Abstract
Purpose
Acromegaly is a rare disease that often requires drug treatment to achieve control, with pegvisomant being one of the most widely used therapies. In the present paper, we aimed to obtain evidence regarding the effectiveness and safety of pegvisomant by reviewing real-world observational longitudinal studies.
Methods
A systematic review was performed with a meta-analysis of event rates (95% confidence interval (CI)) using a random effects model. Sensitivity and subgroup analyses were performed (comprehensive meta-analysis 2.0). The systematic review was performed in accordance to preferred reporting items for systematic reviews and meta-analyses, meta-analysis of observational studies in epidemiology, and Cochrane recommendations (PROSPERO register CRD 42017059880). PubMed, Scopus, Web of Science, and SciELO were used to search for literature. Observational studies in patients using pegvisomant for the treatment of acromegaly were included.
Results
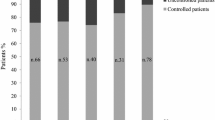
Initially, 552 papers were retrieved from the databases; and 31 articles were included in the qualitative analysis and 14 in the quantitative analysis. Eight primary meta-analyses were performed. The overall rate of patients with disease control was of 60.9% (51.8–69.3%; 95% CI). When considering patients under monotherapy, the control rate was 71.7% (64.0–78.4%; 95% CI). Tumor growth was estimated in 7.3% (4.7–11.1%; 95% CI) and elevation of transaminases in 3.0% (1.7–5.2%; 95% CI).
Conclusions
The real-world data showed that the effectiveness of pegvisomant is not as high as reported in interventional studies. Acromegaly appears to be better controlled when pegvisomant is used as a monotherapy. No serious adverse events were associated with the use of pegvisomant; however, given the high cost of this drug, further studies are required.



Similar content being viewed by others
References
K. Gunawardane, T. Krarup Hansen, J. Sandahl Christiansen, J.O. Lunde Jorgensen, Normal physiology of growth hormone in adults. in L.J. De Groot, G. Chrousos, K. Dungan, K.R. Feingold, A. Grossman, J.M. Hershman, C. Koch, M. Korbonits, R. McLachlan, M. New, J. Purnell, R. Rebar, F. Singer, A. Vinik (eds.) Endotext. (South Dartmouth, MA, 2000)
L. Katznelson, E.R. Laws Jr., S. Melmed, M.E. Molitch, M.H. Murad, A. Utz, J.A. Wass, S. Endocrine, Acromegaly: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 99(11), 3933–3951 (2014). https://doi.org/10.1210/jc.2014-2700
A. Colao, D. Ferone, P. Marzullo, G. Lombardi, Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr. Rev. 25(1), 102–152 (2004). https://doi.org/10.1210/er.2002-0022
S. Melmed, Acromegaly pathogenesis and treatment. J. Clin. Invest 119(11), 3189–3202 (2009). https://doi.org/10.1172/JCI39375
P. Chanson, S. Salenave, P. Kamenicky, Acromegaly. Handb. Clin. Neurol. 124, 197–219 (2014). https://doi.org/10.1016/B978-0-444-59602-4.00014-9
S. Stormann, J. Schopohl, Perspectives on investigational drugs for acromegaly. Expert Opin. Investig. Drugs 25(4), 381–384 (2016). https://doi.org/10.1517/13543784.2016.1142530
J.M. Silverstein, Need for improved monitoring in patients with acromegaly. Endocr. Connect 4(4), R59–67 (2015). https://doi.org/10.1530/EC-15-0064
S.J. Neggers, A. Muhammad, A.J. van der Lely, Pegvisomant treatment in acromegaly. Neuroendocrinology 103(1), 59–65 (2016). https://doi.org/10.1159/000381644
FDA: Pegvisomant Approval (2018), https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=BasicSearch.process. Accessed 19 July 2018
EMA: Pegvisomant Approval (2018).
D.J. Moore, Y. Adi, M.J. Connock, S. Bayliss, Clinical effectiveness and cost-effectiveness of pegvisomant for the treatment of acromegaly: a systematic review and economic evaluation. BMC Endocr. Disord. 9, 20 (2009). https://doi.org/10.1186/1472-6823-9-20
L.P. Leonart, V.L. Ferreira, F.S. Tonin, F. Fernandez-Llimos, R. Pontarolo, Medical treatments for acromegaly: a systematic review and network meta-analysis. Value Health 21(7), 874–880 (2018). https://doi.org/10.1016/j.jval.2017.12.014
P.J. Trainer, W.M. Drake, L. Katznelson, P.U. Freda, V. Herman-Bonert, A.J. van der Lely, E.V. Dimaraki, P.M. Stewart, K.E. Friend, M.L. Vance, G.M. Besser, J.A. Scarlett, M.O. Thorner, C. Parkinson, A. Klibanski, J.S. Powell, A.L. Barkan, M.C. Sheppard, M. Malsonado, D.R. Rose, D.R. Clemmons, G. Johannsson, B.A. Bengtsson, S. Stavrou, D.L. Kleinberg, D.M. Cook, L.S. Phillips, M. Bidlingmaier, C.J. Strasburger, S. Hackett, K. Zib, W.F. Bennett, R.J. Davis, Treatment of acromegaly with the growth hormone-receptor antagonist pegvisomant. N. Engl. J. Med 342(16), 1171–1177 (2000). https://doi.org/10.1056/NEJM200004203421604
A.J. van der Lely, R.K. Hutson, P.J. Trainer, G.M. Besser, A.L. Barkan, L. Katznelson, A. Klibanski, V. Herman-Bonert, S. Melmed, M.L. Vance, P.U. Freda, P.M. Stewart, K.E. Friend, D.R. Clemmons, G. Johannsson, S. Stavrou, D.M. Cook, L.S. Phillips, C.J. Strasburger, S. Hackett, K.A. Zib, R.J. Davis, J.A. Scarlett, M.O. Thorner, Long-term treatment of acromegaly with pegvisomant, a growth hormone-receptor antagonist. Lancet 358(9295), 1754–1759 (2001)
S. de Lusignan, L. Crawford, N. Munro, Creating and using real-world evidence to answer questions about clinical effectiveness. J. Innov. Health Inform. 22(3), 368–373 (2015). https://doi.org/10.14236/jhi.v22i3.177
M.L. Berger, H. Sox, R.J. Willke, D.L. Brixner, H.G. Eichler, W. Goettsch, D. Madigan, A. Makady, S. Schneeweiss, R. Tarricone, S.V. Wang, J. Watkins, C. Daniel Mullins, Good practices for real-world data studies of treatment and/or comparative effectiveness: recommendations from the joint ISPOR-ISPE Special Task Force on real-world evidence in healthcare decision making. Pharmacoepidemiol. Drug Saf. 26(9), 1033–1039 (2017). https://doi.org/10.1002/pds.4297
J.P.T. Higgins, G.S.: Cochrane handbook for systematic reviews of interventions, version 5.1.0. The Cochrane Collaboration (2011).
A. Liberati, D.G. Altman, J. Tetzlaff, C. Mulrow, P.C. Gotzsche, J.P. Ioannidis, M. Clarke, P.J. Devereaux, J. Kleijnen, D. Moher, The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. Br Med J 339, b2700 (2009). https://doi.org/10.1136/bmj.b2700
D.F. Stroup, J.A. Berlin, S.C. Morton, I. Olkin, G.D. Williamson, D. Rennie, D. Moher, B.J. Becker, T.A. Sipe, S.B. Thacker, Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. J Am Med Assoc 283(15), 2008–2012 (2000)
G.A. Wells, B. Shea, D. O’Connell, J. Peterson, V. Welch, M. Losos, P. Tugwell, The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Accessed 13 June 2018
T. Brue, ACROSTUDY: status update on 469 patients. Horm. Res. 71(Suppl 1), S34–S38 (2009). https://doi.org/10.1159/000178035
P.J. Trainer, ACROSTUDY: the first 5 years. Eur. J. Endocrinol. 161(Suppl 1), S19–S24 (2009). https://doi.org/10.1530/EJE-09-0322
A.J. van der Lely, B.M. Biller, T. Brue, M. Buchfelder, E. Ghigo, R. Gomez, J. Hey-Hadavi, F. Lundgren, N. Rajicic, C.J. Strasburger, S.M. Webb, M. Koltowska-Haggstrom, Long-term safety of pegvisomant in patients with acromegaly: comprehensive review of 1288 subjects in ACROSTUDY. J. Clin. Endocrinol. Metab. 97(5), 1589–1597 (2012). https://doi.org/10.1210/jc.2011-2508
P.U. Freda, M.B. Gordon, N. Kelepouris, P. Jonsson, M. Koltowska-Haggstrom, A.J. van der Lely, Long-term treatment with pegvisomant as monotherapy in patients with acromegaly: experience from ACROSTUDY. Endocr. Pract. 21(3), 264–274 (2015). https://doi.org/10.4158/EP14330.OR
N.A. Tritos, P. Chanson, C. Jimenez, D. King, P.J. Jonsson, A. Klibanski, B.M. Biller, Effectiveness of first-line pegvisomant monotherapy in acromegaly: an ACROSTUDY analysis. Eur. J. Endocrinol. 176(2), 213–220 (2017). https://doi.org/10.1530/EJE-16-0697
C.J. Strasburger, A. Mattsson, P. Wilton, F. Aydin, J. Hey-Hadavi, B.M.K. Biller, Increasing frequency of combination medical therapy in the treatment of acromegaly with the GH receptor antagonist pegvisomant. Eur. J. Endocrinol. 178(4), 321–329 (2018). https://doi.org/10.1530/EJE-17-0996
I. Schreiber, M. Buchfelder, M. Droste, K. Forssmann, K. Mann, B. Saller, C.J. Strasburger, I. German Pegvisomant, Treatment of acromegaly with the GH receptor antagonist pegvisomant in clinical practice: safety and efficacy evaluation from the German Pegvisomant Observational Study. Eur. J. Endocrinol. 156(1), 75–82 (2007). https://doi.org/10.1530/eje.1.02312
C.J. Strasburger, M. Buchfelder, M. Droste, K. Mann, G.K. Stalla, B. Saller, I. German Pegvisomant, Experience from the German pegvisomant observational study. Horm. Res. 68(Suppl 5), S70–S73 (2007). https://doi.org/10.1159/000110481
M. Buchfelder, S. Schlaffer, M. Droste, K. Mann, B. Saller, K. Brubach, G.K. Stalla, C.J. Strasburger; German Pegvisomant Observational Study, The German ACROSTUDY: past and present. Eur. J. Endocrinol. 161(Suppl 1), S3–S10 (2009). https://doi.org/10.1530/EJE-09-0350
M. Buchfelder, D. Weigel, M. Droste, K. Mann, B. Saller, K. Brubach, G.K. Stalla, M. Bidlingmaier, C.J. Strasburger, Investigators of German Pegvisomant Observational Study. Pituitary tumor size in acromegaly during pegvisomant treatment: experience from MR re-evaluations of the German Pegvisomant Observational Study. Eur. J. Endocrinol. 161(1), 27–35 (2009). https://doi.org/10.1530/EJE-08-0910
C. Berg, S. Petersenn, H. Lahner, B.L. Herrmann, M. Buchfelder, M. Droste, G.K. Stalla, C.J. Strasburger, U. Roggenbuck, N. Lehmann, S. Moebus, K.H. Jockel, S. Mohlenkamp, R. Erbel, B. Saller, K. Mann; Investigative Group of the Heinz Nixdorf Recall Study and the German Pegvisomant Observational Study Board and Investigators, Cardiovascular risk factors in patients with uncontrolled and long-term acromegaly: comparison with matched data from the general population and the effect of disease control. J. Clin. Endocrinol. Metab. 95(8), 3648–3656 (2010). https://doi.org/10.1210/jc.2009-2570
C. Urbani, C. Sardella, A. Calevro, G. Rossi, I. Scattina, M. Lombardi, I. Lupi, L. Manetti, E. Martino, F. Bogazzi, Effects of medical therapies for acromegaly on glucose metabolism. Eur. J. Endocrinol. 169(1), 99–108 (2013). https://doi.org/10.1530/EJE-13-0032
A. Bianchi, F. Valentini, R. Iuorio, M. Poggi, R. Baldelli, M. Passeri, A. Giampietro, L. Tartaglione, S. Chiloiro, M. Appetecchia, P. Gargiulo, A. Fabbri, V. Toscano, A. Pontecorvi, L. De Marinis, Long-term treatment of somatostatin analog-refractory growth hormone-secreting pituitary tumors with pegvisomant alone or combined with long-acting somatostatin analogs: a retrospective analysis of clinical practice and outcomes. J. Exp. Clin. Cancer Res 32, 40 (2013). https://doi.org/10.1186/1756-9966-32-40
S. Grottoli, P. Maffei, F. Bogazzi, S. Cannavo, A. Colao, E. Ghigo, R. Gomez, E. Graziano, M. Monterubbianesi, P. Jonsson, L. De Marinis, ACROSTUDY: the Italian experience. Endocrine 48(1), 334–341 (2015). https://doi.org/10.1007/s12020-014-0393-9
S. Cannavo, F. Bogazzi, A. Colao, L. De Marinis, P. Maffei, R. Gomez, E. Graziano, M. Monterubbianesi, S. Grottoli, G. Italian Acrostudy, Does pegvisomant treatment expertise improve control of resistant acromegaly? The Italian ACROSTUDY experience. J. Endocrinol. Invest 38(10), 1099–1109 (2015). https://doi.org/10.1007/s40618-015-0289-4
R.S. Auriemma, L.F. Grasso, M. Galdiero, M. Galderisi, C. Pivonello, C. Simeoli, M.C. De Martino, R. Ferrigno, M. Negri, C. de Angelis, R. Pivonello, A. Colao, Effects of long-term combined treatment with somatostatin analogues and pegvisomant on cardiac structure and performance in acromegaly. Endocrine 55(3), 872–884 (2017). https://doi.org/10.1007/s12020-016-0995-5
M. Ragonese, S. Grottoli, P. Maffei, A. Alibrandi, M.R. Ambrosio, G. Arnaldi, A. Bianchi, S. Puglisi, M.C. Zatelli, L. De Marinis, E. Ghigo, A. Giustina, F. Maffezzoni, C. Martini, L. Trementino, S. Cannavo, How to improve effectiveness of pegvisomant treatment in acromegalic patients. J. Endocrinol. Invest. (2017). https://doi.org/10.1007/s40618-017-0773-0
S. Chiloiro, G. Mazziotti, A. Giampietro, A. Bianchi, S. Frara, M. Mormando, A. Pontecorvi, A. Giustina, L. De Marinis, Effects of pegvisomant and somatostatin receptor ligands on incidence of vertebral fractures in patients with acromegaly. Pituitary (2018). https://doi.org/10.1007/s11102-018-0873-7
M. Marazuela, T. Lucas, C. Alvarez-Escola, M. Puig-Domingo, N.G. de la Torre, P. de Miguel-Novoa, A. Duran-Hervada, R. Manzanares, M. Luque-Ramirez, I. Halperin, F.F. Casanueva, I. Bernabeu, Long-term treatment of acromegalic patients resistant to somatostatin analogues with the GH receptor antagonist pegvisomant: its efficacy in relation to gender and previous radiotherapy. Eur. J. Endocrinol. 160(4), 535–542 (2009). https://doi.org/10.1530/EJE-08-0705
M. Marazuela, A.E. Paniagua, M.D. Gahete, T. Lucas, C. Alvarez-Escola, R. Manzanares, J. Cameselle-Teijeiro, M. Luque-Ramirez, R.M. Luque, E. Fernandez-Rodriguez, J.P. Castano, I. Bernabeu, Somatotroph tumor progression during pegvisomant therapy: a clinical and molecular study. J. Clin. Endocrinol. Metab. 96(2), E251–259 (2011). https://doi.org/10.1210/jc.2010-1742
G. Sesmilo, E. Resmini, I. Bernabeu, J. Aller, A. Soto, M. Mora, A. Pico, C. Fajardo, E. Torres, C. Alvarez-Escola, R. Garcia, C. Blanco, R. Camara, S. Gaztambide, I. Salinas, C.D. Pozo, I. Castells, C. Villabona, B. Biagetti, S.M. Webb, Escape and lipodystrophy in acromegaly during pegvisomant therapy, a retrospective multicentre Spanish study. Clin. Endocrinol. 81(6), 883–890 (2014). https://doi.org/10.1111/cen.12440
I. Bernabeu, A. Pico, E. Venegas, J. Aller, C. Alvarez-Escola, J.A. Garcia-Arnes, M. Marazuela, P. Jonsson, N. Mir, M. Garcia Vargas, A.G. Spanish, Safety of long-term treatment with Pegvisomant: analysis of Spanish patients included in global ACROSTUDY. Pituitary 19(2), 127–137 (2016). https://doi.org/10.1007/s11102-015-0691-0
M. Puig-Domingo, A. Soto, E. Venegas, R. Vilchez, C. Blanco, F. Cordido, T. Lucas, M. Marazuela, R. Casany, G. Cuatrecasas, C. Fajardo, M.A. Galvez, S. Maraver, T. Martin, E. Romero, M. Paja, A. Pico, I. Bernabeu, E. Resmini; ACROCOMB study group, Use of lanreotide in combination with cabergoline or pegvisomant in patients with acromegaly in the clinical practice: the ACROCOMB study. Endocrinol. Nutr. 63(8), 397–408 (2016). https://doi.org/10.1016/j.endonu.2016.05.010
A.M. Ramos-Levi, I. Bernabeu, C. Alvarez-Escola, J. Aller, T. Lucas, P. de Miguel, L. Rodriguez-Canete, M.A. Sampedro-Nunez, I. Halperin, M. Puig-Domingo, M. Marazuela, Long-term treatment with pegvisomant for acromegaly: a 10-year experience. Clin. Endocrinol. 84(4), 540–550 (2016). https://doi.org/10.1111/cen.12993
E. Kuhn, L. Maione, A. Bouchachi, M. Roziere, S. Salenave, S. Brailly-Tabard, J. Young, P. Kamenicky, P. Assayag, P. Chanson, Long-term effects of pegvisomant on comorbidities in patients with acromegaly: a retrospective single-center study. Eur. J. Endocrinol. 173(5), 693–702 (2015). https://doi.org/10.1530/EJE-15-0500
P. Chanson, T. Brue, B. Delemer, P. Caron, F. Borson-Chazot, H. Zouater, Medecins de l’Etude ACROSTUDY. Pegvisomant treatment in patients with acromegaly in clinical practice: the French ACROSTUDY. Ann. Endocrinol. 76(6), 664–670 (2015). https://doi.org/10.1016/j.ando.2015.10.003
N.G. Basavilbaso, M. Guitelman, A. Nagelberg, G. Stalldecker, A. Carabelli, O. Bruno, K. Danilowitz, M. Manavela, S. Mallea Gil, C. Ballarino, R. Guelman, D. Katz, S. Fidalgo, R. Leal, H. Fideleff, M. Servidio, D. Bruera, F. Librandi, A. Chervin, M. Vitale, A. Basso, Experience from the Argentine Pegvisomant Observational Study: preliminary data. Front. Horm. Res. 38, 42–49 (2010). https://doi.org/10.1159/000318493
M. Madsen, S. Fisker, U. Feldt-Rasmussen, M. Andreassen, L.O. Kristensen, H. Orskov, J.O. Jorgensen, Circulating levels of pegvisomant and endogenous growth hormone during prolonged pegvisomant therapy in patients with acromegaly. Clin. Endocrinol. 80(1), 92–100 (2014). https://doi.org/10.1111/cen.12239
L. Kasuki, E.O. Machado, L.L. Ogino, M.C. Coelho, C.M. Silva, L.E. Wildemberg, C.H. Lima, M.R. Gadelha, Experience with pegvisomant treatment in acromegaly in a single Brazilian tertiary reference center: efficacy, safety and predictors of response. Arch. Endocrinol. Metab. 60(5), 479–485 (2016). https://doi.org/10.1590/2359-3997000000210
O. Haliloglu, E. Kuruoglu, H.M. Ozkaya, F.E. Keskin, O. Gunaldi, B. Oz, N. Gazioglu, P. Kadioglu, N. Tanriover, Multidisciplinary approach for acromegaly: a single tertiary center’s experience. World Neurosurg. 88, 270–276 (2016). https://doi.org/10.1016/j.wneu.2015.12.092
O. Karapanou, M. Tzanela, M. Christoforaki, L. Papastathopoulou, P. Moutsatsou, E. Botoula, S. Tsagarakis, Therapeutic trends and outcome of acromegaly: a single-center experience over a 40-year period. Hormones 15(3), 368–376 (2016). https://doi.org/10.14310/horm.2002.1680
L. Ferreri, R.S. Auriemma, L.F.S. Grasso, R. Pivonello, A. Colao , Efficacy and tolerability of treatment with pegvisomant in acromegaly: an overview of literature. Expert Opinion on Orphan. Drugs 3(1), 12 (2015). https://doi.org/10.1517/21678707.2014.970171
A. Giustina, G. Arnaldi, F. Bogazzi, S. Cannavo, A. Colao, L. De Marinis, E. De Menis, E. Degli Uberti, F. Giorgino, S. Grottoli, A.G. Lania, P. Maffei, R. Pivonello, E. Ghigo, Pegvisomant in acromegaly: an update. J. Endocrinol. Invest. 40(6), 577–589 (2017). https://doi.org/10.1007/s40618-017-0614-1
A.N. Paisley, P. Trainer, W. Drake, Pegvisomant: a novel pharmacotherapy for the treatment of acromegaly. Expert Opin. Biol. Ther. 4(3), 421–425 (2004). https://doi.org/10.1517/14712598.4.3.421
E. Ghigo, B.M. Biller, A. Colao, I.A. Kourides, N. Rajicic, R.K. Hutson, L. De Marinis, A. Klibanski, Comparison of pegvisomant and long-acting octreotide in patients with acromegaly naive to radiation and medical therapy. J. Endocrinol. Invest. 32(11), 924–933 (2009). https://doi.org/10.3275/672310.1007/BF03345774
A. Colao, R.S. Auriemma, R. Pivonello, The effects of somatostatin analogue therapy on pituitary tumor volume in patients with acromegaly. Pituitary 19(2), 210–221 (2016). https://doi.org/10.1007/s11102-015-0677-y
G. Mazziotti, A. Giustina, Effects of lanreotide SR and autogel on tumor mass in patients with acromegaly: a systematic review. Pituitary 13(1), 60–67 (2010). https://doi.org/10.1007/s11102-009-0169-z
M.J. Rousseau, J.C. Evans, Key statistical assumptions and methods in one arm meta-analyses with binary endpoints and low event rates, including a real-life example in the area of endoscopic colonic stenting. Cogent Med 4(1), 1334318 (2017). https://doi.org/10.1080/2331205X.2017.1334318
Acknowledgments
The authors would like to thank the Brazilian National Counsel of Technological and Scientific Development (CNPq) and the Coordination for the Improvement of Higher Education Personnel (CAPES) for providing scholarships.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Leonart, L.P., Tonin, F.S., Ferreira, V.L. et al. Effectiveness and safety of pegvisomant: a systematic review and meta-analysis of observational longitudinal studies. Endocrine 63, 18–26 (2019). https://doi.org/10.1007/s12020-018-1729-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1729-7