trees Struct Funct 24:855–864
Sharma SK, Kumaria S, Tandon P, Rao SR (2011) Single primer amplification reaction (SPAR) reveals inter-and intra-specific natural genetic variation in five species of Cymbidium (Orchidaceae). Gene 483:54–62
Simmonds NW (1962) Evolution of the bananas. Longmans, London
Simmonds NW, Shepherd K (1955) The taxonomy and origins of cultivated bananas. Bot J Linn Soc 55:302–312
Smith JSC, Chin ECL, Shu H, Smith OS, Wall SJ, Senior ML, Mitchell SE, Kresovich S, Ziegle J (1997) An evaluation of the utility of SSR loci as molecular markers in maize (Zea mays L.): comparisons with data from RFLPs and pedigree. Theor Appl Genet 95:163–173
Sneath PHA, Sokal RR (1973) Numerical taxonomy. The principles and practice of numerical classification
Uma S, Siva SA, Saraswathi MS, Manickavasagam M, Durai P, Selvarajan R, Sathiamoorthy (2006) Variation and intraspecific relationships in Indian wild Musa balbisiana (BB) population as evidenced by random amplified polymorphic DNA. Genet Resour Crop Evol 53:349–355
Varshney RK, Chabane K, Hendre PS, Aggarwal RK, Graner A (2007) Comparative assessment of EST-SSR, EST-SNP and AFLP markers for evaluation of genetic diversity and conservation of genetic resources using wild, cultivated and elite barleys. Plant Sci 173(6):638–649
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new concept for DNA fingerprinting. Nucleic Acids Res 21:4407–4414
Williams JGK, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphism amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18:6531–6535
Wong C, Kiew R, Argent G, Set O, Lee SK, Gan YY (2002) Assessment of the validity of the sections in Musa (Musaceae) using AFLP. Ann Bot 90:231–238
Yeh FC, Young RC, Boyle T (1999) Popgene version 1.31: microsoft window-based freeware for population genetic analysis
Zawko G, Krauss SL, Dixon KW, Sivasithamparam K (2001) Conservation genetics of the rare and endangered Leucopogon obtectus (Ericaceae). Mol Ecol 10:2389–2396
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)—anchored polymerase chain reaction amplification. Genomics 20(2):176–183
Acknowledgments
We thank the Head, Department of Biotechnology and Bioinformatics, NEHU, Shillong for providing the facilities and support to carry out this research. Sincere thanks are also accorded to Dr. C.S. Rao (St. Anthony’s College, Shillong, Meghalaya), Dr. Santosh K. Sharma, Dr. Shrawan Kumar and all members of the Plant Biotechnology Laboratory for their constant encouragement and support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig S1
(PPTX 960 kb)
Supplementary Fig S2
(PPTX 293 kb)
Rights and permissions
About this article
Cite this article
Lamare, A., Rao, S.R. Efficacy of RAPD, ISSR and DAMD markers in assessment of genetic variability and population structure of wild Musa acuminata colla. Physiol Mol Biol Plants 21, 349–358 (2015). https://doi.org/10.1007/s12298-015-0295-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-015-0295-1