Abstract
Objective
Our objective was to correlate staging PSMA PET imaging parameters to final histopathology. Second objective was to assess the performance of standard versus delayed PSMA PET to detect primary prostate tumor.
Methods
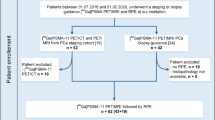
Thirty-one patients (mean age, 61.4 ± 8.2) who underwent radical prostatectomy and preoperative staging PSMA PET scans were included in the study. After defining dominant lesion in pathology, correlations with PET images were performed. Additionally, two physicians blind to clinical and pathological information retrospectively reviewed staging Ga-68 PSMA PET scans with standard and delayed imaging.
Results
Dominant lesion SUV’s increased with time 8.2(± 4.5), 10(± 7.1), and 10.2(± 7.8) at 1, 2, and 3 h (P = .03 T1-T3). WHO Grade group 3 had highest SUV (group 3 11.9 ± 5.6 vs. group 2 7.9 ± 1.5, p = .02). Addition of cribriform pattern on intraductal component was associated with higher SUV’s (11 ± 2.9 vs. 6.5 ± 2.1, p = .01) and higher Gleason four ratios (64 ± 9% vs. 37 ± 17%, p = .01). Intraductal carcinoma was associated with larger tumors (6.3 ± 2.3 cm3 vs. 2.6 ± 1.7 cm3, p < .001). Physician sensitivities ranged from 61 to 81%. Excluding Gleason 3 + 3 lesions and small lesions (< 1 cm3), sensitivities increased to 80–100%. Differences of sensitivity between different time points were not significant. Combined evaluation of all time points did not increase sensitivity.
Conclusions
Cribriform pattern correlates with higher Gleason 4 ratios and SUVs in PSMA PET. Intraductal carcinoma is associated with larger tumors but not higher Gleason 4 ratios and SUVs. Multiple late imaging times did not enhance tumor detection and may pose tolerability issues for some patients.





Similar content being viewed by others
References
Elsässer-Beile U, Reischl G, Wiehr S, Bühler P, Wolf P, Alt K, et al. PET imaging of prostate cancer xenografts with a highly specific antibody against the prostate-specific membrane antigen. J Nucl Med. 2009;50(4):606–11.
NCCN Guidelines Version 4.2022 Prostate cancer 2022. Available from: http://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf.
Derlin T, Weiberg D, von Klot C, Wester HJ, Henkenberens C, Ross TL, et al. (68) Ga-PSMA I & T PET/CT for assessment of prostate cancer: evaluation of image quality after forced diuresis and delayed imaging. Eur Radiol. 2016;26(12):4345–53.
Wen J, Zhu Y, Li L, Liu J, Chen Y, Chen R. Determination of optimal (68) Ga-PSMA PET/CT imaging time in prostate cancers by total-body dynamic PET/CT. Eur J Nucl Med Mol Imaging. 2022;49(6):2086–95.
Afshar-Oromieh A, Eiber M, Fendler W, Schmidt M, Rahbar K, Ahmadzadehfar H, et al. Procedure guideline for prostate cancer imaging with PSMA-ligand PET/CT. Nuklearmedizin. 2023;62(1):5–19.
Alberts I, Sachpekidis C, Gourni E, Boxler S, Gross T, Thalmann G, et al. Dynamic patterns of [(68) Ga] Ga-PSMA-11 uptake in recurrent prostate cancer lesions. Eur J Nucl Med Mol Imaging. 2020;47(1):160–7.
Alberts I, Niklas-Hünermund J, Sachpekidis C, Zacho HD, Mingels C, Dijkstra L, et al. Combination of forced diuresis with additional late imaging in (68) Ga-PSMA-11 PET/CT: effects on lesion visibility and radiotracer uptake. J Nucl Med. 2021;62(9):1252–7.
Kweldam CF, Kümmerlin IP, Nieboer D, Verhoef EI, Steyerberg EW, van der Kwast TH, et al. Disease-specific survival of patients with invasive cribriform and intraductal prostate cancer at diagnostic biopsy. Mod Pathol. 2016;29(6):630–6.
Trinh VQ, Sirois J, Benzerdjeb N, Mansoori BK, Grosset AA, Albadine R, et al. The impact of intraductal carcinoma of the prostate on the site and timing of recurrence and cancer-specific survival. Prostate. 2018;78(10):697–706.
Marra G, van Leenders G, Zattoni F, Kesch C, Rajwa P, Cornford P, et al. Impact of epithelial histological types, subtypes, and growth patterns on oncological outcomes for patients with nonmetastatic prostate cancer treated with curative intent: a systematic review. Eur Urol. 2023.
Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, et al. (68) Ga-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44(6):1014–24.
Afshar-Oromieh A, Sattler LP, Mier W, Hadaschik BA, Debus J, Holland-Letz T, et al. The clinical impact of additional late PET/CT imaging with (68)Ga-PSMA-11 (HBED-CC) in the diagnosis of prostate cancer. J Nucl Med. 2017;58(5):750–5.
Ponto LL, Schoenwald RD. Furosemide (frusemide). A pharmacokinetic/pharmacodynamic review (Part I). Clin Pharmacokinet. 1990;18(5):381–408.
Kweldam CF, van der Kwast T, van Leenders GJ. On cribriform prostate cancer. Transl Androl Urol. 2018;7(1):145–54.
Egevad L, Ahmad AS, Algaba F, Berney DM, Boccon-Gibod L, Compérat E, et al. Standardization of gleason grading among 337 European pathologists. Histopathology. 2013;62(2):247–56.
Kweldam CF, Wildhagen MF, Steyerberg EW, Bangma CH, van der Kwast TH, van Leenders GJ. Cribriform growth is highly predictive for postoperative metastasis and disease-specific death in gleason score 7 prostate cancer. Mod Pathol. 2015;28(3):457–64.
van Houdt PJ, Ghobadi G, Schoots IG, Heijmink S, de Jong J, van der Poel HG, et al. Histopathological features of MRI-invisible regions of prostate cancer lesions. J Magn Reson Imaging. 2020;51(4):1235–46.
Panebianco V, Barchetti G, Simone G, Del Monte M, Ciardi A, Grompone MD, et al. Negative multiparametric magnetic resonance imaging for prostate cancer: what’s next? Eur Urol. 2018;74(1):48–54.
Mikoshi A, Miyai K, Hamabe F, Edo H, Ito K, Matsukuma S, et al. MRI-detectability and histological factors of prostate cancer including intraductal carcinoma and cribriform pattern. Prostate. 2022;82(4):452–63.
Duenweg SR, Fang X, Bobholz SA, Lowman AK, Brehler M, Kyereme F, et al. Diffusion restriction comparison between gleason 4 fused glands and cribriform glands within patient using whole-mount prostate pathology as ground truth. Tomography. 2022;8(2):635–43.
Ho MD, Ross AE, Eggener SE. Risk stratification of low-risk prostate cancer: individualizing care in the era of active surveillance. J Urol. 2023;210(1):38–45.
Epstein JI, Amin MB, Fine SW, Algaba F, Aron M, Baydar DE, et al. The 2019 genitourinary pathology society (GUPS) white paper on contemporary grading of prostate cancer. Arch Pathol Lab Med. 2021;145(4):461–93.
Dinerman BF, Khani F, Golan R, Bernstein AN, Cosiano MF, Margolis DJ, et al. Population-based study of the incidence and survival for intraductal carcinoma of the prostate. Urol Oncol. 2017;35(12):673.e9-e14.
Cohen RJ, Wheeler TM, Bonkhoff H, Rubin MA. A proposal on the identification, histologic reporting, and implications of intraductal prostatic carcinoma. Arch Pathol Lab Med. 2007;131(7):1103–9.
Haffner MC, Weier C, Xu MM, Vaghasia A, Gürel B, Gümüşkaya B, et al. Molecular evidence that invasive adenocarcinoma can mimic prostatic intraepithelial neoplasia (PIN) and intraductal carcinoma through retrograde glandular colonization. J Pathol. 2016;238(1):31–41.
Watts K, Li J, Magi-Galluzzi C, Zhou M. Incidence and clinicopathological characteristics of intraductal carcinoma detected in prostate biopsies: a prospective cohort study. Histopathology. 2013;63(4):574–9.
Vetrone L, Mei R. Histology and PSMA expression on immunohistochemistry in high-risk prostate cancer patients: comparison with (68) Ga-PSMA PET/CT features in primary staging. Cancers (Basel). 2023. https://doi.org/10.3390/cancers15061716.
Schmuck S, Nordlohne S, von Klot CA, Henkenberens C, Sohns JM, Christiansen H, et al. Comparison of standard and delayed imaging to improve the detection rate of [(68) Ga] PSMA I &T PET/CT in patients with biochemical recurrence or prostate-specific antigen persistence after primary therapy for prostate cancer. Eur J Nucl Med Mol Imaging. 2017;44(6):960–8.
Schmuck S, Mamach M, Wilke F, von Klot CA, Henkenberens C, Thackeray JT, et al. Multiple time-point 68Ga-PSMA I&T PET/CT for characterization of primary prostate cancer: value of early dynamic and delayed imaging. Clin Nucl Med. 2017;42(6):e286–93.
Funding
This study did not have an external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guner, L.A., Unal, K., Beylergil, V. et al. Enhancing PSMA PET/CT imaging of prostate cancer: investigating the impact of multiple time point evaluation, diuretic administration, cribriform pattern, and intraductal carcinoma. Ann Nucl Med 37, 618–628 (2023). https://doi.org/10.1007/s12149-023-01864-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-023-01864-7