Abstract
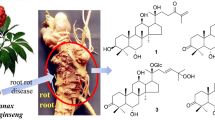
Phytochemical investigation of the ethanol extract of the roots of Sanguisorba officinalis resulted in the isolation of three new triterpene glycosides, 3β-[(α-L-arabinopyranosyl)oxy]-19α,23-dihydroxyolean-12-en-28-oic acid 28-[6-O-acetyl-β-D-glucopyranosyl] ester (1), 2α,3β,19α,23-tetrahydroxyurs-12-en-28-oic acid 28-[6-O-acetyl-β-D-glucopyranosyl] ester (2), and 3β-[(α-L-arabinopyranosyl)oxy]-19α-hydroxyurs-12,20(30)-dien-28-oic acid 28-[6-O-acetyl-β-D-glucopyranosyl] ester (3). All the triterpene glycosides exhibited the significant cytotoxic potential with low IC50 values (IC50 < 5.0 μM) against six tumor cell lines (MCF-7, HeLa, HepG2, SGC-7901, NCI-H460, and BGC-823).


Similar content being viewed by others
References
Chen, J.J., H.C. Hung, P.J. Sung, I.S. Chen, and W.L. Kuo. 2011. Aporphine alkaloids and cytotoxic lignans from the roots of Illigera luzonensis. Phytochemistry 72: 523–532.
Cheng, D.L., and X.P. Cao. 1992. Pomolic acid derivatives from the root of Sanguisorba officinalis. Phytochemistry 31: 1317–1320.
Hara, S., H. Okabe, and K. Mihashi. 1987. Gas-Liquid chromatographic separation of aldose enantiomers as Trimethylsilyl ethers of methyl 2-(Polyhydroxyalkyl)-thiazolidine-4 (R)-carboxylates. Chemical & Pharmaceutical Bulletin 35: 501–506.
Liang, J.G., J.P. Chen, Z.W. Tan, J. Peng, X. Zheng, K.J. Nishiura, J. Ng, Z.Y. Wang, D.M. Wang, Z.W. Chen, and L. Liu. 2013. Extracts of the medicinal herb Sanguisorba officinalis inhibit the entry of human immunodeficiency virus-1. Journal of Food and Drug Analysis 21: S52–S58.
Liu, X., B.F. Shi, and B. Yu. 2004. Four new dimeric triterpene glucosides from Sanguisorba officinalis. Tetrahedron 60: 11647–11654.
Liu, X., Y. Cui, Q. Yu, and B. Yu. 2005. Triterpenoids from Sanguisorba officinalis. Phytochemistry 66: 1671–1679.
Mimaki, Y., M. Fukushima, A. Yokosuka, Y. Sashida, S. Furuya, and H. Sakagami. 2001. Triterpene glycosides from the roots of Sanguisorba officinalis. Phytochemistry 57: 773–779.
Shu, J.C., J.Q. Liu, G.X. Chou, and Z.T. Wang. 2012. Two new triterpenoids from Psidium guajava. Chinese Chemical Letters 23: 827–830.
Thuong, P.T., C.H. Lee, T.T. Dao, P.H. Nguyen, W.G. Kim, S.J. Lee, and W.K. Oh. 2008. Triterpenoids from the leaves of Diospyros kaki (Persimmon) and their inhibitory effects on protein tyrosine phosphatase 1B. Journal of Natural Products 71: 1775–1778.
Yu, T., Y.J. Lee, H.M. Yang, S. Han, J.H. Kim, Y. Lee, C. Kim, M.H. Han, M.Y. Kim, J. Lee, and J.Y. Cho. 2011. Inhibitory effect of Sanguisorba officinalis ethanol extract on NO and PGE2 production is mediated by suppression of NF-κB and AP-1 activation signaling cascade. Journal of Ethnopharmacology 134: 11–17.
Zhang, L., S.R. Koyyalamudi, S.C. Jeong, N. Reddy, P.T. Smith, R. Ananthan, and T. Longvah. 2012a. Antioxidant and immunomodulatory activities of polysaccharides from the roots of Sanguisorba officinalis. International Journal of Biological Macromolecules 51: 1057–1062.
Zhang, L.J., J.J. Cheng, C.C. Liao, H.L. Cheng, H.T. Huang, L.M.Y. Kuo, and Y.H. Kuo. 2012b. Triterpene acids from Euscaphis japonica and assessment of their cytotoxic and anti-NO activities. Planta Medica 78: 1584–1590.
Zhang, Y., Y.J. Zhang, M.R. Jacob, X.C. Li, and C.R. Yang. 2008. Steroidal saponins from the stem of Yucca elephantipes. Phytochemistry 69: 264–270.
Acknowledgments
The above research was made possible by the grant from the Scientific Planning Project of the Applied Basic Research of Yunnan Province (S2012FZ0005) and the Develo** Key Subject of Ecology of Qu**g Normal University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jiang Hu and Yan Song have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Hu, J., Song, Y., Li, H. et al. Cytotoxic triterpene glycosides from the roots of Sanguisorba officinalis . Arch. Pharm. Res. 38, 984–990 (2015). https://doi.org/10.1007/s12272-014-0477-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-014-0477-3