Abstract
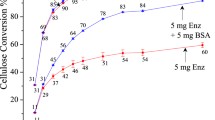
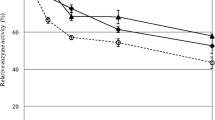
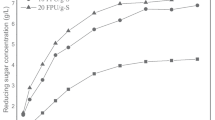
Bovine serum albumin (BSA) was added to filter paper during the hydrolysis of cellulase. Adding BSA before the addition of the cellulase enhances enzyme activity in the solution, thereby increasing the conversion rate of cellulose. After 48 h of BSA treatment, the BSA adsorption quantities are 3.3, 4.6, 7.8, 17.2, and 28.3 mg/g substrate, each with different initial BSA concentration treatments at 50 °C; in addition, more cellulase was adsorbed onto the filter paper at 50 °C compared with 35 °C. After 48 h of hydrolysis, the free-enzyme activity could not be measured without the BSA treatment, whereas the remaining activity of the filter paper activity was approximately 41 % when treated with 1.0 mg/mL BSA. Even after 96 h of hydrolysis, 25 % still remained. Meanwhile, after 48 h of incubation without substrate, the remaining enzyme activities were increased 20.7 % (from 43.7 to 52.7 %) and 94.8 % (from 23.3 to 45.5 %) at 35 and 50 °C, respectively. Moreover, the effect of the BSA was more obvious at 35 °C compared with 50 °C. When using 15 filter paper cellulase units per gram substrate cellulase loading at 50 °C, the cellulose conversion was increased from 75 % (without BSA treatment) to ≥90 % when using BSA dosages between 0.1 and 1.5 mg/mL. Overall, these results suggest that there are promising strategies for BSA treatment in the reduction of enzyme requirements during the hydrolysis of cellulose.







Similar content being viewed by others
References
Mansfield, S. D., Mooney, C., & Saddler, J. N. (1999) Substrate and enzyme characteristics that limit cellulose hydrolysis. Biotechnology Progress, 15, 804–816.
Lynd, L. R., Weimer, P. J., Van Zyl, W. H., & Pretorius, I. S. (2002). Microbial cellulose utilization: fundamentals and biotechnology. Microbiology and Molecular Biology Reviews, 66, 506–577.
Lynd, L. R., Laser, M. S., Bransby, D., Dale, B. E., Davison, B., Hamilton, R., et al. (2008). How biotech can transform biofuels. Nature Biotechnology, 26, 169–172.
Wyman, C. E. (1999). Biomass ethanol: technical progress, opportunities, and commercial challenges. Annual Review of Energy and the Environment, 24, 189–226.
McMillan, J. D. (1994). Pretreatment of lignocellulosic biomass. ACS symposium series. ACS Publications, pp. 292–324.
Castanon, M., & Wilke, C. R. (1981). Effects of the surfactant Tween 80 on enzymatic hydrolysis of newspaper. Biotechnology and Bioengineering, 23, 1365–1372.
Seo, D. J., Fujita, H., & Sakoda, A. (2011). Effects of a non-ionic surfactant, Tween 20, on adsorption/desorption of saccharification enzymes onto/from lignocelluloses and saccharification rate. Adsorption, 17, 813–822.
Eriksson, T., Börjesson, J., & Tjerneld, F. (2002). Mechanism of surfactant effect in enzymatic hydrolysis of lignocellulose. Enzyme and Microbial Technology, 31, 353–364.
Kaar, W. E., & Holtzapple, M. T. (1998). Benefits from Tween during enzymic hydrolysis of corn stover. Biotechnology and Bioengineering, 59, 419–427.
Park, J. W., Takahata, Y., Kajiuchi, T., & Akehata, T. (1992). Effects of nonionic surfactant on enzymatic hydrolysis of used newspaper. Biotechnology and Bioengineering, 39, 117–120.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
Pan, X., **e, D., Gilkes, N., Gregg, D. J., & Saddler, J. N. (2005). Strategies to enhance the enzymatic hydrolysis of pretreated softwood with high residual lignin content. Applied Biochemistry and Biotechnology, 124, 1069–1079.
Kim, M. H., Lee, S. B., Ryu, D. D. Y., & Reese, E. T. (1982). Surface deactivation of cellulase and its prevention. Enzyme and Microbial Technology, 4, 99–103.
Howell, J. A., & Mangat, M. (1978). Enzyme deactivation during cellulose hydrolysis. Biotechnology and Bioengineering, 20, 847–863.
Klyosov, A. A., Rabinowitch, M. L. (1980). Enzymatic conversion of cellulose to glucose: present state of the art and potential. Enzyme Engineering: Future Directions, 83–165.
Ooshima, H., Sakata, M., & Harano, Y. (1986). Enhancement of enzymatic hydrolysis of cellulose by surfactant. Biotechnology and Bioengineering, 28, 1727–1734.
Zheng, Y., Pan, Z., Zhang, R., Wang, D., & Jenkins, B. (2008). Non-ionic surfactants and non-catalytic protein treatment on enzymatic hydrolysis of pretreated cree** wild ryegrass. Applied Biochemistry and Biotechnology, 146, 231–248.
Acknowledgments
This work was supported by a grant from the National High Technology Research and Development Program of China (863 Program; no. 2012AA101803) and JST/JICA-SATREPS, titled “Sustainable Integration of Local Agriculture and Biomass Industries.”
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, H., Mochidzuki, K., Kobayashi, S. et al. Effect of Bovine Serum Albumin (BSA) on Enzymatic Cellulose Hydrolysis. Appl Biochem Biotechnol 170, 541–551 (2013). https://doi.org/10.1007/s12010-013-0208-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0208-0