Abstract
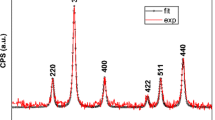
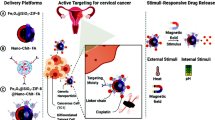
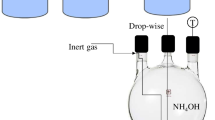
Magnetite nanoparticles are particularly attractive for drug delivery applications because of their size-dependent superparamagnetism, low toxicity, and biocompatibility with cells and tissues. Surface modification of iron oxide nanoparticles with biocompatible polymers is potentially beneficial to prepare biodegradable nanocomposite-based drug delivery agents for in vivo and in vitro applications. In the present study, the bare (10 nm) and polyethylene glycol (PEG)–(3-aminopropyl)triethoxysilane (APTES) (PA) modified (17 nm) superparamagnetic iron oxide nanoparticles (SPIO NPs) were synthesized by coprecipitation method. The anticancer drugs, doxorubicin (DOX) and paclitaxel (PTX), were separately encapsulated into the synthesized polymeric nanocomposites for localized targeting of human ovarian cancer in vitro. Surface morphology analysis by scanning electron microscopy showed a slight increase in particle size (27 ± 0.7 and 30 ± 0.45 nm) with drug loading capacities of 70 and 61.5 % and release capabilities of 90 and 93 % for the DOX- and PTX-AP-SPIO NPs, respectively (p < 0.001). Ten milligrams/milliliter DOX- and PTX-loaded AP-SPIO NPs caused a significant amount of cytotoxicity and downregulation of antiapoptotic proteins, as compared with same amounts of free drugs (p < 0.001). In vivo antiproliferative effect of present formulation on immunodeficient female Balb/c mice showed ovarian tumor shrinkage from 2,920 to 143 mm3 after 40 days. The present formulation of APTES–PEG-SPIO-based nanocomposite system of targeted drug delivery proved to be effective enough in order to treat deadly solid tumor of ovarian cancer in vitro and in vivo.








Similar content being viewed by others
References
Jeun, M., Lee, S., Kang, J. K., Tomitaka, A., & Kang, K. W. (2012). Physical limits of pure superparamagnetic Fe3O4 nanoparticles for a local hyperthermia agent in nanomedicine. Applied Physics Letters, 100, 092406.
Salehizadeh, H., Hekmatian, H., Sadeghi, M., & Kennedy, K. (2012). Synthesis and characterization of core-shell Fe3O4-gold-chitosan nanostructure. Journal of Nanobiotechnology, 10, 3–9.
Cai, H., Xu, C., He, P., & Fang, Y. (2001). Nanoparticles: from theory to applications. Electroanalytical Chemistry, 510, 78–85.
Wu, W., & Jiang, X. (2012). Long-circulating polymeric drug nanocarriers. Functional nanoparticles for bioanalysis, nanomedicine, and bioelectronic devices. Volume 2, ACS Symposium Series, Vol. 1113, ISBN: 9780841228283, Chapter 2, pp 27–36. 4, 5.
Sun, C., Jerry, S., Lee, H., & Zhang, M. (2008). Magnetic nanoparticles in MR imaging and drug delivery. Advanced Drug Delivery Reviews, 60, 1252–1265.
Neuberger, T., Schopf, B., Hofmann, H., Hofmann, H., & von Rechenberg, B. (2005). Superparamagnetic nanoparticles for biomedical applications: Possibilities and limitations of a new drug delivery system. Journal of Magnetism and Magnetic Materials, 293, 483–496.
Torchilin, V. P. (2006). Multifunctional nanocarriers. Advanced Drug Delivery Reviews, 58, 1532–1555.
Zhang, G., Lai, B. B., Zhou, Y. Y., Chen, B. A., & Wang, A. M. (2011). Fe3O4 nanoparticles with daunorubicin induce apoptosis through caspase 8-PARP pathway and inhibit K562 leukemia cell-induced tumor growth in vivo. Nanomedicine: Nanotechnology, Biology, and Medicine, 7, 595–603.
Biranwar, Y. A., Bare, K. R., Mahajan, V. R., Sapate, M. K., & Baviskar, D. T. (2011). Erythrocyte drug delivery system. International Journal of Pharmaceutical Science Review and Research, 8, 124–128.
Inui, O., Teramura, Y., & Iwata, H. (2010). Retention dynamics of amphiphilic polymers PEG-lipids and PVA-Alkyl on the cell surface. ACS Applied Material Interfaces, 2, 1514–1520.
Chao, Y., Su, A., Lin, Y., Hsu, W., & Huang, K. (2011). Synthesis and application of polyethylene glycol/vinyltriethoxy silane (PEG/VTES) copolymers. African Journal of Biotechnology, 10, 12754–12761.
Kitagawa, K., Kubota, K., Sueyoshi, K., & Otsuka, O. (2010). One-step preparation of amino-PEG modified poly(methyl methacrylate) microchips for electrophoretic separation of biomolecules. Journal of Pharmaceutical Biomedicine and Analysis, 53, 1272–1277.
Cole, A. J., David, A. E., Wang, J., Galbán, C. J., Hill, H. L., & Yang, V. C. (2011). Polyethylene glycol modified, cross-linked starch coated iron oxide nanoparticles for enhanced magnetic tumor targeting. Biomaterials, 32, 2183–2193.
**e, J., Xu, C., Kohler, N., Hou, Y., & Sun, S. (2007). Controlled PEGylation of monodisperse Fe3O4 nanoparticles for reduced non-specific uptake by macrophage cells. Advanced Materials, 19, 3163–3166.
Zhang, Y., Kohler, N., & Zhang, M. (2002). Surface modification of superparamagnetic magnetite nanoparticles and their intracellular uptake. Biomaterials, 23, 1553–1561.
Yue-Jian, C., Juan, T., Fei, X., Jia-Bi, Z., Ning, G., & Yi-Hua, Z. (2010). Synthesis, self-assembly, and characterization of PEG-coated iron oxide nanoparticles as potential MRI contrast agent. Drug Development and Industrial Pharmacy, 36, 1235–1244.
Chen, Y. J., Tao, J., **ong, F., Zhu, J. B., Gu, N., & Geng, K. K. (2010). Characterization and in vitro cellular uptake of PEG coated iron oxide nanoparticles as MRI contrast agent. Pharmazie, 65, 481–486.
Mikhayaylvo, M., Kim, D. K., & Berry, C. C. (2004). BSA immobilization on amine functionalized superparamagnetic iron oxide nanoparticles. Chemical Materials, 16, 2344–2354.
Naseri, G. M. (2012). The amounts of loaded DOX and PTX were 35 and 30.75 mg/g SPIO NPs. Carbohydrate Polymers, 90, 1265–1272.
Jain, K. K. (2005). The role of nanobiotechnology in drug discovery. Drug Discovery Today, 10, 1435–1442.
Wang, H., Shrestha, T. B., Basel, M. T., Dani, R. K., Seo, G., & Balivada, S. (2012). Magnetic-Fe/Fe3O4-nanoparticle-bound SN38 as carboxylesterase-cleavable prodrug for the delivery to tumors within monocytes/macrophages. Beilstein Journal of Nanotechnology, 3, 444–455.
Mahmoudi, M., Simchi, A., Imani, M., Milani, A. S., & Stroeve, P. (2009). An in vitro study of bare and poly(ethylene glycol)-co-fumarate-coated superparamagnetic iron oxide nanoparticles: a new toxicity identification procedure. Nanotechnology, 20, 225104.
Fonseca, C., Simoes, S., & Gaspar, R. (2002). Paclitaxel-loaded PLGA nanoparticles: preparation, physicochemical characterization and in vitro anti-tumoral activity. Journal of Controlled Release, 82, 273–286.
Wang, Q., Zheng, X., Yang, L., Shi, Y., Gao, L., & Zhong, L. (2010). Reactive oxygen species-mediated apoptosis contributes to chemosensitization effect of saikosaponins on cisplatin-induced cytotoxicity in cancer cells. Journal of Experimental and Clinical Cancer Research, 29, 159–167.
Lane, D., Côté, M., & Grondin, R. (2006). Acquired resistance to TRAIL-induced apoptosis in human ovarian cancer cells is conferred by increased turnover of mature caspase-3. Molecular Cancer Therapy, 5, 509–521.
Vanderhyden, B. C., Shaw, T. J., & Ethier, J. (2003). Animal models of ovarian cancer. Reproductive Biology and Endocrinology, 1, 67–77.
Ferrari, M. (2005). Cancer nanotechnology: opportunities and challenges. Nature Reviews Cancer, 5, 161–171.
Pardakhty, A., & Moazeni, E. (2013). Nano-niosomes in drug, vaccine and gene delivery: a rapid overview. Nanomedicine Journal, 1, 1–13.
Akbarzadeh, A., Mikaeili, H., Zarghami, N., Rahmati, M., Barkhordari, A., & Davaran, S. (2012). Preparation and in vitro evaluation of doxorubicin-loaded Fe3O4 magnetic nanoparticles modified with biocompatible copolymers. International Journal of Nanomedicine, 7, 144–156.
Brullot, W., Reddy, N. K., Wouters, J., Valev, V. K., Goderis, B., & Vermant, J. (2012). Versatile ferrofluids based on polyethylene glycol coated iron oxide nanoparticles. Journal of Magnetism and Magnetic Materials, 324, 1919–1925.
Gubin, S. P. (2009). Magnetic Nanoparticles. Hoboken: Wiley.
Sah, H. (1999). Stabilization of protein against methylene chloride water interface-induced denaturation and aggregation. Journal of Controlled Release, 58, 143–151.
Yoo, H. S., & Park, T. G. (2000). In vitro and in vivo anti-tumor activities of nanoparticles based on doxorubicin-PLGA conjugates. Journal of Controlled Release, 68, 419–431.
Lu, A. H., Salabas, E. L., & Schuth, F. (2007). Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angewandte Chemie International Edition, 46, 1222–1244.
Wang, L. Y., Bai, J. W., Li, Y. J., & Huang, Y. (2008). Multifunctional nanoparticles displaying magnetization and near-IR absorption. Angewandte Chemie International Edition, 47, 2439–2442.
Hu, L., McArthur, C., & Jaffe, R. B. (2010). Ovarian cancer stem-like side-population cells are tumourigenic and chemoresistant. British Journal of Cancer, 102, 1276–1283.
Tsuruta, Y., Mandai, M., & Konishi, I. (2001). Combination effect of adenovirus-mediated pro-apoptotic bax gene transfer with cisplatin or paclitaxel treatment in ovarian cancer cell lines. European Journal of Cancer, 37, 531–541.
Muscolini, M., Cianfrocca, R., & Sajeva, A. (2008). Trichostatin A up-regulates p73 and induces Bax-dependent apoptosis in cisplatin-resistant ovarian cancer cells. Molecular Cancer Therapeutics, 7, 1410–1419.
Villedieu, M., Louis, M. H., & Dutoit, S. (2007). Absence of Bcl-xL downregulation in response to cisplatin is associated with chemoresistance in ovarian carcinoma cells. Gynecologic Oncology, 105, 31–44.
Hine, C. M., Seluanov, A., & Gorbunova, V. (2012). Rad51 promoter-targeted gene therapy is effective for in vivo visualization and treatment of cancer. Molecular Therapy, 20, 347–355.
Acknowledgments
The authors are grateful for the financial support of the Research Council of the University of Tehran, Tehran, Iran, to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Javid, A., Ahmadian, S., Saboury, A.A. et al. Biocompatible APTES–PEG Modified Magnetite Nanoparticles: Effective Carriers of Antineoplastic Agents to Ovarian Cancer. Appl Biochem Biotechnol 173, 36–54 (2014). https://doi.org/10.1007/s12010-014-0740-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0740-6