Abstract
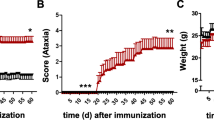
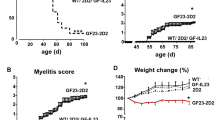
Interleukin 23 (IL-23) is a key mediator in neuroinflammation in numerous autoimmune diseases including multiple sclerosis (MS). However, the pathophysiology of IL-23 and how it contributes to neuroinflammation is poorly defined. To further clarify the role of IL-23 in CNS inflammation, we generated a transgenic mouse model (GF-IL23) with astrocyte-targeted expression of both IL-23 subunits, IL-23p19, and IL-23p40. These GF-IL23 mice spontaneously develop a progressive ataxic phenotype, which corresponds to cerebellar tissue destruction, and inflammatory infiltrates most prominent in the subarachnoidal and perivascular space. The CNS-cytokine milieu was characterized by numerous inflammatory mediators such as IL-17a and IFNγ. However, the leukocytic infiltrates were surprisingly predominated by B cells. To further examine the impact of the CNS-specific IL-23 synthesis on an additional systemic inflammatory stimulus, we applied the LPS-induced endotoxemia model. Administration of LPS in GF-IL23 mice resulted in early and pronounced microglial activation, enhanced cytokine production and, in sharp contrast to control animals, in the formation of lymphocytic infiltrates. Our model confirms a critical role for IL-23 in the induction of inflammation in the CNS, in particular facilitating the accumulation of lymphocytes in and around the brain. Thereby, CNS-specific synthesis of IL-23 is able to induce a cascade of inflammatory cytokines leading to microglia activation, astrocytosis, and ultimately tissue damage. The presented transgenic model will be a useful tool to further dissect the role of IL-23 in neuroinflammation.








Similar content being viewed by others
References
Becher B, Durell BG, Noelle RJ (2003) IL-23 produced by CNS-resident cells controls T cell encephalitogenicity during the effector phase of experimental autoimmune encephalomyelitis. J Clin Invest 112:1186–1191. https://doi.org/10.1172/JCI19079
Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, Lucian L, W T et al (2003) Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 421:744–748. https://doi.org/10.1038/nature01355
Alunno A, Carubbi F, Cafaro G, Pucci G, Battista F, Bartoloni E, Giacomelli R, Schillaci G et al (2015) Targeting the IL-23/IL-17 axis for the treatment of psoriasis and psoriatic arthritis. Expert Opin Biol Ther 15:1727–1737. https://doi.org/10.1517/14712598.2015.1084284
Cho JH, Feldman M (2015) Heterogeneity of autoimmune diseases: pathophysiologic insights from genetics and implications for new therapies. Nat Med 21:730–738. https://doi.org/10.1038/nm.3897
Fragoulis GE, Siebert S, McInnes IB (2016) Therapeutic targeting of IL-17 and IL-23 cytokines in immune-mediated diseases. Annu Rev Med 67:337–353. https://doi.org/10.1146/annurev-med-051914-021944
Furue M, Kadono T (2016) Psoriasis: behind the scenes. J Dermatol 43:4–8. https://doi.org/10.1111/1346-8138.13186
Zaky DS, El-Nahrery EM (2016) Role of interleukin-23 as a biomarker in rheumatoid arthritis patients and its correlation with disease activity. Int Immunopharmacol 31:105–108. https://doi.org/10.1016/j.intimp.2015.12.011
Javan MR, Shahraki S, Safa A, Zamani MR, Salmaninejad A, Aslani S (2017) An interleukin 12 B single nucleotide polymorphism increases IL-12p40 production and is associated with increased disease susceptibility in patients with relapsing-remitting multiple sclerosis. Neurol Res 39:435–441. https://doi.org/10.1080/01616412.2017.1301623
Sie C, Korn T, Mitsdoerffer M (2014) Th17 cells in central nervous system autoimmunity. Exp Neurol 262(Pt A):18–27. https://doi.org/10.1016/j.expneurol.2014.03.009
Kebir H, Ifergan I, Alvarez JI, Bernard M, Poirier J, Arbour N, Duquette P, Prat A (2009) Preferential recruitment of interferon-gamma-expressing TH17 cells in multiple sclerosis. Ann Neurol 66:390–402. https://doi.org/10.1002/ana.21748
Wen SR, Liu GJ, Feng RN, Gong FC, Zhong H, Duan SR, Bi S (2012) Increased levels of IL-23 and osteopontin in serum and cerebrospinal fluid of multiple sclerosis patients. J Neuroimmunol 244:94–96. https://doi.org/10.1016/j.jneuroim.2011.12.004
Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B, Sedgwick JD, McClanahan T, Kastelein RA et al (2005) IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med 201:233–240. https://doi.org/10.1084/jem.20041257
Shajarian M, Alsahebfosoul F, Etemadifar M, Sedaghat N, Shahbazi M, Firouzabadi FP, Dezashibi HM (2015) IL-23 plasma level measurement in relapsing remitting multiple sclerosis (RRMS) patients compared to healthy subjects. Immunol Investig 44:36–44. https://doi.org/10.3109/08820139.2014.930477
Hu Y, Zheng Y, Wu Y, Ni B, Shi S (2014) Imbalance between IL-17A-producing cells and regulatory T cells during ischemic stroke. Mediat Inflamm 2014:813045. https://doi.org/10.1155/2014/813045
Lv M, Liu Y, Zhang J, Sun L, Liu Z, Zhang S, Wang B, Su D et al (2011) Roles of inflammation response in microglia cell through Toll-like receptors 2/interleukin-23/interleukin-17 pathway in cerebral ischemia/reperfusion injury. Neuroscience 176:162–172. https://doi.org/10.1016/j.neuroscience.2010.11.066
Shichita T, Sugiyama Y, Ooboshi H, Sugimori H, Nakagawa R, Takada I, Iwaki T, Okada Y et al (2009) Pivotal role of cerebral interleukin-17-producing gamma deltaT cells in the delayed phase of ischemic brain injury. Nat Med 15:946–950. https://doi.org/10.1038/nm.1999
Wang M, Zhong D, Zheng Y, Li H, Chen H, Ma S, Sun Y, Yan W et al (2015) Damage effect of interleukin (IL)-23 on oxygen-glucose-deprived cells of the neurovascular unit via IL-23 receptor. Neuroscience 289:406–416. https://doi.org/10.1016/j.neuroscience.2015.01.012
Zheng Y, Zhong D, Chen H, Ma S, Sun Y, Wang M, Liu Q, Li G (2015) Pivotal role of cerebral interleukin-23 during immunologic injury in delayed cerebral ischemia in mice. Neuroscience 290:321–331. https://doi.org/10.1016/j.neuroscience.2015.01.041
Abbas A, Gregersen I, Holm S, Daissormont I, Bjerkeli V, Krohg-Sørensen K, Skagen KR, Dahl TB et al (2015) Interleukin 23 levels are increased in carotid atherosclerosis: possible role for the interleukin 23/interleukin 17 axis. Stroke 46:793–799. https://doi.org/10.1161/STROKEAHA.114.006516
Vom Berg J, Prokop S, Miller KR, Obst J, Kälin RE, Lopategui-Cabezas I, Wegner A, Mair F et al (2012) Inhibition of IL-12/IL-23 signaling reduces Alzheimer’s disease-like pathology and cognitive decline. Nat Med 18:1812–1819. https://doi.org/10.1038/nm.2965
Town T, Bai F, Wang T, Kaplan AT, Qian F, Montgomery RR, Anderson JF, Flavell RA et al (2009) Toll-like receptor 7 mitigates lethal West Nile encephalitis via interleukin 23-dependent immune cell infiltration and homing. Immunity 30:242–253. https://doi.org/10.1016/j.immuni.2008.11.012
Oppmann B, Lesley R, Blom B, Timans JC, Xu Y, Hunte B, Vega F, Yu N et al (2000) Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity 13:715–725
Parham C, Chirica M, Timans J, Vaisberg E, Travis M, Cheung J, Pflanz S, Zhang R et al (2002) A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rbeta1 and a novel cytokine receptor subunit, IL-23R. J Immunol 168:5699–5708
Pirhonen J, Matikainen S, Julkunen I (2002) Regulation of virus-induced IL-12 and IL-23 expression in human macrophages. J Immunol 169:5673–5678
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, Weiner HL, Kuchroo VK (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441:235–238. https://doi.org/10.1038/nature04753
Mangan PR, Harrington LE, O'Quinn DB, Helms WS, Bullard DC, Elson CO, Hatton RD, Wahl SM et al (2006) Transforming growth factor-beta induces development of the T(H)17 lineage. Nature 441:231–234. https://doi.org/10.1038/nature04754
Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B (2006) TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24:179–189. https://doi.org/10.1016/j.immuni.2006.01.001
Haak S, Croxford AL, Kreymborg K, Heppner FL, Pouly S, Becher B, Waisman A (2009) IL-17A and IL-17F do not contribute vitally to autoimmune neuro-inflammation in mice. J Clin Invest 119:61–69. https://doi.org/10.1172/JCI35997
Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y (2006) IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 177:566–573
Kreymborg K, Etzensperger R, Dumoutier L, Haak S, Rebollo A, Buch T, Heppner FL, Renauld JC et al (2007) IL-22 is expressed by Th17 cells in an IL-23-dependent fashion, but not required for the development of autoimmune encephalomyelitis. J Immunol 179:8098–8104
Codarri L, Gyülvészi G, Tosevski V, Hesske L, Fontana A, Magnenat L, Suter T, Becher B (2011) RORγt drives production of the cytokine GM-CSF in helper T cells, which is essential for the effector phase of autoimmune neuroinflammation. Nat Immunol 12:560–567. https://doi.org/10.1038/ni.2027
McQualter JL, Darwiche R, Ewing C, Onuki M, Kay TW, Hamilton JA, Reid HH, Bernard CC (2001) Granulocyte macrophage colony-stimulating factor: a new putative therapeutic target in multiple sclerosis. J Exp Med 194:873–882
Akwa Y, Hassett DE, Eloranta ML, Sandberg K, Masliah E, Powell H, Whitton JL, Bloom FE et al (1998) Transgenic expression of IFN-alpha in the central nervous system of mice protects against lethal neurotropic viral infection but induces inflammation and neurodegeneration. J Immunol 161:5016–5026
Campbell IL, Abraham CR, Masliah E, Kemper P, Inglis JD, Oldstone MB, Mucke L (1993) Neurologic disease induced in transgenic mice by cerebral overexpression of interleukin 6. Proc Natl Acad Sci U S A 90:10061–10065
Pagenstecher A, Lassmann S, Carson MJ, Kincaid CL, Stalder AK, Campbell IL (2000) Astrocyte-targeted expression of IL-12 induces active cellular immune responses in the central nervous system and modulates experimental allergic encephalomyelitis. J Immunol 164:4481–4492
Zimmermann J, Krauthausen M, Hofer MJ, Heneka MT, Campbell IL, Müller M (2013) CNS-targeted production of IL-17A induces glial activation, microvascular pathology and enhances the neuroinflammatory response to systemic endotoxemia. PLoS One 8:e57307. https://doi.org/10.1371/journal.pone.0057307
Boztug K, Carson MJ, Pham-Mitchell N, Asensio VC, DeMartino J, Campbell IL (2002) Leukocyte infiltration, but not neurodegeneration, in the CNS of transgenic mice with astrocyte production of the CXC chemokine ligand 10. J Immunol 169:1505–1515
Metten P, Best KL, Cameron AJ, Saultz AB, Zuraw JM, Yu CH, Wahlsten D, Crabbe JC (2004) Observer-rated ataxia: rating scales for assessment of genetic differences in ethanol-induced intoxication in mice. J Appl Physiol (1985) 97:360–368. https://doi.org/10.1152/japplphysiol.00086.2004
Quintana A, Müller M, Frausto RF, Ramos R, Getts DR, Sanz E, Hofer MJ, Krauthausen M et al (2009) Site-specific production of IL-6 in the central nervous system retargets and enhances the inflammatory response in experimental autoimmune encephalomyelitis. J Immunol 183:2079–2088. https://doi.org/10.4049/jimmunol.0900242
de Haas AH, Boddeke HW, Biber K (2008) Region-specific expression of immunoregulatory proteins on microglia in the healthy CNS. Glia 56:888–894. https://doi.org/10.1002/glia.20663
Bodea LG, Wang Y, Linnartz-Gerlach B, Kopatz J, Sinkkonen L, Musgrove R, Kaoma T, Muller A et al (2014) Neurodegeneration by activation of the microglial complement-phagosome pathway. J Neurosci 34:8546–8556. https://doi.org/10.1523/JNEUROSCI.5002-13.2014
Dong R, Hu D, Yang Y, Chen Z, Fu M, Wang DW, Xu X, Tu L (2017) EETs reduces LPS-induced hyperpermeability by targeting GRP78 mediated Src activation and subsequent Rho/ROCK signaling pathway. Oncotarget 8:50958–50971. https://doi.org/10.18632/oncotarget.17331
Li CC, Munitic I, Mittelstadt PR, Castro E, Ashwell JD (2015) Suppression of dendritic cell-derived IL-12 by endogenous glucocorticoids is protective in LPS-induced sepsis. PLoS Biol 13:e1002269. https://doi.org/10.1371/journal.pbio.1002269
Hauser SL, Bar-Or A, Comi G, Giovannoni G, Hartung HP, Hemmer B, Lublin F, Montalban X et al (2017) Ocrelizumab versus interferon Beta-1a in relapsing multiple sclerosis. N Engl J Med 376:221–234. https://doi.org/10.1056/NEJMoa1601277
Li R, Bar-Or A (2018) The multiple roles of B cells in multiple sclerosis and their implications in multiple sclerosis therapies. Cold Spring Harb Perspect Med 9. https://doi.org/10.1101/cshperspect.a029108
Li R, Patterson KR, Bar-Or A (2018) Reassessing B cell contributions in multiple sclerosis. Nat Immunol 19:696–707. https://doi.org/10.1038/s41590-018-0135-x
Pöllinger B, Krishnamoorthy G, Berer K, Lassmann H, Bösl MR, Dunn R, Domingues HS, Holz A et al (2009) Spontaneous relapsing-remitting EAE in the SJL/J mouse: MOG-reactive transgenic T cells recruit endogenous MOG-specific B cells. J Exp Med 206:1303–1316. https://doi.org/10.1084/jem.20090299
Batista FD, Harwood NE (2009) The who, how and where of antigen presentation to B cells. Nat Rev Immunol 9:15–27. https://doi.org/10.1038/nri2454
Crawford A, Macleod M, Schumacher T, Corlett L, Gray D (2006) Primary T cell expansion and differentiation in vivo requires antigen presentation by B cells. J Immunol 176:3498–3506
McLaughlin KA, Wucherpfennig KW (2008) B cells and autoantibodies in the pathogenesis of multiple sclerosis and related inflammatory demyelinating diseases. Adv Immunol 98:121–149. https://doi.org/10.1016/S0065-2776(08)00404-5
Ferretti E, Ponzoni M, Doglioni C, Pistoia V (2016) IL-17 superfamily cytokines modulate normal germinal center B cell migration. J Leukoc Biol 100:913–918. https://doi.org/10.1189/jlb.1VMR0216-096RR
Luther SA, Lopez T, Bai W, Hanahan D, Cyster JG (2000) BLC expression in pancreatic islets causes B cell recruitment and lymphotoxin-dependent lymphoid neogenesis. Immunity 12:471–481
Phares TW, DiSano KD, Stohlman SA, Segal BM, Bergmann CC (2016) CXCL13 promotes isotype-switched B cell accumulation to the central nervous system during viral encephalomyelitis. Brain Behav Immun 54:128–139. https://doi.org/10.1016/j.bbi.2016.01.016
Mitsdoerffer M, Lee Y, Jäger A, Kim HJ, Korn T, Kolls JK, Cantor H, Bettelli E et al (2010) Proinflammatory T helper type 17 cells are effective B-cell helpers. Proc Natl Acad Sci U S A 107:14292–14297. https://doi.org/10.1073/pnas.1009234107
Pfeifle R, Rothe T, Ipseiz N, Scherer HU, Culemann S, Harre U, Ackermann JA, Seefried M et al (2017) Regulation of autoantibody activity by the IL-23-TH17 axis determines the onset of autoimmune disease. Nat Immunol 18:104–113. https://doi.org/10.1038/ni.3579
Chognard G, Bellemare L, Pelletier AN, Dominguez-Punaro MC, Beauchamp C, Guyon MJ, Charron G, Morin N et al (2014) The dichotomous pattern of IL-12r and IL-23R expression elucidates the role of IL-12 and IL-23 in inflammation. PLoS One 9:e89092. https://doi.org/10.1371/journal.pone.0089092
Cocco C, Morandi F, Airoldi I (2011) Interleukin-27 and interleukin-23 modulate human plasmacell functions. J Leukoc Biol 89:729–734. https://doi.org/10.1189/jlb.1210660
Harbour SN, Maynard CL, Zindl CL, Schoeb TR, Weaver CT (2015) Th17 cells give rise to Th1 cells that are required for the pathogenesis of colitis. Proc Natl Acad Sci U S A 112:7061–7066. https://doi.org/10.1073/pnas.1415675112
Hirota K, Duarte JH, Veldhoen M, Hornsby E, Li Y, Cua DJ, Ahlfors H, Wilhelm C et al (2011) Fate map** of IL-17-producing T cells in inflammatory responses. Nat Immunol 12:255–263. https://doi.org/10.1038/ni.1993
Kurschus FC, Croxford AL, Heinen AP, Wörtge S, Ielo D, Waisman A (2010) Genetic proof for the transient nature of the Th17 phenotype. Eur J Immunol 40:3336–3346. https://doi.org/10.1002/eji.201040755
Ahern PP, Schiering C, Buonocore S, McGeachy MJ, Cua DJ, Maloy KJ, Powrie F (2010) Interleukin-23 drives intestinal inflammation through direct activity on T cells. Immunity 33:279–288. https://doi.org/10.1016/j.immuni.2010.08.010
Annunziato F, Cosmi L, Santarlasci V, Maggi L, Liotta F, Mazzinghi B, Parente E, Filì L et al (2007) Phenotypic and functional features of human Th17 cells. J Exp Med 204:1849–1861. https://doi.org/10.1084/jem.20070663
Ip JP, Noçon AL, Hofer MJ, Lim SL, Müller M, Campbell IL (2011) Lipocalin 2 in the central nervous system host response to systemic lipopolysaccharide administration. J Neuroinflammation 8:124. https://doi.org/10.1186/1742-2094-8-124
Kawanokuchi J, Shimizu K, Nitta A, Yamada K, Mizuno T, Takeuchi H, Suzumura A (2008) Production and functions of IL-17 in microglia. J Neuroimmunol 194:54–61. https://doi.org/10.1016/j.jneuroim.2007.11.006
Sonobe Y, Liang J, ** S, Zhang G, Takeuchi H, Mizuno T, Suzumura A (2008) Microglia express a functional receptor for interleukin-23. Biochem Biophys Res Commun 370:129–133. https://doi.org/10.1016/j.bbrc.2008.03.059
Kivity S, Agmon-Levin N, Blank M, Shoenfeld Y (2009) Infections and autoimmunity--friends or foes? Trends Immunol 30:409–414. https://doi.org/10.1016/j.it.2009.05.005
Acknowledgments
We thank Marco Hessler for his expert technical assistance. We further thank Jens Reimann for his support in routine histological procedures. MM was a post-doctoral fellow from the Deutsche Forschungsgemeinschaft (DFG, Mu17-07/3-1) and was also supported by the fund ‘Innovative Medical Research’ of the University of Muenster Medical School, Germany. ILC was supported by a start-up grant from the University of Sydney. GCP was supported by grants from the European Union Joint Program, Neurodegenerative Disease Research program (JPND; Horizon 2020 Framework Programme, grant agreement 643417/DACAPO-AD) and a DZNE Intersite grant (DEMDAS 2). JZ was funded by the fund “Bonfor” from the University of Bonn Medical School, Germany and the DFG (KFO177, University of Bonn). LN was funded by the DFG (KFO177, University of Bonn) and the “Oppenheim Foerderpreis” Novartis GmbH.
Author information
Authors and Affiliations
Contributions
Development of the transgenic construct: MM, MJH, ILC. Conceived and designed the experiments: LN, JZ, MK, MM. Performed the experiments: LN, JZ, MK, MM. Analyzed the data: LN, JZ, MK, AB, DG, MM. GCP planned and discussed the MRI experiments. RS developed and optimized the MRI sequences, performed the MRI experiments, analyzed the data, and wrote the MRI methods and results section. Contributed reagents/materials/analysis tools: LN, MTH, ILC, AB, DG, MM. Prepared the manuscript: LN, MM. Corrected and modified the manuscript: all authors.
Corresponding author
Ethics declarations
Ethical Approval
All applicable national and institutional guidelines for the care and use of animals were followed. All animal experiments were approved by the Animal Care Commission of Nordrhein-Westfalen.
Conflict of Interest
The authors declare that they have no conflict of interest.
LN and MM received travel grants from Merck, Sanofi Genzyme, TEVA, Novartis GmbH, honoraria for talks and research support from Novartis GmbH.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Louisa Nitsch and Julian Zimmermann shared authorship
Electronic supplementary material
Figure S1
CNS-specific expression of IL-23 leads to cerebellar infiltrates of the founder mice. The GF-IL23 transgenic founder mice GF-IL23–13 (A), GF-IL23–20 (B), GF-IL23–22 (C), GF-IL23–43 (D) display perivascular and subarachnoidal infiltrates in H&E-staining of the cerebellum. In the founder animals GF-IL23–77 and GF-p19 no infiltrates were observed. Scale bar: 100 μm (PDF 141 kb)
Figure S2
B cell differentiation and activation in GF-IL23 mice Representative flow cytometry profiles profiles of the of single cell suspensions of GF-IL23 mice. (A) CD19+ gated cells of the cerebellum and spleen of GF-IL23 mice were analyzed for CD21 and CD23 expression. We found follicular (CD21intCD23hi) and marginal zone (CD21hiCD23low) B cells. (B) CD45 gated cells of the cerebellum were stained against B220 and CD5 for analysis of a B1 (B220intCD5int) B cell population. (C, D). CD19+ gated cells of GF-IL23 transgenic cerebellum and spleen were analyzed for expression of IgM and IgD, CD19, CD80 and CD86 (C, D). Data are representative for at least n = 6–9 GF-IL23 mice with *p < 0.05. (PDF 58 kb)
Rights and permissions
About this article
Cite this article
Nitsch, L., Zimmermann, J., Krauthausen, M. et al. CNS-Specific Synthesis of Interleukin 23 Induces a Progressive Cerebellar Ataxia and the Accumulation of Both T and B Cells in the Brain: Characterization of a Novel Transgenic Mouse Model. Mol Neurobiol 56, 7977–7993 (2019). https://doi.org/10.1007/s12035-019-1640-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-019-1640-0