Abstract
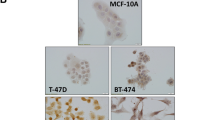
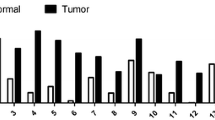
Argonaute (Ago) proteins have been demonstrated to be widely expressed and involved in post-transcriptional gene silencing and thus play key roles in carcinogenesis. Nevertheless, little is known about the specific role of Ago2 in gastric cancer (GC). Thus, we aimed to study the expression of Ago2 in 363 primary GC, 8 corresponding lymph node metastases and 10 non-neoplastic surrounding gastric epithelium tissues by immunohistochemical analyses and tissue microarray. The expression of Ago2 was also correlated with clinicopathological characteristics and HER-2 status. Ago2 expression levels in primary GC and corresponding lymph node metastases were significantly higher compared with healthy controls (P < 0.05). But, there was no difference of Ago2 between GC and its metastatic lymph node (P > 0.05). Ago2 up-regulation had no correlation with GC patients’ age, tumor location, tumor size, gross morphology or tumor infiltration. However, we found that Ago2 was different between HER-2 positive and HER-2 negative groups (P = 0.044), which had been demonstrated to be related to GC prognosis. And there was a great correlation between Ago2 expression and the tumor differentiation (P = 0.007), lymph node invasion (P = 0.000) and clinical stage (P = 0.006). Interestingly, Ago2 was also correlated to patients’ gender (P = 0.004), which may suggest a possible role of hormonal signal in the mechanisms of Ago2. Thus, our results suggested that up-regulation of Ago2 may play an important role in GC carcinogenesis and progression. Further studies on the cellular functions of Ago2 need to address these issues.




Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, et al. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12):2893–917.
**g JJ, Liu HY, Hao JK, Wang LN, Wang YP, et al. Gastric cancer incidence and mortality in Zhuanghe, China, between 2005 and 2010. World J Gastroenterol. 2012;18(11):1262–9.
Lima RT, Busacca S, Almeida GM, Gaudino G, Fennell DA, et al. MicroRNA regulation of core apoptosis pathways in cancer. Eur J Cancer. 2011;47(2):163–74.
Van Wynsberghe PM, Chan SP, Slack FJ, Pasquinelli AE. Analysis of microRNA expression and function. Methods Cell Biol. 2011;106:219–52.
Sinkovics JG. Molecular biology of oncogenic inflammatory processes. I. Non-oncogenic and oncogenic pathogens, intrinsic inflammatory reactions without pathogens, and microRNA/DNA interactions (Review). Int J Oncol. 2012;40(2):305–49.
Sandhu S, Garzon R. Potential applications of microRNAs in cancer diagnosis, prognosis, and treatment. Semin Oncol. 2011;38(6):781–7.
Hui A, How C, Ito E, Liu FF. Micro-RNAs as diagnostic or prognostic markers in human epithelial malignancies. BMC Cancer. 2011;30(11):500.
Murray MY, Rushworth SA, MacEwan DJ. MicroRNAs as a new therapeutic target towards leukaemia signalling. Cell Signal. 2012;24(2):363–8.
Song MY, Pan KF, Su HJ, Zhang L, Ma JL, et al. Identification of Serum MicroRNAs as novel non-invasive biomarkers for early detection of gastric cancer. PLoS ONE. 2012;7(3):e33608.
Wang D, Zhang Z, O’Loughlin E, Lee T, Houel S, et al. Quantitative functions of Argonaute proteins in mammalian development. Genes Dev. 2012;26(7):693–704.
O’Carroll D, Mecklenbrauker I, Das PP, Santana A, Koenig U, et al. A Slicer-independent role for Argonaute 2 in hematopoiesis and the microRNA pathway. Genes Dev. 2007;21(16):1999–2004.
Meister G, Landthaler M, Patkaniowska A, Dorsett Y, Teng G, et al. Human Argonaute2 mediates RNA cleavage targeted by miRNAs and siRNAs. Mol Cell. 2004;15(2):185–97.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors. AJCC cancer staging manual (7th ed). New York: Springer; 2010.
Rüschoff J, Dietel M, Baretton G, Arbogast S, Walch A, et al. HER2 diagnostics in gastric cancer-guideline validation and development of standardized immunohistochemical testing. Virchows Arch. 2010;457(3):299–307.
Kavanagh DO, Chambers G, O’Grady L, Barry KM, Waldron RP, et al. Is overexpression of HER-2 a predictor of prognosis in colorectal cancer? BMC Cancer. 2009;1(9):1.
Zhang J, Zhan N, Dong WG. Altered expression of selenium-binding protein 1 in gastric carcinoma and precursor lesions. Med Oncol. 2011;28(4):951–7.
Papaconstantinou I, Karakatsanis A, Gazouli M, Polymeneas G, Voros D. The role of microRNAs in liver cancer. Eur J Gastroenterol Hepatol. 2012;24(3):223–8.
Majumder S, Jacob ST. Emerging role of microRNAs in drug-resistant breast cancer. Gene Expr. 2011;15(3):141–51.
Fassan M, Croce CM, Rugge M. miRNAs in precancerous lesions of the gastrointestinal tract. World J Gastroenterol. 2011;17(48):5231–9.
Schoenberg DR. Mechanisms of endonuclease-mediated mRNA decay. Wiley Interdiscip Rev RNA. 2011;2(4):582–600.
Cullen BR. Viruses and microRNAs: RISCy interactions with serious consequences. Genes Dev. 2011;25(18):1881–94.
Diederichs S, Haber DA. Dual role for argonautes in microRNA processing and posttranscriptional regulation of microRNA expression. Cell. 2007;131(6):1097–108.
Matranga C, Tomari Y, Shin C, Bartel DP, Zamore PD. Passenger-strand cleavage facilitates assembly of siRNA into Ago2-containing RNAi enzyme complexes. Cell. 2005;123(4):607–20.
Leuschner PJ, Ameres SL, Kueng S, Martinez J. Cleavage of the siRNA passenger strand during RISC assembly in human cells. EMBO Rep. 2006;7(3):314–20.
Rand TA, Petersen S, Du F, Wang X. Argonaute2 cleaves the anti-guide strand of siRNA during RISC activation. Cell. 2005;123(4):621–9.
Cifuentes D, Xue H, Taylor DW, Patnode H, Mishima Y, et al. A novel miRNA processing pathway independent of Dicer requires Argonaute2 catalytic activity. Science. 2010;328(5986):1694–8.
Yoo NJ, Hur SY, Kim MS, Lee JY, Lee SH. Immunohistochemical analysis of RNA-induced silencing complex-related proteins AGO2 and TNRC6A in prostate and esophageal cancers. APMIS. 2010;118(4):271–6.
Li L, Yu C, Gao H, Li Y. Argonaute proteins: Potential biomarkers for human colon cancer. BMC Cancer. 2010;10:38.
Chang SS, Smith I, Glazer C, Hennessey P, Califano JA. EIF2C is overexpressed and amplified in head and neck squamous cell carcinoma. ORL J Otorhinolaryngol Relat Spec. 2010;72:337–43.
Sand M, Skrygan M, Georgas D, Arenz C, Gambichler T, et al. Expression levels of the microRNA maturing microprocessor complex component DGCR8 and the RNA-induced silencing complex (RISC) components Argonaute-1, Argonaute-2, PACT, TARBP1, and TARBP2 in epithelial skin cancer. Mol Carcinog; 2011.
Papachristou DJ, Sklirou E, Corradi D, Grassani C, Kontogeorgakos V, et al. Immunohistochemical analysis of the endoribonucleases Drosha, Dicer and Ago2 in smooth muscle tumours of soft tissues. Histopathology; 2012.
Zhang X, Graves P, Zeng Y. Overexpression of human Argonaute2 inhibits cell and tumor growth. Biochim Biophys Acta. 2012;1830(3):2553–61.
Burstein HJ. The distinctive nature of HER2-positive breast cancers. N Engl J Med. 2005;353:1652–4.
Gschwind A, Fischer OM, Ullrich A. The discovery of receptor tyrosine kinases: targets for cancer therapy. Nat Rev Cancer. 2004;4:361–70.
Baselga J, Swain SM. Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer. 2009;9:463–75.
Naoghare PK, Tak YK, Kim MJ, Han E, Song JM. Knock-down of argonaute 2 (AGO2) induces apoptosis in myeloid leukaemia cells and inhibits siRNA-mediated silencing of transfected oncogenes in HEK-293 cells. Basic Clin Pharmacol Toxicol. 2011;109(4):274–82.
Cheng N, Li Y, Han ZG. Ago2 promotes tumor metastasis via upregulating FAK expression in hepatocellular carcinoma. Hepatology. 2013;57:1906–18.
Acknowledgments
This work was supported by research grants from the National Natural Science Foundation of China (NO. 81171391), China Postdoctoral Science Foundation (No. 201150M1575) and Special Program of the Postdoctoral Science Foundation of China (NO. 2012T50895).
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Fan, Xs., Wang, Cx. et al. Up-regulation of Ago2 expression in gastric carcinoma. Med Oncol 30, 628 (2013). https://doi.org/10.1007/s12032-013-0628-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0628-2