Abstract
Background
Midline shift (MLS) has been associated with unfavorable outcome in patients with intracerebral hemorrhage (ICH). However, the optimal criteria to define the MLS measurements that indicate future outcome in ICH patients are absent, and the quantitative threshold of MLS that differentiates favorable and poor clinical outcome should be further explored.
Methods
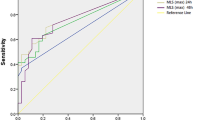
We enrolled patients with ICH who underwent admission computed tomography (CT) within 6 h after onset of symptoms. We assessed MLS at several locations, including the pineal gland, septum pellucidum, and cerebral falx. MLS(max) was defined as the maximum midline shift among these locations. Functional outcomes were assessed with the Modified Rankin Scale (mRS) at 3 months. We performed multivariate logistic regression analysis to investigate the MLS locations for predicting poor outcome. ROC curve analysis was used to establish whether MLS values were predictive of 90-day poor outcome.
Results
In 199 patients with ICH, 78 (39.2%) patients had poor functional outcome at 3-month follow-up. Pineal gland shift, septum pellucidum shift, cerebral falx shift, and MLS(max) all showed a significant difference between poor outcome and favorable outcome (p < 0.001). After adjustment for age, baseline Glasgow Coma Scale score, ICH location, time to initial CT, baseline ICH volume, and intraventricular hemorrhage, the MLS(max) was independently associated with poor outcome (p = 0.032). MLS(max) > 4 mm (our proposed optimal threshold) was more likely to have poorer outcomes than those without (p < 0.001).
Conclusions
MLS(max) can be a good independent predictor of clinical outcome, and MLS(max) > 4 mm is an optimal threshold associated with poor outcome in patients with ICH.



Similar content being viewed by others
References
Qureshi AI, Mendelow AD, Hanley DF. Intracerebral haemorrhage. Lancet. 2009;373:1632–44.
de Oliveira Manoel AL, Goffi A, et al. The critical care management of spontaneous intracranial hemorrhage: a contemporary review. Crit Care. 2016;20:272.
Cheung RT, Zou LY. Use of the original, modified, or new intracerebral hemorrhage score to predict mortality and morbidity after intracerebral hemorrhage. Stroke. 2003;34:1717–22.
Broderick JP, Brott TG, Duldner JE, Tomsick T, Huster G. Volume of intracerebral hemorrhage. A powerful and easy-to-use predictor of 30-day mortality. Stroke. 1993;24:987–93.
Hemphill JC 3rd, Bonovich DC, Besmertis L, Manley GT, Johnston SC. The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke. 2001;32:891–7.
Diringer MN, Edwards DF, Zazulia AR. Hydrocephalus: a previously unrecognized predictor of poor outcome from supratentorial intracerebral hemorrhage. Stroke. 1998;29:1352–7.
Portenoy RK, Lipton RB, Berger AR, Lesser ML, Lantos G. Intracerebral haemorrhage: a model for the prediction of outcome. J Neurol Neurosurg Psychiatry. 1987;50:976–9.
Gebel JM Jr, Jauch EC, Brott TG, et al. Natural history of perihematomal edema in patients with hyperacute spontaneous intracerebral hemorrhage. Stroke. 2002;33:2631–5.
Li Q, Zhang G, Huang YJ, et al. Blend sign on computed tomography: novel and reliable predictor for early hematoma growth in patients with intracerebral hemorrhage. Stroke. 2015;46:2119–23.
Li Q, Zhang G, **ong X, et al. Black Hole Sign: novel imaging marker that predicts hematoma growth in patients with intracerebral hemorrhage. Stroke. 2016;47:1777–81.
Jauch EC, Pineda JA, Hemphill JC. Emergency neurological life support: intracerebral hemorrhage. Neurocrit Care. 2015;23(Suppl 2):S83–93.
Zazulia AR, Diringer MN, Derdeyn CP, Powers WJ. Progression of mass effect after intracerebral hemorrhage. Stroke. 1999;30:1167–73.
Ropper AH. Lateral displacement of the brain and level of consciousness in patients with an acute hemispheral mass. N Engl J Med. 1986;314:953–8.
Boulouis G, Morotti A, Brouwers HB, et al. Noncontrast computed tomography hypodensities predict poor outcome in intracerebral hemorrhage patients. Stroke. 2016;47:2511–6.
Qureshi AI, Palesch YY, Barsan WG, et al. Intensive blood-pressure lowering in patients with acute cerebral hemorrhage. N Engl J Med. 2016;375:1033–43.
Kothari RU, Brott T, Broderick JP, et al. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996;27:1304–5.
Sun W, Pan W, Kranz PG, et al. Predictors of late neurological deterioration after spontaneous intracerebral hemorrhage. Neurocrit Care. 2013;19:299–305.
Hays A, Diringer MN. Elevated troponin levels are associated with higher mortality following intracerebral hemorrhage. Neurology. 2006;66:1330–4.
Fan JS, Huang HH, Chen YC, et al. Emergency department neurologic deterioration in patients with spontaneous intracerebral hemorrhage: incidence, predictors, and prognostic significance. Acad Emerg Med. 2012;19:133–8.
Kowalski RG, Buitrago MM, Duckworth J, et al. Neuroanatomical predictors of awakening in acutely comatose patients. Ann Neurol. 2015;77:804–16.
Samarasekera N, Fonville A, Lerpiniere C, et al. Influence of intracerebral hemorrhage location on incidence, characteristics, and outcome: population-based study. Stroke. 2015;46:361–8.
Miyai I, Suzuki T, Kang J, Volpe BT. Improved functional outcome in patients with hemorrhagic stroke in putamen and thalamus compared with those with stroke restricted to the putamen or thalamus. Stroke. 2000;31:1365–9.
Keong LH, Ghani AR, Awang MS, Sayuthi S, Idris B, Abdullah JM. The role of a high augmentation index in spontaneous intracerebral hemorrhage to prognosticate mortality. Acta Neurochir Suppl. 2011;111:375–9.
Chung CS, Caplan LR, Han W, Pessin MS, Lee KH, Kim JM. Thalamic haemorrhage. Brain. 1996;119:1873–86.
Delcourt C, Sato S, Zhang S, et al. Intracerebral hemorrhage location and outcome among INTERACT2 participants. Neurology. 2017;88:1408–14.
Uddin LQ, Iacoboni M, Lange C, Keenan JP. The self and social cognition: the role of cortical midline structures and mirror neurons. Trends Cogn Sci. 2007;11:153–7.
Cavanna AE, Trimble MR. The precuneus: a review of its functional anatomy and behavioural correlates. Brain. 2006;129:564–83.
Gerner ST, Kuramatsu JB, Moeller S, et al. Specific lobar affection reveals a rostrocaudal gradient in functional outcome in spontaneous intracerebral hemorrhage. Stroke. 2017;48:587–95.
Wiggins WS, Moody DM, Toole JF, Laster DW, Ball MR. Clinical and computerized tomographic study of hypertensive intracerebral hemorrhage. Arch Neurol. 1978;35:832–3.
Fogelholm R, Murros K, Rissanen A, Avikainen S. Long term survival after primary intracerebral haemorrhage: a retrospective population based study. J Neurol Neurosurg Psychiatry. 2005;76:1534–8.
Fogelholm R, Murros K, Rissanen A, Avikainen S. Admission blood glucose and short term survival in primary intracerebral haemorrhage: a population based study. J Neurol Neurosurg Psychiatry. 2005;76:349–53.
Bhatia R, Singh H, Singh S, et al. A prospective study of in-hospital mortality and discharge outcome in spontaneous intracerebral hemorrhage. Neurol India. 2013;61:244–8.
Daverat P, Castel JP, Dartigues JF, Orgogozo JM. Death and functional outcome after spontaneous intracerebral hemorrhage. A prospective study of 166 cases using multivariate analysis. Stroke. 1991;22:1–6.
Masè G, Zorzon M, Biasutti E, Tasca G, Vitrani B, Cazzato G. Immediate prognosis of primary intracerebral hemorrhage using an easy model for the prediction of survival. Acta Neurol Scand. 1995;91:306–9.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (Grant Nos. 81200899, 81371310) and the National Basic Research Program of China (973 Program) (Grant No. 2009CB918300).
Author information
Authors and Affiliations
Contributions
W-SY, QL, and PX contributed to the conception and design of the article. W-SY, QL, Q-JL, RL, X-CW, L-BZ, and PX all contributed to data collection, data interpretation, and data analysis. W-SY contributed to drafting of the manuscript. PX and QL contributed to administrative, technical, or material support. All authors contributed to revising the article for important intellectual content and gave final approval of the version to be published.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Yang, WS., Li, Q., Li, R. et al. Defining the Optimal Midline Shift Threshold to Predict Poor Outcome in Patients with Supratentorial Spontaneous Intracerebral Hemorrhage. Neurocrit Care 28, 314–321 (2018). https://doi.org/10.1007/s12028-017-0483-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-017-0483-7