Abstract
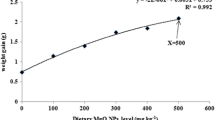
The present study was conducted to assess the influence of dietary zinc nanoparticles (size 50 nm) on the growth, biochemical constituents, enzymatic antioxidant levels and the nonspecific immune response of the freshwater prawn, Macrobrachium rosenbergii post larvae (PL). The concentrations of dietary supplement zinc nanoparticles (ZnNPs) were 0, 10, 20, 40, 60 and 80 mg kg−1 with the basal diet, and the level of Zn in ZnNP-supplemented diets were 0.71, 10.61, 20.73, 40.73, 60.61 and 80.60 mg kg−1, respectively. ZnNP-incorporated diets were fed to M. rosenbergii PL (initial body weight, 0.18 ± 0.02 g) in a triplicate experimental setup for a period of 90 days. ZnNP supplemented feed fed PL up to 60 mg kg−1 showed significantly (P < 0.05) improved performance in survival, growth and activities of digestive enzymes (protease, amylase and lipase). The concentrations of biochemical constituents (total protein, total amino acid, total carbohydrate and total lipid), total haemocyte count and differential haemocyte count were elevated in 10–60 mg kg−1 ZnNP supplemented feed fed PL. However, the PL fed with 80 mg ZnNPs kg−1 showed negative results. Activities of enzymatic antioxidants [superoxide dismutase (SOD) and catalase (CAT)], metabolic enzymes [glutamate–oxaloacetate transaminase (GOT) and glutamate–pyruvate transaminase (GPT)] and the process of lipid peroxidation (LPO) in the hepatopancreas and muscle showed no significant alterations in 10–60 mg kg−1 ZnNP supplemented feed fed PL. Whereas, 80 mg ZnNPs kg−1 supplemented feed fed PL showed significant elevations in SOD, CAT, LPO, GOT and GPT. Therefore, 80 mg ZnNPs kg−1 was found to be toxic to M. rosenbergii PL. Thus, the study suggests that up to 60 mg ZnNPs kg−1 can be supplemented for regulating survival, growth and immunity of M. rosenbergii.
Similar content being viewed by others
References
Salgueiro MJ, Zubillaga M, Lysionek A, Sarabia MI, Caro R, Paoli DT, Hager A, Weill R, Boccio J (2000) Zinc as an essential micronutrient: a review. Nutr Res 20:737–755
Tan B, Mai K (2001) Zinc methionine and zinc sulfate as sources of dietary zinc for juvenile abalone, Haliotis discus hannai Ino. Aquaculture 192:67–84
Gammanpila M, Age AY, Bart AN (2007) Evaluation of the effects of dietary vitamin C, E and Zinc supplementation on reproductive performance of Nile tilapia (Oreochromis niloticus). Sri Lanka Jo Aquat Sci 12:39–60
Herland H, Cooper M (2011) Effects of dietary mineral supplementation on quality of fresh and salt-cured fillets from farmed Atlantic cod Gadus morhua. J World Aquacult Soc 42:261–267
Davis DA, Lawrence AL, Galtin DM III (1993) Evaluation of the dietary zinc requirement of Penaeus vannamei and effects of phytic acid on zinc and phosphorus bioavailability. J World Aquacult Soc 24:40–47
Shiau SY, Jiang LC (2006) Dietary zinc requirements of grass shrimp, Penaeus monodon, and effects on immune responses. Aquaculture 254:476–482
Rani B, Hasnat A, Kohli MPS, Chandraprakash G (2012) Zinc supplementation and its effect on thermal stress resistance in Carassius auratus Fry. Isr J Aquacult Bamidgeh 64:779–786
Manusadzianas L, Grigutyte R, Jurkoniene S, Karitonas R, Sadauskas K, Ferard JF, Cotelle S, Foucaud L (2009) Toxicity of zinc oxide nanoparticle suspensions to aquatic biota. 14th international Symposium on Toxicity and Assement (ISTA-14).University of Metz France
**ong D, Fang T, Yu L, Sima X, Zhu W (2011) Effects of nano-scale TiO2, ZnO and their bulk counterparts on zebrafish: acute toxicity, oxidative stress and oxidative damage. Sci Total Environ 409:1444–1452
Hao L, Chen L, Hao J, Zhong N (2013) Bioaccumulation and sub-acute toxicity of zinc oxide nanoparticles in juvenile carp (Cyprinus carpio): a comparative study with its bulk counterparts. Ecotoxicol Environ Saf 91:52–60
Rather MA, Sharma R, Aklakur M, Ahmad S, Kumar N, Khan M, Ramya VL (2011) Nanotechnology, a novel tool for aquaculture and fisheries development a prospective mini-review. Fish Aquacult J 16:1–5
Zhou X, Wang Y, Gu Q, Li W (2009) Effects of different dietary selenium sources (selenium nanoparticle and selenomethionine) on growth performance muscle composition and glutathione peroxidase enzyme activity of crucian carp (Carassius auratus gibelio). Aquaculture 29:78–81
AOAC (1995) Official methods of analysis, 16th edn. Arlinton, VA, AOAC International
Tekinay AA, Davies SJ (2001) Dietary carbohydrate level influencing feed intake, nutrient utilization and plasma glucose concentration in the rainbow trout, Oncorhynchus mykiss. Tur J Vet Anim Sci 25:657–666
Furne M, Hidalgo MC, Lopez A, Garcia-Gallego M, Morales AE, Domenzain A, Domezain J, Sanz A (2005) Digestive enzyme activities in Adriatic sturgeon Acipenser naccarii and rainbow trout Oncorhynchus mykiss A comparative study. Aquaculture 250:391–398
Bernfeld P (1955) Amylases alpha and beta. Methods Enzymol 1:149–158
Lowry OH, Rosenbrough WJ, Fair AL, Randall RJ (1951) Protein measurement with the folinphenol reagent. J Biol Chem 193:265–275
Moore S, Stein WH (1948) Photometric ninhydrin method for use in the chromatography of amino acid. J Biol Chem 176:367–388
Roe JH (1955) The determination of sugar and blood and spinal fluid with anthrone reagent. J Biol Chem 212:335–343
Folch J, Lees M, Bloane-Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 266:497–509
Barnes H, Black Stock J (1973) Estimation of lipids in marine animals and tissues Detailed investigation of the Sulphophosphovanillin method for total lipids. J Exper Mar Biol Ecol 12:103–118
Tsing A, Arcier JM, Brehelin M (1989) Hemocytes of penaeid and palaemonid shrimps: morphology, cytochemistry and hemograms. J Invert Pathol 53:64–77
Kakkar R, Kalra J, Mantha SV, Prasad K (1995) Lipid peroxidation and activity of antioxidant enzymes in diabetic rats. Mol Cell Biochem 151:113–119
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47:389–394
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Reitman S, Frankel S (1957) A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol 28:56–63
Ho E (2004) Zinc deficiency, DNA damage and cancer risk. J Nutr Biochem 15:572–578
Tapiero H, Tew KD (2003) Trace elements in human physiology and pathology: zinc and metallothioneins. Biomed Pharmacother 57:399–411
Hedemann MS, Jensen BB, Poulsen HD (2006) Influence of dietary zinc and copper on digestive enzyme activity and intestinal morphology in weaned pigs. J Anim Sci 84:3310–3320
Ramseyer L, Garling D, Hill G, Link J (1999) Effect of dietary zinc supplementation and phytase pre- treatment of soybean meal or corn gluten meal on growth, zinc status and zinc-related metabolism in rainbow trout, Oncorhynchus mykiss. Fish Physiol Biochem 20:251–261
Satoh S, Izume K, Takeuchi T, Watanabe T (1987) Availability to rainbow trout of zinc contained in various types of fish meals. Nippon Suisan Gakkaishi 53(1861–1):866
Mohanty M, Adhikari S, Mohanty P, Sarangi N (2009) Effect of waterborne zinc on survival, growth, and feed intake of Indian major carp, Cirrhinus mrigala (Hamilton). Water Air Soil Pollut 201:3–7
Li WW, Gong YN, ** XK, He L, Jiang H, Ren F, Wang Q (2010) The effect of dietary zinc supplementation on the growth hepatopancreas fatty acid composition and gene expression in the Chinese mitten crab Eriocheir sinensis (H Milne-Edwards) (Decapoda, Grapsidae). Aquacult Res 41:828–837
Li JS, Li JL, Wu TT (2007) The effects of copper, iron and zinc on digestive enzymeactivity in the hybrid tilapia Oreochromis niloticus (L.) Oreochromis aureus (Steindachner). J Fish Biol 71:1788–1798
**g MY, Sun JY, Weng XY, Wang JF (2009) Effects of zinc levels on activities of gastrointestinal enzymes in growing rats. J Anim Physiol Anim Nutr 93:606–612
Lovett DL, Felder DL (1990) Ontogenic change in digestive enzyme activityof larval and postlarval white shrimp Penaeus setiferus (Crustacea, Decapoda, Penaeidae). Biol Bull 178:144–159
Lall SP (2002) The minerals. In: Halver JE, Hardy RD (eds) Fish nutrition, 3rd edn. Academic Press, New York, pp 259–308
Gatlin DM III, Wilson RP (1984) Dietary selenium requirements of fingerling channel catfish. J Nutr 114:627–633
Ward JD, Spears JW, Kegley EB (1993) Effect of copper level and source (copper lysine vs copper sulfate) on copper status performance and immune response in growing steers fed diets with or without supplemental molybdenum and sulfur. J Anim Sci 71:2748–2755
Ates A, Daniels J, Arslan Z, Farah IO, Rivera HF (2013) Comparative evaluation of impact of Zn and ZnO nanoparticles on brine shrimp (Artemia salina) larvae: effects of particle size and solubility on toxicity. Environ Sci Process Impact 15:225–233
Acknowledgments
Bharathiar University, Coimbatore, Tamil Nadu, India, is gratefully acknowledged for the financial support provided in the form of University Research Fellowship to the first author. The University Grants Commission, Government of India, New Delhi, is also gratefully acknowledged for the acquired laboratory facility by the second author through a Major Research Project operated (2009–2012) on prawn nutrition.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muralisankar, T., Bhavan, P.S., Radhakrishnan, S. et al. Dietary Supplementation of Zinc Nanoparticles and Its Influence on Biology, Physiology and Immune Responses of the Freshwater Prawn, Macrobrachium rosenbergii . Biol Trace Elem Res 160, 56–66 (2014). https://doi.org/10.1007/s12011-014-0026-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0026-4