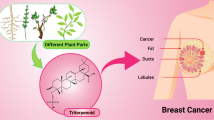
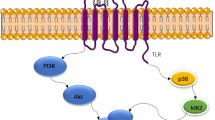
Abstract
Breast cancer is the most common cause of cancer-related deaths among women worldwide. Thus, the development of new and effective low-toxicity drugs is vital. The specific characteristics of breast cancer have allowed for the development of targeted therapy towards each breast cancer subtype. Nevertheless, increasing drug resistance is displayed by the changing phenotype and microenvironments of the tumor through mutation or dysregulation of various mechanisms. Recently, emerging data on the therapeutic potential of biocompounds isolated from ginseng have been reported. Therefore, in this review, various roles of ginsenosides in the treatment of breast cancer, including apoptosis, autophagy, metastasis, epithelial-mesenchymal transition, epigenetic changes, combination therapy, and drug delivery system, have been discussed.




Similar content being viewed by others
References
Abdel-Hafiz HA, Horwitz KB (2015) Role of epigenetic modifications in luminal breast cancer. Epigenomics 7(5):847–862. https://doi.org/10.2217/epi.15.10
Balanis N, Carlin CR (2017) Stress-induced EGF receptor signaling through STAT3 and tumor progression in triple-negative breast cancer. Mol Cell Endocrinol 451:24–30. https://doi.org/10.1016/j.mce.2017.01.013
Chaudhary P, Ha E, Vo TTL, Seo JH (2019) Diverse roles of arrest defective 1 in cancer development. Arch Pharm Res 42(12):1040–1051. https://doi.org/10.1007/s12272-019-01195-0
Chen L, Yan KP, Liu XC, Wang W, Li C, Li M, Qiu CG (2018) Valsartan regulates TGF-beta/Smads and TGF-beta/p38 pathways through lncRNA CHRF to improve doxorubicin-induced heart failure. Arch Pharm Res 41(1):101–109. https://doi.org/10.1007/s12272-017-0980-4
Chen WW, Huang YF, Hu ZB, Liu YM, **ao HX, Liu DB, Zhuang YZ (2019) Microarray analysis of altered long non-coding RNA expression profile in liver cancer cells treated by ginsenoside Rh2. J Asian Nat Prod Res 21(8):742–753. https://doi.org/10.1080/10286020.2018.1490273
Cho ES, Kang HE, Kim NH, Yook JI (2019) Therapeutic implications of cancer epithelial-mesenchymal transition (EMT). Arch Pharm Res 42(1):14–24. https://doi.org/10.1007/s12272-018-01108-7
Choi S, Kim TW, Singh SV (2009) Ginsenoside Rh2-mediated G1 phase cell cycle arrest in human breast cancer cells is caused by p15 Ink4B and p27 Kip1-dependent inhibition of cyclin-dependent kinases. Pharm Res 26(10):2280–2288. https://doi.org/10.1007/s11095-009-9944-9
Choi S, Oh JY, Kim SJ (2011) Ginsenoside Rh2 induces Bcl-2 family proteins-mediated apoptosis in vitro and in xenografts in vivo models. J Cell Biochem 112(1):330–340. https://doi.org/10.1002/jcb.22932
Christensen LP (2009) Ginsenosides chemistry, biosynthesis, analysis, and potential health effects. Adv Food Nutr Res 55:1–99. https://doi.org/10.1016/S1043-4526(08)00401-4
Chung Y, Jeong S, Choi HS, Ro S, Lee JS, Park JK (2018) Upregulation of autophagy by ginsenoside Rg2 in MCF-7 cells. Anim Cells Syst (Seoul) 22(6):382–389. https://doi.org/10.1080/19768354.2018.1545696
Cong Z, Zhao Q, Yang B, Cong D, Zhou Y, Lei X, Zhang X (2019) Ginsenoside Rh3 inhibits proliferation and induces apoptosis of colorectal cancer cells. Pharmacology. https://doi.org/10.1159/000503821
Cui L, Bu W, Song J, Feng L, Xu T, Liu D, Ding W, Wang J, Li C, Ma B, Luo Y, Jiang Z, Wang C, Chen J, Hou J, Yan H, Yang L, Jia X (2018) Apoptosis induction by alantolactone in breast cancer MDA-MB-231 cells through reactive oxygen species-mediated mitochondrion-dependent pathway. Arch Pharm Res 41(3):299–313. https://doi.org/10.1007/s12272-017-0990-2
de Oliveira TM, Nabavi S, Wang Y, Tonellato P, Esteva FJ, Cantley LC, Wulf GM (2017) Genomic characteristics of trastuzumab-resistant Her2-positive metastatic breast cancer. J Cancer Res Clin Oncol 143(7):1255–1262. https://doi.org/10.1007/s00432-017-2358-x
Dong B, Pang TT (2017) LncRNA H19 contributes to Rh2-mediated MC3T3-E1cell proliferation by regulation of osteopontin. Cell Mol Biol (Noisy-le-grand) 63(8):1–6. https://doi.org/10.14715/cmb/2017.63.8.1
Dong Y, Fu R, Yang J, Ma P, Liang L, Mi Y, Fan D (2019) Folic acid-modified ginsenoside Rg5-loaded bovine serum albumin nanoparticles for targeted cancer therapy in vitro and in vivo. Int J Nanomed 14:6971–6988. https://doi.org/10.2147/IJN.S210882
Duan Z, Wei B, Deng J, Mi Y, Dong Y, Zhu C, Fu R, Qu L, Fan D (2018) The anti-tumor effect of ginsenoside Rh4 in MCF-7 breast cancer cells in vitro and in vivo. Biochem Biophys Res Commun 499(3):482–487. https://doi.org/10.1016/j.bbrc.2018.03.174
Elzoghby AO, Samy WM, Elgindy NA (2012) Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release 157(2):168–182. https://doi.org/10.1016/j.jconrel.2011.07.031
Escriva-de-Romani S, Arumi M, Bellet M, Saura C (2018) HER2-positive breast cancer: current and new therapeutic strategies. Breast 39:80–88. https://doi.org/10.1016/j.breast.2018.03.006
Feng Y, Sun T, Yu Y, Gao Y, Wang X, Chen Z (2018) MicroRNA-370 inhibits the proliferation, invasion and EMT of gastric cancer cells by directly targeting PAQR4. J Pharmacol Sci 138(2):96–106. https://doi.org/10.1016/j.jphs.2018.08.004
Gao Q, Zheng J (2018) Ginsenoside Rh2 inhibits prostate cancer cell growth through suppression of microRNA-4295 that activates CDKN1A. Cell Prolif 51(3):e12438. https://doi.org/10.1111/cpr.12438
Gao H, Liang D, Li C, Xu G, Jiang M, Li H, Yin J, Song Y (2020) 2-Deoxy-Rh2: A novel ginsenoside derivative, as dual-targeting anti-cancer agent via regulating apoptosis and glycolysis. Biomed Pharmacother 124:109891. https://doi.org/10.1016/j.biopha.2020.109891
Ge G, Yan Y, Cai H (2017) Ginsenoside Rh2 inhibited proliferation by inducing ROS mediated ER stress dependent apoptosis in lung cancer cells. Biol Pharm Bull 40(12):2117–2124. https://doi.org/10.1248/bpb.b17-00463
Gong J, Li Y, Liu CJ, **ang Y, Li C, Ye Y, Zhang Z, Hawke DH, Park PK, Diao L, Putkey JA, Yang L, Guo AY, Lin C, Han L (2017) A pan-cancer analysis of the expression and clinical relevance of small nucleolar RNAs in human cancer. Cell Rep 21(7):1968–1981. https://doi.org/10.1016/j.celrep.2017.10.070
Gu M, Wang L, Yang C, Li X, Jia C, Croteau S, Ruan X, Hardy P (2018) Micro-RNA-181a suppresses progestin-promoted breast cancer cell growth. Maturitas 114:60–66. https://doi.org/10.1016/j.maturitas.2018.06.004
Ham J, Lee S, Lee H, Jeong D, Park S, Kim SJ (2018) Genome-wide methylation analysis identifies NOX4 and KDM5A as key regulators in inhibiting breast cancer cell proliferation by ginsenoside Rg3. Am J Chin Med 46(6):1333–1355. https://doi.org/10.1142/s0192415x18500702
Ham J, Jeong D, Park S, Kim HW, Kim H, Kim SJ (2019) Ginsenoside Rg3 and Korean red ginseng extract epigenetically regulate the tumor-related long noncoding RNAs RFX3-AS1 and STXBP5-AS1. J Ginseng Res 43(4):625–634. https://doi.org/10.1016/j.jgr.2019.02.004
Hong Y, Fan D (2019) Ginsenoside Rk1 induces cell cycle arrest and apoptosis in MDA-MB-231 triple negative breast cancer cells. Toxicology 418:22–31. https://doi.org/10.1016/j.tox.2019.02.010
Hong C, Wang D, Liang J, Guo Y, Zhu Y, **a J, Qin J, Zhan H, Wang J (2019) Novel ginsenoside-based multifunctional liposomal delivery system for combination therapy of gastric cancer. Theranostics 9(15):4437–4449. https://doi.org/10.7150/thno.34953
Hou JG, Jeon BM, Yun YJ, Cui CH, Kim SC (2019) Ginsenoside Rh2 ameliorates doxorubicin-induced senescence bystander effect in breast carcinoma cell MDA-MB-231 and normal epithelial cell MCF-10A. Int J Mol Sci. https://doi.org/10.3390/ijms20051244
Huynh DTN, Heo KS (2019) Therapeutic targets for endothelial dysfunction in vascular diseases. Arch Pharm Res 42(10):848–861. https://doi.org/10.1007/s12272-019-01180-7
Huynh DTN, ** Y, Myung CS, Heo KS (2020) Inhibition of p90RSK is critical to abolish Angiotensin II-induced rat aortic smooth muscle cell proliferation and migration. Biochem Biophys Res Commun 523(1):267–273. https://doi.org/10.1016/j.bbrc.2019.12.053
Jaenisch R, Bird A (2003) Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat Genet 33(Suppl):245–254. https://doi.org/10.1038/ng1089
Jeong D, Ham J, Park S, Kim HW, Kim H, Ji HW, Kim SJ (2019) Ginsenoside Rh2 suppresses breast cancer cell proliferation by epigenetically regulating the long noncoding RNA C3orf67-AS1. Am J Chin Med 47(7):1643–1658. https://doi.org/10.1142/s0192415x19500848
Ji X, Lu Y, Tian H, Meng X, Wei M, Cho WC (2019) Chemoresistance mechanisms of breast cancer and their countermeasures. Biomed Pharmacother 114:108800. https://doi.org/10.1016/j.biopha.2019.108800
Jimenez-Garduno AM, Mendoza-Rodriguez MG, Urrutia-Cabrera D, Dominguez-Robles MC, Perez-Yepez EA, Ayala-Sumuano JT, Meza I (2017) IL-1beta induced methylation of the estrogen receptor ERalpha gene correlates with EMT and chemoresistance in breast cancer cells. Biochem Biophys Res Commun 490(3):780–785. https://doi.org/10.1016/j.bbrc.2017.06.117
** Y, Heo K-S (2019) p90RSK activation promotes epithelial-mesenchymal transition in cisplatin-treated triple-negative breast cancer cells. J Bacteriol Virol 49:221. https://doi.org/10.4167/jbv.2019.49.4.221
** X, Yang C, Fan P, **ao J, Zhang W, Zhan S, Liu T, Wang D, Wu H (2017) CDK5/FBW7-dependent ubiquitination and degradation of EZH2 inhibits pancreatic cancer cell migration and invasion. J Biol Chem 292(15):6269–6280. https://doi.org/10.1074/jbc.M116.764407
** Y, Baek N, Back S, Myung C-S, Heo K-S (2018) Inhibitory effect of ginsenosides Rh1 and Rg2 on oxidative stress in LPS-stimulated RAW 264.7 cells. J Bacteriol Virol 48:156. https://doi.org/10.4167/jbv.2018.48.4.156
** Y, Huynh DTN, Kang KW, Myung CS, Heo KS (2019) Inhibition of p90RSK activation sensitizes triple-negative breast cancer cells to cisplatin by inhibiting proliferation, migration and EMT. BMB Rep 52(12):706–711
Jung J, Song DY, Hwang JJ, Park HJ, Lee JS, Song SY, Jeong SY, Choi EK (2019) Induction of p53-mediated senescence is essential for the eventual anticancer therapeutic effect of RH1. Arch Pharm Res 42(9):815–823. https://doi.org/10.1007/s12272-019-01132-1
Kang JH, Song KH, Woo JK, Park MH, Rhee MH, Choi C, Oh SH (2011) Ginsenoside Rp1 from Panax ginseng exhibits anti-cancer activity by down-regulation of the IGF-1R/Akt pathway in breast cancer cells. Plant Foods Hum Nutr 66(3):298–305. https://doi.org/10.1007/s11130-011-0242-4
Kim BJ (2013) Involvement of melastatin type transient receptor potential 7 channels in ginsenoside Rd-induced apoptosis in gastric and breast cancer cells. J Ginseng Res 37(2):201–209. https://doi.org/10.5142/jgr.2013.37.201
Kim DH (2018) Gut microbiota-mediated pharmacokinetics of ginseng saponins. J Ginseng Res 42(3):255–263. https://doi.org/10.1016/j.jgr.2017.04.011
Kim SJ, Kim AK (2015) Anti-breast cancer activity of fine black ginseng (panax ginseng meyer) and ginsenoside Rg5. J Ginseng Res 39(2):125–134. https://doi.org/10.1016/j.jgr.2014.09.003
Kim BM, Kim DH, Park JH, Surh YJ, Na HK (2014) Ginsenoside Rg3 inhibits constitutive activation of NF-kappaB signaling in human breast cancer (MDA-MB-231) cells: ERK and Akt as potential upstream targets. J Cancer Prev 19(1):23–30. https://doi.org/10.15430/jcp.2014.19.1.23
Kim H, Lee JH, Kim JE, Kim YS, Ryu CH, Lee HJ, Kim HM, Jeon H, Won HJ, Lee JY, Lee J (2018) Micro-/nano-sized delivery systems of ginsenosides for improved systemic bioavailability. J Ginseng Res 42(3):361–369. https://doi.org/10.1016/j.jgr.2017.12.003
Kim JH, Yi YS, Kim MY, Cho JY (2017) Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J Ginseng Res 41(4):435–443. https://doi.org/10.1016/j.jgr.2016.08.004
Kim YJ, Perumalsamy H, Castro-Aceituno V, Kim D, Markus J, Lee S, Kim S, Liu Y, Yang DC (2019) Photoluminescent and self-assembled hyaluronic acid-zinc oxide-ginsenoside Rh2 nanoparticles and their potential caspase-9 apoptotic mechanism towards cancer cell lines. Int J Nanomed 14:8195–8208. https://doi.org/10.2147/IJN.S221328
Klutstein M, Nejman D, Greenfield R, Cedar H (2016) DNA methylation in cancer and aging. Cancer Res 76(12):3446–3450. https://doi.org/10.1158/0008-5472.CAN-15-3278
Lee H, Lee S, Jeong D, Kim SJ (2018) Ginsenoside Rh2 epigenetically regulates cell-mediated immune pathway to inhibit proliferation of MCF-7 breast cancer cells. J Ginseng Res 42(4):455–462. https://doi.org/10.1016/j.jgr.2017.05.003
Lee JH, Ahn JY, Shin TJ, Choi SH, Lee BH, Hwang SH, Kang J, Kim HJ, Park CW, Nah SY (2011) Effects of minor ginsenosides, ginsenoside metabolites, and ginsenoside epimers on the growth of caenorhabditis elegans. J Ginseng Res 35(3):375–383. https://doi.org/10.5142/jgr.2011.35.3.375
Li J, Qi Y (2019) Ginsenoside Rg3 inhibits cell growth, migration and invasion in Caco-2 cells by downregulation of lncRNA CCAT1. Exp Mol Pathol 106:131–138. https://doi.org/10.1016/j.yexmp.2019.01.003
Li L, Wang Y, Qi B, Yuan D, Dong S, Guo D, Zhang C, Yu M (2014) Suppression of PMA-induced tumor cell invasion and migration by ginsenoside Rg1 via the inhibition of NF-kappaB-dependent MMP-9 expression. Oncol Rep 32(5):1779–1786. https://doi.org/10.3892/or.2014.3422
Li H, Huang N, Zhu W, Wu J, Yang X, Teng W, Tian J, Fang Z, Luo Y, Chen M, Li Y (2018a) Modulation the crosstalk between tumor-associated macrophages and non-small cell lung cancer to inhibit tumor migration and invasion by ginsenoside Rh2. BMC cancer 18(1):579. https://doi.org/10.1186/s12885-018-4299-4
Li Y, Hu K, **ao X, Wu W, Yan H, Chen H, Chen Z, Yin D (2018b) FBW7 suppresses cell proliferation and G2/M cell cycle transition via promoting gamma-catenin K63-linked ubiquitylation. Biochem Biophys Res Commun 497(2):473–479. https://doi.org/10.1016/j.bbrc.2018.01.192
Li C, Du L, Ren Y, Liu X, Jiao Q, Cui D, Wen M, Wang C, Wei G, Wang Y, Ji A, Wang Q (2019) SKP2 promotes breast cancer tumorigenesis and radiation tolerance through PDCD4 ubiquitination. J Exp Clin Cancer Res 38(1):76. https://doi.org/10.1186/s13046-019-1069-3
Liu Y, Fan D (2018) Ginsenoside Rg5 induces apoptosis and autophagy via the inhibition of the PI3K/Akt pathway against breast cancer in a mouse model. Food & function 9(11):5513–5527. https://doi.org/10.1039/c8fo01122b
Liu Y, Fan D (2020) The preparation of ginsenoside Rg5, its antitumor activity against breast cancer cells and its targeting of PI3K. Nutrients. https://doi.org/10.3390/nu12010246
Liu B, Cheng Y, Liu Q, Bao JK, Yang JM (2010) Autophagic pathways as new targets for cancer drug development. Acta Pharmacol Sin 31(9):1154–1164. https://doi.org/10.1038/aps.2010.118
Liu J, Shaik S, Dai X, Wu Q, Zhou X, Wang Z, Wei W (2015a) Targeting the ubiquitin pathway for cancer treatment. Biochim Biophys Acta 1855(1):50–60. https://doi.org/10.1016/j.bbcan.2014.11.005
Liu ZH, Li J, **a J, Jiang R, Zuo GW, Li XP, Chen Y, **ong W, Chen DL (2015b) Ginsenoside 20(s)-Rh2 as potent natural histone deacetylase inhibitors suppressing the growth of human leukemia cells. Chem Biol Interact 242:227–234. https://doi.org/10.1016/j.cbi.2015.10.014
Liu F, Ma F, Wang Y, Hao L, Zeng H, Jia C, Wang Y, Liu P, Ong IM, Li B, Chen G, Jiang J, Gong S, Li L, Xu W (2017a) PKM2 methylation by CARM1 activates aerobic glycolysis to promote tumorigenesis. Nat Cell Biol 19(11):1358–1370. https://doi.org/10.1038/ncb3630
Liu T, Zhao L, Hou H, Ding L, Chen W, Li X (2017b) Ginsenoside 20(S)-Rg3 suppresses ovarian cancer migration via hypoxia-inducible factor 1 alpha and nuclear factor-kappa B signals. Tumour Biol 39(5):1010428317692225. https://doi.org/10.1177/1010428317692225
Lu G, Li Y, Ma Y, Lu J, Chen Y, Jiang Q, Qin Q, Zhao L, Huang Q, Luo Z, Huang S, Wei Z (2018) Long noncoding RNA LINC00511 contributes to breast cancer tumourigenesis and stemness by inducing the miR-185-3p/E2F1/Nanog axis. J Exp Clin Cancer Res 37(1):289. https://doi.org/10.1186/s13046-018-0945-6
Luo Y, Wang X, Niu W, Wang H, Wen Q, Fan S, Zhao R, Li Z, **ong W, Peng S, Zeng Z, Li X, Li G, Tan M, Zhou M (2017) Elevated microRNA-125b levels predict a worse prognosis in HER2-positive breast cancer patients. Oncol Lett 13(2):867–874. https://doi.org/10.3892/ol.2016.5482
Mai TT, Moon J, Song Y, Viet PQ, Phuc PV, Lee JM, Yi TH, Cho M, Cho SK (2012) Ginsenoside F2 induces apoptosis accompanied by protective autophagy in breast cancer stem cells. Cancer Lett 321(2):144–153. https://doi.org/10.1016/j.canlet.2012.01.045
Malicka I, Hanuszkiewicz J, Wozniewski M (2019) Barriers of physical activity of women post breast cancer treatment in rural Poland. Health Care Women Int 40(6):682–695. https://doi.org/10.1080/07399332.2019.1608208
Mathiyalagan R, Wang C, Kim YJ, Castro-Aceituno V, Ahn S, Subramaniyam S, Simu SY, Jimenez-Perez ZE, Yang DC, Jung SK (2019) Preparation of polyethylene glycol-ginsenoside Rh1 and Rh2 conjugates and their efficacy against lung cancer and inflammation. Molecules. https://doi.org/10.3390/molecules24234367
McGuire A, Brown JA, Kerin MJ (2015) Metastatic breast cancer: the potential of miRNA for diagnosis and treatment monitoring. Cancer Metastasis Rev 34(1):145–155. https://doi.org/10.1007/s10555-015-9551-7
Mekala JR, Naushad SM, Ponnusamy L, Arivazhagan G, Sakthiprasad V, Pal-Bhadra M (2018) Epigenetic regulation of miR-200 as the potential strategy for the therapy against triple-negative breast cancer. Gene 641:248–258. https://doi.org/10.1016/j.gene.2017.10.018
Morrow JK, Lin HK, Sun SC, Zhang S (2015) Targeting ubiquitination for cancer therapies. Future Med Chem 7(17):2333–2350. https://doi.org/10.4155/fmc.15.148
Nakhjavani M, Palethorpe HM, Tomita Y, Smith E, Price TJ, Yool AJ, Pei JV, Townsend AR, Hardingham JE (2019) Stereoselective anti-cancer activities of ginsenoside Rg3 on triple negative breast cancer cell models. Pharmaceuticals (Basel, Switzerland). https://doi.org/10.3390/ph12030117
Nosrati H, Sefidi N, Sharafi A, Danafar H, Kheiri Manjili H (2018) Bovine serum albumin (BSA) coated iron oxide magnetic nanoparticles as biocompatible carriers for curcumin-anticancer drug. Bioorg Chem 76:501–509. https://doi.org/10.1016/j.bioorg.2017.12.033
Omarini C, Guaitoli G, Pipitone S, Moscetti L, Cortesi L, Cascinu S, Piacentini F (2018) Neoadjuvant treatments in triple-negative breast cancer patients: where we are now and where we are going. Cancer Manag Res 10:91–103. https://doi.org/10.2147/CMAR.S146658
Otto T, Sicinski P (2017) Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer 17(2):93–115. https://doi.org/10.1038/nrc.2016.138
Peng B, He R, Xu Q, Yang Y, Hu Q, Hou H, Liu X, Li J (2019) Ginsenoside 20(S)-protopanaxadiol inhibits triple-negative breast cancer metastasis in vivo by targeting EGFR-mediated MAPK pathway. Pharmacol Res 142:1–13. https://doi.org/10.1016/j.phrs.2019.02.003
Pokharel YR, Kim ND, Han HK, Oh WK, Kang KW (2010) Increased ubiquitination of multidrug resistance 1 by ginsenoside Rd. Nutr Cancer 62(2):252–259. https://doi.org/10.1080/01635580903407171
Qiu R, Qian F, Wang X, Li H, Wang L (2019) Targeted delivery of 20(S)-ginsenoside Rg3-based polypeptide nanoparticles to treat colon cancer. Biomed Microdevices 21(1):18. https://doi.org/10.1007/s10544-019-0374-0
Reinbolt RE, Mangini N, Hill JL, Levine LB, Dempsey JL, Singaravelu J, Koehler KA, Talley A, Lustberg MB (2015) Endocrine therapy in breast cancer: the neoadjuvant, adjuvant, and metastatic approach. Semin Oncol Nurs 31(2):146–155. https://doi.org/10.1016/j.soncn.2015.02.002
Ren Z, Chen X, Hong L, Zhao X, Cui G, Li A, Liu Y, Zhou L, Sun R, Shen S, Li J, Lou J, Zhou H, Wang J, Xu G, Yu Z, Song Y, Chen X (2020) Nanoparticle conjugation of ginsenoside Rg3 inhibits hepatocellular carcinoma development and metastasis. Small 16(2):e1905233. https://doi.org/10.1002/smll.201905233
Schwartz GK, Shah MA (2005) Targeting the cell cycle: a new approach to cancer therapy. J Clin Oncol 23(36):9408–9421. https://doi.org/10.1200/JCO.2005.01.5594
Shi Y, Su C, Cui W, Li H, Liu L, Feng B, Liu M, Su R, Zhao L (2014) Gefitinib loaded folate decorated bovine serum albumin conjugated carboxymethyl-beta-cyclodextrin nanoparticles enhance drug delivery and attenuate autophagy in folate receptor-positive cancer cells. J Nanobiotechnology 12:43. https://doi.org/10.1186/s12951-014-0043-7
Shi Y, Gong W, Lu L, Wang Y, Ren J (2019) Upregulation of miR-129-5p increases the sensitivity to Taxol through inhibiting HMGB1-mediated cell autophagy in breast cancer MCF-7 cells. Braz J Med Biol Res 52(11):e8657. https://doi.org/10.1590/1414-431X20198657
Shin SA, Moon SY, Park D, Park JB, Lee CS (2019) Apoptotic cell clearance in the tumor microenvironment: a potential cancer therapeutic target. Arch Pharm Res 42(8):658–671. https://doi.org/10.1007/s12272-019-01169-2
Tang YC, Zhang Y, Zhou J, Zhi Q, Wu MY, Gong FR, Shen M, Liu L, Tao M, Shen B, Gu DM, Yu J, Xu MD, Gao Y, Li W (2018) Ginsenoside Rg3 targets cancer stem cells and tumor angiogenesis to inhibit colorectal cancer progression in vivo. Int J Oncol 52(1):127–138. https://doi.org/10.3892/ijo.2017.4183
Toh TB, Lim JJ, Chow EK (2017) Epigenetics in cancer stem cells. Mol Cancer 16(1):29. https://doi.org/10.1186/s12943-017-0596-9
Wu TTL, Tong YC, Chen IH, Niu HS, Li Y, Cheng JT (2018) Induction of apoptosis in prostate cancer by ginsenoside Rh2. Oncotarget 9(13):11109–11118. https://doi.org/10.18632/oncotarget.24326
Waks AG, Winer EP (2019) Breast cancer treatment: a review. JAMA 321(3):288–300. https://doi.org/10.1001/jama.2018.19323
Wang W, Zhang X, Qin JJ, Voruganti S, Nag SA, Wang MH, Wang H, Zhang R (2012) Natural product ginsenoside 25-OCH3-PPD inhibits breast cancer growth and metastasis through down-regulating MDM2. PLoS ONE 7(7):e41586. https://doi.org/10.1371/journal.pone.0041586
Wang B, Zhang Y, Mao Z, Yu D, Gao C (2014) Toxicity of ZnO nanoparticles to macrophages due to cell uptake and intracellular release of zinc ions. J Nanosci Nanotechnol 14(8):5688–5696. https://doi.org/10.1166/jnn.2014.8876
Wang P, Du X, **ong M, Cui J, Yang Q, Wang W, Chen Y, Zhang T (2016) Ginsenoside Rd attenuates breast cancer metastasis implicating derepressing microRNA-18a-regulated Smad2 expression. Sci Rep 6:33709. https://doi.org/10.1038/srep33709
Wang RX, Chen S, Huang L, Zhou Y, Shao ZM (2019) Monitoring serum VEGF in neoadjuvant chemotherapy for patients with triple-negative breast cancer: a new strategy for early prediction of treatment response and patient survival. Oncologist 24(6):753–761. https://doi.org/10.1634/theoncologist.2017-0602
Wen X, Zhang HD, Zhao L, Yao YF, Zhao JH, Tang JH (2015) Ginsenoside Rh2 differentially mediates microRNA expression to prevent chemoresistance of breast cancer. Asian Pac J Cancer Prev 16(3):1105–1109. https://doi.org/10.7314/apjcp.2015.16.3.1105
**ng Z, Zhang Y, Liang K, Yan L, **ang Y, Li C, Hu Q, ** F, Putluri V, Putluri N, Coarfa C, Sreekumar A, Park PK, Nguyen TK, Wang S, Zhou J, Zhou Y, Marks JR, Hawke DH, Hung MC, Yang L, Han L, Ying H, Lin C (2018) Expression of long noncoding RNA YIYA promotes glycolysis in breast cancer. Cancer Res 78(16):4524–4532. https://doi.org/10.1158/0008-5472.CAN-17-0385
Yang JY, Zong CS, **a W, Wei Y, Ali-Seyed M, Li Z, Broglio K, Berry DA, Hung MC (2006) MDM2 promotes cell motility and invasiveness by regulating E-cadherin degradation. Mol Cell Biol 26(19):7269–7282. https://doi.org/10.1128/MCB.00172-06
Yang J, Zeng Z, Peng Y, Chen J, Pan L, Pan D (2014a) IL-7 splicing variant IL-7delta5 induces EMT and metastasis of human breast cancer cell lines MCF-7 and BT-20 through activation of PI3K/Akt pathway. Histochem Cell Biol 142(4):401–410. https://doi.org/10.1007/s00418-014-1222-1
Yang R, An Y, Miao F, Li M, Liu P, Tang Q (2014b) Preparation of folic acid-conjugated, doxorubicin-loaded, magnetic bovine serum albumin nanospheres and their antitumor effects in vitro and in vivo. Int J Nanomed 9:4231–4243. https://doi.org/10.2147/IJN.S67210
Yang J, Yuan D, **ng T, Su H, Zhang S, Wen J, Bai Q, Dang D (2016) Ginsenoside Rh2 inhibiting HCT116 colon cancer cell proliferation through blocking PDZ-binding kinase/T-LAK cell-originated protein kinase. J Ginseng Res 40(4):400–408. https://doi.org/10.1016/j.jgr.2016.03.007
Yang X, Hu Q, Hu LX, Lin XR, Liu JQ, Lin X, Dinglin XX, Zeng JY, Hu H, Luo ML, Yao HR (2017) miR-200b regulates epithelial-mesenchymal transition of chemo-resistant breast cancer cells by targeting FN1. Discov Med 24(131):75–85
Yu X, Xu H, Hu M, Luan X, Wang K, Fu Y, Zhang D, Li J (2015) Ginsenoside Rg3 bile salt-phosphatidylcholine-based mixed micelles: design, characterization, and evaluation. Chem Pharm Bull (Tokyo) 63(5):361–368. https://doi.org/10.1248/cpb.c15-00045
Yu L, Dong L, Wang Y, Liu L, Long H, Li H, Li J, Yang X, Liu Z, Duan G, Dai X, Lin Z (2019) Reversible regulation of SATB1 ubiquitination by USP47 and SMURF2 mediates colon cancer cell proliferation and tumor progression. Cancer Lett 448:40–51. https://doi.org/10.1016/j.canlet.2019.01.039
Yuan Z, Jiang H, Zhu X, Liu X, Li J (2017) Ginsenoside Rg3 promotes cytotoxicity of Paclitaxel through inhibiting NF-κB signaling and regulating Bax/Bcl-2 expression on triple-negative breast cancer. Biomed Pharmacother 89:227–232. https://doi.org/10.1016/j.biopha.2017.02.038
Zare-Zardini H, Taheri-Kafrani A, Amiri A, Bordbar AK (2018) New generation of drug delivery systems based on ginsenoside Rh2-, Lysine- and Arginine-treated highly porous graphene for improving anticancer activity. Sci Rep 8(1):586. https://doi.org/10.1038/s41598-017-18938-y
Zhang K, Li Y (2016) Effects of ginsenoside compound K combined with cisplatin on the proliferation, apoptosis and epithelial mesenchymal transition in MCF-7 cells of human breast cancer. Pharm Biol 54(4):561–568. https://doi.org/10.3109/13880209.2015.1101142
Zhang J, Lu M, Zhou F, Sun H, Hao G, Wu X, Wang G (2012) Key role of nuclear factor-κB in the cellular pharmacokinetics of adriamycin in MCF-7/Adr cells: the potential mechanism for synergy with 20(S)-ginsenoside Rh2. Drug Metab Dispos 40(10):1900–1908. https://doi.org/10.1124/dmd.112.045187
Zhang Y, Liu QZ, **ng SP, Zhang JL (2016) Inhibiting effect of Endostar combined with ginsenoside Rg3 on breast cancer tumor growth in tumor-bearing mice. Asian Pac J Trop Med 9(2):180–183. https://doi.org/10.1016/j.apjtm.2016.01.010
Zhang E, Shi H, Yang L, Wu X, Wang Z (2017) Ginsenoside Rd regulates the Akt/mTOR/p70S6K signaling cascade and suppresses angiogenesis and breast tumor growth. Oncol Rep 38(1):359–367. https://doi.org/10.3892/or.2017.5652
Zhang J, Jiang Y, Li Y, Li W, Zhou J, Chen J, Shang Z, Gu Q, Wang W, Shen T, Hu W (2020) Micelles modified with a chitosan-derived homing peptide for targeted intracellular delivery of ginsenoside compound K to liver cancer cells. Carbohydr Polym 230:115576. https://doi.org/10.1016/j.carbpol.2019.115576
Zhao HG, Zhou SL, Lin YY, Wang H, Dai HF, Huang FY (2018) Autophagy plays a protective role against apoptosis induced by toxicarioside N via the Akt/mTOR pathway in human gastric cancer SGC-7901 cells. Arch Pharm Res 41(10):986–994. https://doi.org/10.1007/s12272-018-1049-8
Zhao L, Shou H, Chen L, Gao W, Fang C, Zhang P (2019) Effects of ginsenoside Rg3 on epigenetic modification in ovarian cancer cells. Oncol Rep 41(6):3209–3218. https://doi.org/10.3892/or.2019.7115
Zhu Y, Li L, Hou D, Ouyang Y, Guo X, Wang Y, Li J, Gong K (2019) MicroRNA-19a regulates the proliferation, migration and invasion of human gastric cancer cells by targeting CUL5. Arch Biochem Biophys 662:93–100. https://doi.org/10.1016/j.abb.2018.11.023
Zou J, Su H, Zou C, Liang X, Fei Z (2020) Ginsenoside Rg3 suppresses the growth of gemcitabine-resistant pancreatic cancer cells by upregulating lncRNA-CASC2 and activating PTEN signaling. J Biochem Mol Toxicol. https://doi.org/10.1002/jbt.22480
Acknowledgements
This research was supported by National Research Foundation of Korea (KNRF-2016232004 and -2019025901).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declares that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
**, Y., Huynh, D.T.N., Nguyen, T.L.L. et al. Therapeutic effects of ginsenosides on breast cancer growth and metastasis. Arch. Pharm. Res. 43, 773–787 (2020). https://doi.org/10.1007/s12272-020-01265-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01265-8