Abstract
Purpose
The present work was intended to develop the ibuprofen-Kollicoat® MAE 100P delayed-release pellets using hot-melt extrusion technology, which exhibits pH-dependent solubility. Ibuprofen irritates the gastric lining, so its release in the gastric environment is not desired. Conventionally, Kollicoat® MAE 100P has been used as an enteric coating polymer, and we have explored its application using hot-melt extrusion technology in our work.
Methods
Three different drug loadings were processed at various extrusion temperatures using HME to produce pellets of uniform size. DSC was performed to study the drug’s state, the polymer’s thermal behavior, and drug-polymer miscibility. An in vitro drug release study was performed in 0.1N HCl followed by pH 6.8 phosphate buffer to understand the ability of the polymer to impede the release of ibuprofen in the stomach. Furthermore, the lead batch was characterized by DSC, FTIR, HS-PLM, and optical microscopy to study the interaction between the drug and polymer.
Results
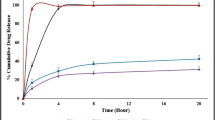
The thermogram of the pellets indicated no drug-polymer immiscibility. This work also demonstrates proof of the plasticizing ability of ibuprofen. Drug release studies showed less than 1.5% drug release in 0.1N HCl in 2 h, and complete drug release was obtained in the next 2 h in pH 6.8 phosphate buffer, indicating the delayed-release characteristics of the pellets.
Conclusion
This work proves that Kollicoat® MAE 100P could be used in modified-release dosage forms to attain the delayed-release pellets.
Graphical Abstract
Schematic representation of development of ibuprofen delayed release pellets








Similar content being viewed by others
References
Liu F, et al. Patient-centered pharmaceutical design to improve acceptability of medicines: similarities and differences in paediatric and geriatric populations. Drugs. 2014;74:1871–89.
Perico N, et al. Home as the new frontier for the treatment of COVID-19: the case for anti-inflammatory agents. Lancet Infect Dis. 2022.
Nagaich U, Chaudhary V. Nonsteroidal anti-inflammatory drugs: concepts and innovations. In: Biologically Active Small Molecules. Apple Academic Press; 2023. p. 165–244.
Vemula SK. Formulation and pharmacokinetics of colon-specific double-compression coated mini-tablets: Chronopharmaceutical delivery of ketorolac tromethamine. Int J Pharm. 2015;491(1–2):35–41.
Debotton N, Dahan A. Applications of polymers as pharmaceutical excipients in solid oral dosage forms. Med Res Rev. 2017;37(1):52–97.
Prabhakar RV, Sateesh KV. Formulation, evaluation and pharmacokinetics of colon targeted pulsatile system of flurbiprofen. J Drug Target. 2012;20(8):703–14.
Nakkala RK, et al. Duloxetine hydrochloride enteric-coated pellets in capsules with delayed release: formulation and evaluation. Smart Science; 2023. p. 1–13.
Vemula SK, Veerareddy PR. Development, evaluation and pharmacokinetics of time-dependent ketorolac tromethamine tablets. Expert Opin Drug Deliv. 2013;10(1):33–45.
Khan Z, et al. Recent advances in microbeads-based drug delivery system for achieving controlled drug release. J Biomater Sci Polym Ed. 2023;34(4):541–64.
** fast release pellets via wet extrusion-spheronization. Pharm Dev Technol. 2018;23(5):432–41.
Narala S, et al. Preparation and in vitro evaluation of hot-melt extruded pectin-based pellets containing ketoprofen for colon targeting. Int J Pharm: X. 2023;5.
Nambiar AG, et al. Continuous manufacturing and molecular modeling of pharmaceutical amorphous solid dispersions. AAPS PharmSciTech. 2022;23(7):249.
Crowley MM, et al. Pharmaceutical applications of hot-melt extrusion: part I. Drug Dev Ind Pharm. 2007;33(9):909–26.
Tiwari RV, Patil H, Repka MA. Contribution of hot-melt extrusion technology to advance drug delivery in the 21st century. Expert Opin Drug Deliv. 2016;13(3):451–64.
Karnik I, et al. Formulation development and characterization of dual drug loaded hot-melt extruded inserts for better ocular therapeutic outcomes: sulfacetamide/prednisolone. J Drug Deliv Sci Technol. 2023;84.
Repka MA, Langley N, DiNunzio J. Melt extrusion: materials, technology and drug product design. 2013; 4(5).
Srinivasan P, et al. Numerical simulation of five different screw configurations used during the preparation of hot-melt extruded Kollidon® and Soluplus® based amorphous solid dispersions containing indomethacin. J Drug Deliv Sci Technol. 2023;85.
Akay S, et al. Degradation, solubility and chromatographic studies of Ibuprofen under high temperature water conditions. Chemosphere. 2021;277.
Wu C-Y, Benet LZ. Predicting drug disposition via application of BCS: transport/absorption/elimination interplay and development of a biopharmaceutics drug disposition classification system. Pharm Res. 2005;22(1):11–23.
Czyrski A. Determination of the lipophilicity of ibuprofen, naproxen, ketoprofen, and flurbiprofen with thin-layer chromatography. J Chem. 2019.
Man AM, et al. Ibuprofen-associated hypokalemia and metabolic acidosis: systematic literature review. Ann Pharmacother. 2022;56(11):1250–7.
Kushner P, et al. The use of non-steroidal anti-inflammatory drugs (NSAIDs) in COVID-19. NPJ Primary Care Respiratory Medicine. 2022;32(1):35.
de Lima CAA, et al. Gastroprotective mechanisms. Peptic Ulcer Disease-What’s New; 2022.
Forssell H. Gastric mucosal defence mechanisms: a brief review. Scand J Gastroenterol. 1988;23(sup155):23–8.
Fini A, et al. Fast dispersible/slow releasing ibuprofen tablets. Eur J Pharm Biopharm. 2008;69(1):335–41.
Bjarnason I, et al. Mechanisms of damage to the gastrointestinal tract from nonsteroidal anti-inflammatory drugs. Gastroenterology. 2018;154(3):500–14.
Russell RI. Protective effects of the prostaglandins on the gastric mucosa. Am J Med. 1986;81(2):2–4.
Goldstein JL, Cryer B. Gastrointestinal injury associated with NSAID use: a case study and review of risk factors and preventative strategies. Drug, healthcare and patient safety. 2015;7:31.
Domper Arnal M-J, Hijos-Mallada G, Lanas A. Gastrointestinal and cardiovascular adverse events associated with NSAIDs. Expert Opin Drug Saf. 2022;21(3):373–84.
Sostres C, Gargallo CJ, Lanas A. Nonsteroidal anti-inflammatory drugs and upper and lower gastrointestinal mucosal damage. Arthritis Res Ther. 2013;15(3):1–8.
Bühler V. Kollicoat grades: functional polymers for the pharmaceutical industry. BASF; 2007.
Medarević D, et al. Analytical and computational methods for the estimation of drug-polymer solubility and miscibility in solid dispersions development. Pharmaceutics. 2019;11(8):372.
Martinez-Marcos L, et al. A novel hot-melt extrusion formulation of albendazole for increasing dissolution properties. Int J Pharm. 2016;499(1–2):175–85.
Bottom R. Thermogravimetric analysis. Principles and applications of thermal analysis. 2008;1:87–118.
Gabbott P. Principles and applications of thermal analysis. John Wiley & Sons; 2008.
Ghebre-Selassie I. Pharmaceutical pelletization technology. CRC Press; 2022.
Ghebre-Sellassie I, et al. Pharmaceutical extrusion technology. CRC Press; 2003.
Kolter K, et al. Hot-melt extrusion with BASF pharma polymers: extrusion compendium. BASF; 2012.
Thakkar R, et al. Systematic screening of pharmaceutical polymers for hot melt extrusion processing: a comprehensive review. Int J Pharm. 2020;576.
De Brabander C, et al. Characterization of ibuprofen as a nontraditional plasticizer of ethyl cellulose. J Pharm Sci. 2002;91(7):1678–85.
Wu C, McGinity JW. Non-traditional plasticization of polymeric films. Int J Pharm. 1999;177(1):15–27.
Thumma S, et al. Influence of plasticizers on the stability and release of a prodrug of Δ9-tetrahydrocannabinol incorporated in poly (ethylene oxide) matrices. Eur J Pharm Biopharm. 2008;70(2):605–14.
Wu C, McGinity JW. Influence of an enteric polymer on drug release rates of theophylline from pellets coated with Eudragit® RS 30D. Pharm Dev Technol. 2003;8(1):103–10.
Chan S-Y, Qi S, Craig DQ. An investigation into the influence of drug–polymer interactions on the miscibility, processability and structure of polyvinylpyrrolidone-based hot melt extrusion formulations. Int J Pharm. 2015;496(1):95–106.
Nollenberger K, Albers J. Poly (meth) acrylate-based coatings. Int J Pharm. 2013;457(2):461–9.
Kim NA, et al. Investigation of polymeric excipients for dutasteride solid dispersion and its physicochemical characterization. Arch Pharmacal Res. 2014;37(2):214–24.
Lee SY, Yu G, Kim IW. Effects of polymeric additives on the crystallization and release behavior of amorphous ibuprofen. J Nanomater. 2013.
Ryabenkova Y, et al. Mechanism of hydrogen-bonded complex formation between ibuprofen and nanocrystalline hydroxyapatite. Langmuir. 2017;33(12):2965–76.
Shan X, et al. Mutual effects of hydrogen bonding and polymer hydrophobicity on ibuprofen crystal inhibition in solid dispersions with poly (N-vinyl pyrrolidone) and poly (2-oxazolines). Pharmaceutics. 2021;13(5):659.
Lu J, et al. Understanding the processing window of hypromellose acetate succinate for hot-melt extrusion, part I: polymer characterization and hot-melt extrusion. Adv Polym Technol. 2018;37(1):154–66.
Wong RSH, Dodou K. Effect of drug loading method and drug physicochemical properties on the material and drug release properties of poly (ethylene oxide) hydrogels for transdermal delivery. Polymers. 2017;9(7):286.
Zhang J, et al. Hydroxypropyl methylcellulose-based controlled release dosage by melt extrusion and 3D printing: structure and drug release correlation. Carbohyd Polym. 2017;177:49–57.
Hancock BC, Zografi G. The relationship between the glass transition temperature and the water content of amorphous pharmaceutical solids. Pharm Res. 1994;11(4):471–7.
Acknowledgements
We are deeply grateful to the late Dr. Suresh Bandari for his contributions to this research. He provided instrumental guidance to the project at crucial steps and helped to move it forward.
Funding
This project was partially supported by the National Institute of General Medical Sciences (NIGMS; Grant Number P30GM122733), which is a component of the National Institutes of Health (NIH) serving as one of its Centers of Biomedical Research Excellence (COBRE).
Author information
Authors and Affiliations
Contributions
Mittal Darji: design experiment, acquire data, analyze data, and interpret data, manuscript writing. Adwait Pradhan: acquire data, analyze data, and interpret data. K. Kolter and Nigel Langley: funding acquisition, resources. Sateesh Kumar Vemula and Michael A. Repka: interpret data, and draft manuscript. Michael A. Repka: supervise the study.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Darji, M., Pradhan, A., Vemula, S.K. et al. Development of Delayed-Release Pellets of Ibuprofen Using Kollicoat® MAE 100P via Hot-Melt Extrusion Technology. J Pharm Innov 18, 1827–1837 (2023). https://doi.org/10.1007/s12247-023-09758-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-023-09758-x